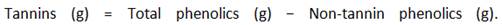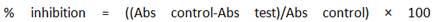Introduction
The consumption of herbal medicines is increasing over the world, primarily due to its benefits. The World Health Organization estimates, that nearly 80 % of the world’s population in developing countries relies primarily on herbal medicine to satisfy their healthcare needs.1 Besides, in recent years, compounds derived from medicinal and aromatics plants have been commercially exploited by the pharma, food, flavor, fragrance, dyeing and pesticide industries.
Among the various biological activities reported for plants, the antioxidant stands out, related to the need to have protectors against oxidative stress. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are continuously generated under physiological conditions and are involved in the growth, differentiation, progression and death of cells. Low concentrations of these species are beneficial and even indispensable for processes such as intracellular signaling and defense against microorganisms.2 However, when the body's natural antioxidant defenses are overcome by excessive production of ROS/RNS, the so-called "oxidative stress" occurs. In this state, cellular and extracellular macromolecules (lipids, proteins and nucleic acids) can suffer oxidative damage, causing harm to different tissues.
Because in certain occasions the innate defenses are not enough against a severe or continuous oxidative stress, it is necessary to supply certain amounts of exogenous antioxidants to maintain an adequate value of them in order to balance ROS/RNS levels. Because of this, there has been an increase in interest regarding natural antioxidants, such as polyphenols, present in medicinal and edible plants, which could help in the prevention of oxidative stress.
The Polygonaceae are a family of plants belonging to the order Caryophyllales, and comprise approximately 1 200 species distributed in approximately 48 genera. The largest genera are Eriogonum (240 species), Rumex (200 species), Coccoloba (120 species), Persicaria (100 species) and Calligonum (80 species). The family is present all over the world, but it is more diverse in the Northern Temperate Zone.
The genus Coccoloba comprises approximately 120-150 shrubs and trees, mostly perennialsflowering plants, of which more than 25 occurs in Cuba. It is native to the tropical and subtropical regions of America, in South America, the Caribbean and Central America, with two species that extend to Florida.
Biological assays revealed antifungic activity in C. acrosticoides3 and C. dugandiana4, antibacterial in C. crosticoides3 and C. Cozumelensis5 and alelochemical in C. uvifera.6 In relation to the chemical profile of the genus Coccoloba, the species until now investigated are represented by flavonoids 4,7, terpenoids 3,8, benzenoids 4 and carboxylic acids and esters.9
C. cowellii, a critically endangered plant, endemic of serpentinitic savannas of Camagüey province, is locallyknown as "Uverillo" and "Moco de guanajo”. In the scientific literature, there are no publications related to the phytochemistry and biological activity of this specie. Taking into account endemism of C. cowellii and biological activities and chemical composition reported in allied species, this work is aimed to determine and quantify the phenolics content of the leaves of C. cowellii and to evaluate its antioxidant properties in vitro.
Materials and methods
Plant Material
Leaves of C. cowellii (fig. 1) were collected in april 2018 near to Albaisa, in the municipality of Camagüey (Lat 21.43615, Lon -77.83253). Plant was taxonomically identified by the curator of “Julián Acuña Galé” herbarium at the University of Camagüey (HIPC), where a voucher specimen was deposited (number 12057).
Plantmaterial after cleaned was dried on the shadow at room temperature until constant weight. Afterward, it was milled in a blade mill and stored in the dark at a dry place until further use.
Chemical reagents
Folin-Ciocalteu, DPPH (1,1-diphenyl-2-picryl-hydrazyl), ferric chloride, gelatin and quercetin reagents were purchased from Sigma(USA). Tannic acid was from BDH Laboratory reagents (England). All other used reagents were purchased from UNI-CHEM Chemical Reagents (China). All solvents used for extraction and chromatography were of analytical purity grade.
Extracts preparation
A dynamic ultrasound-assisted extraction device, based on the method suggested by Xungang et al.10 with some modifications was constructed for the extraction. Five grams of plant material were filled into extraction cell (9 x 2 cm, i.d.). The column was prepared by adding a small plug of cotton, plant material, and suitable amount of cottonas shown in fig. 2, to form an extraction column. The column was set in an ultrasonic cleaner from Scientz, model SB-3200STD (China), vibration frequency at 40 kHz, 80 % power, with inlet connected to a peristaltic pump by a tube, and outlet to a flask. Dynamic ultrasound-assisted extraction was carried out by continuously feeding extraction solvent into the column by the pump and assisting by ultrasonic wave, where a flow rate of 3 mL/min was used, and temperature was set at 40 ºC by adjusting the regulation of ultrasonic bath. The plant material was firstly defatted with 1 mL of ethyl ether: petroleum ether (1:1, v/v) for 1 min and dynamically extracted for 35 min. Then a second dynamic extraction was performed using aqueous ethanol (8:2, v/v) for 35 min to obtain the ethanolic extract. After the procedure, the fraction of the effluent (≈100 mL) was collected into 150 mL of volumetric flask.
Qualitative phytochemical studies
The ethanolic extract was subjected to qualitative phytochemical analysis. Presence of various phytochemicals such as phenols, steroids, glycosides, saponins, flavonoids, terpenoids, alkaloids and reducing sugars were determined by various phytochemical tests.11
Quantification of Total Phenolic content (TPC)
The total phenol concentration of the sample was determined using the Folin-Ciocalteu method described by Makkar.12 Briefly, 50 μL of ethanolic extract in 950 μL of methanol were mixed with 1,0 mL of distilled water and 0,5 mL of Folin-Ciocaleu reagent (1N) in a test tube. After vortexed the tube and allowed to stand for 5 min at room temperature, 2,5 mL of 5 % aqueous sodium carbonate were added, the tube was vortexed again and incubated for 40 min in a dark place. The absorbance was measured at 725 nm using a UV/Vis spectrophotometer from Rayleigh, model UV-1601 (China). A standard curve using tannic acid (TA) was created with 5 points. Total phenolic concentration was expressed as tannic acid equivalent in mg per gram of defatted extract (mg TA/g extract). The methanol solution was used as a blank. All assays were carried out in triplicate.
Quantification of Total Tannin Content (TTC)
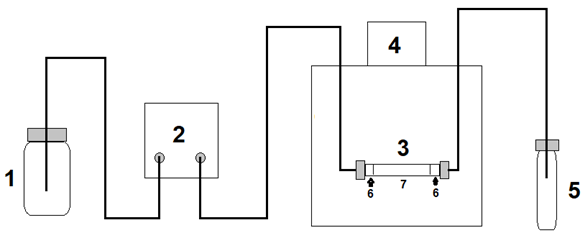
The gelatin reagent for the precipitation of tannins was prepared according to Velázquez 13, mixing in a volumetric flask 5 mL of the 10 % gelatin solution and 10 mL of 10 % NaCl solution in 1 % HCl. The mixture was stirred well and adjusted to 50 mL with distilled water, then allowed to stand for 30 minutes to allow the insoluble solids to settle, filtered and the filtered solution was used as a reagent for the analysis of total tannins. After that, 500 μL of the ethanolic extract was mixed with 500 μL of the gelatin reagent and incubated for 30 minutes in a water bath at 37 °C, then centrifuged at 5 000 rpm for 10 min using a centrifuge from Yingtai Instrument, model TG16 (China), and 500 μL of the supernatant was taken for the determination of total phenols by the method of Folin-Ciocalteu as previously described.12 The total content of tannins was calculated using the following formula:
Quantification of Total Flavonoid Content (TFC)
The amount of total flavonoids in the extracts was measured spectrophotometrically as previously reported.14 Briefly, 500 μL of the extract were mixed with 1,50 mL of 95 % ethanol, 0,10 mL of 10 % aluminum chloride (AlCl3.6H2O), 0,10 mL of sodium acetate (NaC2H3O2) (1 M) and 2,80 mL of distilled water. After incubation for 40 min, absorbance was measured at 415 nm using a UV/Vis spectrophotometer from Rayleigh, model UV-1601 (China). To calculate the concentration of flavonoids, a calibration curve was prepared using quercetin (QE) as standard. The flavonoid concentration is expressed as quercetin equivalents in mg per gram of defatted extract (mg QE/g extract). All assays were carried out in triplicate.
Evaluation of antioxidant activity
Free radical-scavenging ability by the use of a stable DPPH radical
The DPPH radical-scavenging activity was determined using the method described by Blois 15 with slightly modifications. Briefly, 5 mL of a 0,1 mM DPPH solution was mixed with sample solutions at different concentrations (10, 20, 30, 40, 50 µg/mL). A control (Abs Control) containing methanol and DPPH solution was also prepared. The mixture of methanol, DPPH and ascorbic acid served as positive control. All solutions obtained were then incubated for 20 min at room temperature. The radical scavenging capacity was evaluated by measuring the decrease of absorbance at 517 nm using a UV/Vis spectrophotometer from Rayleigh, model UV-1601 (China). The percentage of inhibition of samples was calculated from obtained absorbances by the equation:
Then, a curve was constructed by plotting percentage of inhibition against concentration in µg/mL. All assays were carried out in triplicate.
TLC-DPPH test
In order to relate specific compounds with the antioxidant activity, the assay previously described was develop on a Thin Layer Chromatography plate (TLC). The stable 2,2-diphenyl-1-picrylhydrazyl radical (DPPH) has an absorption maximum at 517 nm, which decreases upon reduction through reaction with a radical scavenger. The corresponding color change can thus be observed in a TLC bioassay.(16) The TLC plate with the sample was developed with the elution solvent (chloroform:ethyl acetate:methanol 2:3:1, v/v/v) and then dried. It was then sprayed with a solution of 0,2 % (DPPH) in methanol. The plate was examined in daylight after 30 min. Active (free-radical scavenging) compounds appear as yellow-white spots against a purple background. To compare, another two plates were developed with the same solvent, dried and revealed with 254 nm UV light and sulfuric acid-vanillin reagent, respectively.
Reducing power ability
The reducing power of plant extracts can be detected based on the ability to reduce ferric ions in the reaction mixture to ferrous ions. The ferricyanide reagent is first incubated in phosphate buffer at pH 6.6 with antioxidants, and the reduction product, ferrocyanide, combines with the later added Fe3+ to produce Prussian blue, which is detected at 700 nm. The assay is based in the following chemical reactions:
The reduction will favor the production of thecolored complex where the intensity is dependent on the concentration of reductants. Reducing power was determined using the method described by Oyaizu 17 with slightly modifications. Briefly, sample solutions at different concentrations (10, 20, 30, 40, 50 µg/mL) were mixed with 2,5 mL of 0,2 M phosphate buffer (pH 6,6) and 2,5 mL of 1 % potassium ferricyanide and incubated at 50 ºC for 20 min. Then, 2.5 mL of 10% trichloroacetic acid were added, and the tubes were centrifuged at 10,000 rpm for 10 min using a centrifuge from Yingtai Instrument, model TG16 (China). Then, 2.5 mL of the upper layer were mixed with 2,5 mL of distilled water and 0,5 mL of 0,1 % ferric chloride, and the absorbance of the reaction mixtures was measured at 700 nm using a UV/Vis spectrophotometer from Rayleigh, model UV-1601 (China). Ascorbic acid was used as a positive control. All assays were carried out in triplicate.
Results and discussion
Preliminary phytochemical tests for the leaves extract of Coccoloba cowellii
The qualitative analysis of the ethanol extract is showed in table 1. The main phytochemical constituents of the extract were the phenolic compounds, represented by tannins and flavonoids, and in a minor degree, reducing sugars. The phenolics compounds are present in the polar extracts of other species of Coccolobagenus, specifically tannins (both condensed and hydrolysables) and flavonoids glucosides of quercetin, catechins and myricetin.3,18
Table 1 Phytochemical screening of the ethanolic extract of Coccoloba cowellii
| Chemical constituents | Results of chemical reaction |
|---|---|
| Alkaloids | - |
| Triterpenes and steroids | - |
| Coumarins | - |
| Phenols and tannins | +++ |
| Free aminoacids | - |
| Cardiac glycosides | - |
| Flavonoids | ++ |
| Quinones | - |
| Reducing sugars | + |
| Saponins | - |
Precipitate or coloration: very abundant, +++; abundant, ++; middle, +; not detected,
- (the number of positive signs indicated the intensity of the reactions).
Phenolic, flavonoid and tannin contents
The total phenolic content of the ethanolic extract, calculated from the calibration curve (Y = 0,018 03*X + 0,013 8; R2 = 0,999) was 264,77 ± 5,47 tannic acid equivalents/g, the total tannin content (Y = 0,018 03*X + 0,013 8; R2 = 0,999) was 148,02 ± 2,63 tannic acid equivalents/g and the total flavonoid content (Y = 0,006 806*X + 0,474 7; R2 =0,983) was 177,04 ± 1,08 quercetin equivalents/g (table 2). These results were comparable to those obtained for the acetone extract of C. uviferaleaves 19, and demonstrate the high content of this type of secondary metabolites in the evaluated extract. No other scientific reports regarding the metabolites concentration of other species for this genus were found in the consulted literature.
Phenolic compounds have redox properties, which allow them to act as antioxidants.20 As their free radical scavenging ability is facilitated by their hydroxyl groups, the total phenolic concentration could be used as a basis for rapid screening of antioxidant activity. Antioxidant activity of flavonoids, including flavones, flavanols and condensed tannins, is related to the presence of free OH groups, especially 3-OH. Plant flavonoids have antioxidant activity in vitro and also act as antioxidants in vivo.21
Table 2 Total phenolics, tannins and flavonoids content of ethanolic extract of C. cowelliileaves
| Total phenolics contenta | 264,77 ± 5,47 |
| Total tannins contenta | 148,02 ± 2,63 |
| Total flavonoids contentb | 177,04 ± 1,08 |
amg tannic acid equivalent (TAE)/g DW.
bmg quercetin equivalent (QE)/g DW.
Values are mean ±standard deviation of three replicates.
Antioxidant activity
Several methods have been developed for the assessment of antioxidant capacity. Because many active species and reaction mechanisms are involved in oxidative stress processes, no simple universal method can be applied for an accurate and quantitative measurement of antioxidant capacity. Generally, in these methods, a radical is generated and the antioxidant capability of a sample against the radical is evaluated. In the present study, the antioxidant activity of C. cowellii ethanolic leaf extract was determined using the DPPH radical scavenging and the reducing power assay. In the present study, the antioxidant activity of C. cowellii ethanolic leaf extract was determined using the DPPH radical scavenging, alone and combined with TLC plates, and the reducing power assay.
DPPH free radical scavenging assay
The radical DPPH has the advantage of being unaffected by certain side reactions, such as metal-ion chelation and enzyme inhibition, brought about by various additives. A freshly prepared DPPH solution exhibits a deep purple color with a maximum absorption at 517nm.22 This purple color generally fades/disappears when an antioxidant is present in the medium. Thus, antioxidant molecules can quench DPPH free radicals (i.e., by providing hydrogen atoms or by electron donation, conceivably via a free-radical attack on the DPPH molecule) and convert them to a colorless/bleached product, DPPH-H (i.e., 2,2-diphenyl-1-hydrazine, or a substituted analogous hydrazine), resulting in a decrease in absorbance at 517nm.
It has been found that ascorbic acid, tocopherol and polyhydroxy aromatic compounds reduce and decolorize DPPH by their hydrogen donating ability.22 It appears that C. cowellii possesses hydrogen donating abilities and can act as an antioxidant through this pathway.DPPH scavenging activity of C. cowellii ethanolic extract was 34,01 % at a concentration of 50 µg/mL, while that of the control, ascorbic acid, was 29,35 % (fig. 4, a).These results were also comparable to those obtained for C. uvifera23, and showed that the extract exhibited an antioxidant potential comparable to the antioxidant capacity of commonly used ascorbic acid.
TLC-DPPH Test
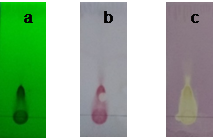
The results showed the presence of radical scavengers in the ethanolic extract as a yellow-white zone over a purple background (fig. 3, c). The same compounds can be visualized using 254 nm UV light (fig. 3, a) and the sulfuric acid-vanillin reagent (fig. 3, b). Taking into account the chromatographic behavior of the extract and the color developed with the sulfuric acid-vanillin reagent, the presence of proanthocyanidins or condensed tannins would be possible. Those images were taken from the one of the ten different solvent systems (data not showed) that displayed a better TLC resolution. Further analyses are necessary to corroborate this hypothesis.
Ferric-reducing power assay
The reducing power of a compound is related to its electron transfer ability and may serve as a significant indicator of its potential antioxidant activity. In this assay, the yellow color of the test solution changes to green and blue depending on the reducing power of test specimen. Greater absorbance at 700 nm indicates higher reducing power.
In the concentration range investigated, the extract demonstrated reducing power that increased linearly with concentration. Significant changes in absorbance at 700 nm were observed (0,43-0,63) with increasing concentrations of extract (10-50 µg/mL) (fig. 4, b). The reducing power of the C. cowellii leaves might be due to its hydrogen-donating ability.
The high phenolic and flavonoid content is possibly responsible for the antioxidant activity of thisextract. Flavonoids are highly effective scavengers of most oxidizing molecules, including singlet oxygen, and various other free radicals implicated in several diseases.24 Flavonoids suppress reactive oxygen formation, chelate trace elements involved in free-radical production, scavenge reactive species and up-regulate and protect antioxidant defenses.25 Similarly, phenolics compounds confer oxidative stress tolerance on plants. Crude extracts of fruits, herbs, vegetables, cereals and other plant materials rich in phenolics are increasingly being used by the food industry for their antioxidative properties and health benefits.
Conclusions
In vitro antioxidant activities of C. cowelliiare consistent with the presence of phenolic compounds, like flavonoids and tannins, and the results are similar to other species of the Coccoloba genus. Results suggested that C. cowellii is a potential source of antioxidant agents, possibly due to the presence of phenolic compounds. Therefore, it may be considered a viable specimen for vegetal biotechnology techniques of in vitro propagation. Nevertheless, more definitive phytochemical analysis must be required to isolate and to characterize plant metabolites that show the pharmacological effects. These results constitute the first report of the phytochemical composition and potential antioxidant activity of C. cowellii.