Tropics are abundant in forage resources that are feasible to use in animal feed. They have advantages from the agronomic and nutritional standpoint. In the international literature it has been reported a great number of resources including Legumes, which represent sources of protein and certain minerals and vitamins (Díaz et al. 2014). In this sense, the grains of Mucuna pruriens, a legume widely studied in the literature, is noted for having concentrations between 250-350 g crude protein (CP)/kg dry matter (DM) (Chikagwa et al. 2009), 31.2-39.5 % of starch (Vadivel and Pugalenthi 2010) and 2.74 % to 3.41 % of ash (Adebowale et al. 2005). However, their inclusion in non-ruminant feeds is limited due to the high content of fiber (NDF: 21.3 %) (Pugalenthi et al. 2005) and the presence of several antinutritional factors (ANF) (Safwat et al. 2014).
In order to reduce some of these limitations, different processing methods such as seed germination, shelling, roasting, soaking in water or alkaline solutions and cooking have been used (Chaparro et al. 2009). It is necessary to know its chemical composition and digestibility to be recommended as animal food. However, the in vivo determination of digestibility is laborious and costly process and requires the use of large quantities of food, then various methods have been proposed for the in vitro estimation (Lemos and Nunes 2008). Among them, pH-stat method has been used to analyze the degree of hydrolysis of different substrates with satisfactory results (Rutherfurd 2010). Itzá (2012) studied the protein digestibility of four autoclaved legumes and concluded that the method was reliable for predicting the digestive utilization of proteins of legumes as compared to an in vivo assay in mice.
Considering the above, the objective of this study was to determine the chemical composition and in vitro protein digestibility of processed Mucuna pruriens seeds.
Materials and Methods
The experiment was conducted at the Animal Nutrition laboratory of the Faculty of Veterinary Medicine and Animal Science, University of Yucatan, Merida, Mexico. The experimental treatments consisted of 500 g of seeds Mucuna pruriens var. ceniza which were subjected to different processing methods:
Raw Mucuna pruriens seeds (MPS).
MPS soaking in water for 24 and 48 hours and cooking at 100°C for 1 hour.
MPS soaking in 0.1% NaHCO3 (pH 8.5) and 0.2 % NaHCO3 (pH 8.6) for 4 hours and pressure cooking (autoclaving) at 121°C for 30 minutes (Chaparro et al. 2009 and Vadivel et al. 2011, respectively).
Four samples were processed by treatment, dried in oven at 60 °C for 48 hours, ground to particle size of 1 mm in hammer mill and stored in plastic bags until chemical analysis in laboratory.
Chemical analysis. Each chemical analysis was performed in duplicate. For individual sample the content of dry matter (DM) (AOAC 2005) and nitrogen (N) in Leco equipment were determined; with the latter the crude protein (CP) content (Nx6.25) was calculated.
A pool of four samples were prepared weighing 75 g each, homogenized and then DM, ash and gross energy (GE) were determined according to AOAC (2005). Fibrous fractionation (neutral detergent fiber (NDF), acid detergent fiber (ADF) and lignin were performed according to Daenlon et al. (2013), tannin content (Herald et al. 2014) and total phenols (Margraf et al. 2015) were also analyzed.
In vitro protein digestibility. The technique based on the pH-stat method, which measures the degree of hydrolysis of the protein (DHP) from the processed legume seeds (Tibbetts et al. 2011) was used. A 736 Stat Titrino device, (Metrohm, Herisau, Switzerland), that automatically titled and controlled to maintain a stable pH, was used.
Pancreatin and trypsin (SIGMA Trypsin T0303-1G, P-1750 Pancreatin of porcine pancreas) according to the methodology described by Itza (2012) were used. A solution of the two previously mentioned enzymes (0.8 mg/mL trypsin and 20 mg/mL pancreatin) was prepared, which was adjusted to pH 8 and 37°C.
Two hundred milligrams of each protein sample of raw and processed velvet bean seeds were weighed, added 25 mL of bidistilled water to ensure an aqueous suspension of 8 mg protein/mL. An adjustment to pH 8.0 and 37°C under constant stirring was carried out. Then, 0.5 mL of the enzyme solution to protein suspension was added and the enzymatic activity was measured for 900 seconds of incubation at pH-stat. An alkaline solution of 0.1 N sodium hydroxide sufficient to maintain the pH at 8 was added; the PC Control program was used to manage the Titrant.
The DPH was determined from the volume of 0.1 N NaOH used to maintain pH 8 and calculated from the equation described by Meinlschmidt et al. (2016):
Where:
htot is the total number of peptide bonds per equivalent protein and depends on the amino acid composition of the protein substrate. The value of 7.8 for soybean reported by Meinlschmidt et al. (2016) as a standard value for the legume studied was used and, h is the number of hydrolyzed peptide bonds that was calculated according to:
where:
Vb is the base intake in mL, Nb; normality of the titrant, α; the degree of dissociation of the α-NH2 group which is 1.3 to 37 °C and pH 8.0 (Itza 2012), MP; protein mass in the reaction mixture.
Experimental design and statistical analysis. Data were analyzed by ANOVA with five treatments and four replications for each. For analysis of the results INFOSTAT computerized statistical package (Balzarini et al. 2001) version 5.1 on Windows XP was used. The comparison of means was performed through the Duncan (1955) test, if necessary.
Results and Discussion
Table 1 shows the chemical composition of processed velvet bean seeds. Values are in the range reported by Ezeagu et al. (2003), Dahouda et al. (2009) and Nwaoguikpe et al. (2011).
The lower values of FDA, FDN and lignin in the treatment with 0.1 and 0.2 % bicarbonate could be explained by the results reported by Vadivel and Pugalenthi (2010) using similar methodology for processing mucuna seeds. These authors argued that it might be due to the soaking in the alkaline solution improving the seed permeability because of the cuticle degradation and the subsequent heat applied by pressure cooking (autoclaving).
The average concentration of total phenolics and condensed tannins in all treatments are lower than those reported by Adebowale et al. (2005), however, they are in the range reported by Pugalenthi et al. (2005) and Chikagwa et al. (2009). Chaparro et al. (2009) explain that the total phenolic content varies depending on several factors such as genotype, agronomic practices, maturity at harvest, post-harvest storage as well as the weather and growing conditions.
The DM was lower in water treatment after 24 and 48 hours, which was expected due to the high humidity level that the velvet beans were submitted (table 2). The protein fraction in water treatment after 24 and 48 hours and 0.2 % bicarbonate was increased in comparison to raw legume (table 2). According to Bergeson et al. (2016) the boiling process increases the protein content in both immature and mature seeds. In addition, water cooking, with or without pressure, increases the quality and digestibility of protein and carbohydrates and reduces protease inhibitors, amylase and lectins (Savón and Scull 2006).
Table 2 Dry Matter (DM) and Crude Protein (CP) content of processed Mucuna pruriens seeds
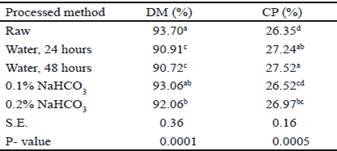
a,b,c,d Values with different letters within the same column differ significantly
Figure 1 shows the degree of hydrolysis of protein under different velvet bean processing methods. The protein digestibility improved after soaking in water and alkaline solution, boiling and cooking pressure (autoclaving). These values are found to be similar when compared with those reported by Itza (2012) for velvet bean autoclaving at 30 minutes and higher than those reported by Barać et al. (2011) in Pisum sativum seeds (4.7 %) and soybean flour treated at 121°C in autoclaving for 40 minutes (7.9 %) by Wang et al. (2009). The differences between studies indicate that the species of legume and the different methods of processing are responsible for the variation of the indicator. Apparently the treatments used, modify the protein structures and eliminate ANF in Mucuna pruriens, so the enzymes used in the pH-stat digestion process probably broke the peptide bonds in the hydrolysis or digestion according to Wang et al. (2009).
Torres et al. (2013) notified differences in both the nutritional composition and in vitro digestibility of nutrients from different tropical legume beans and they were positively correlated. Khattab et al. (2009) stated that the heat treatment either by cooking or autoclaving, denatured proteins of legumes, their structure was opened and thus become less resistant to digestive proteases. Moreover, these physical methods reduce ANF (Pugalenthi and Vadivel 2010) such as thermolabile proteases and phytates, among others, that interact with proteins of legumes forming insoluble complexes that block the action of digestive enzymes, so their reduction improves the digestibility of the protein fraction.
The results show that in Mucuna pruriens seeds soaking in water and cooking as well as soaking in 0.1-0.2 % NaHCO3 and pressure cooking are equally effective methods to improve in vitro protein digestibility.











 texto en
texto en 




