Introduction
With the increase in life expectancy and population aging, the incidence and prevalence of chronic diseases and their consequences have increased. Diseases associated with the Central Nervous System (CNS), including stroke and neurodegenerative diseases have become a major health problem, not only because of their morbidity and mortality but also because of the lack of effective treatments.1,2,3
In recent decades, intense research has been carried out in the search for new therapies to treat neurological diseases. Different variants of erythropoietin (EPO) have been evaluated for the treatment of neurological diseases, reporting satisfactory and encouraging results.4,5
NeuroEPO, a variant of EPO with a low sialic acid content, is obtained in Cuba at the Center for Molecular Immunology (CIM) in the production process of recombinant human EPO (rHu-EPO). Preclinical studies have demonstrated the effects of NeuroEPO on CNS protection in models of Alzheimer's, stroke, and glutamate-induced excitotoxicity.6,7,8,9 In clinical studies, intranasal administration has shown to be safe and well tolerated in healthy volunteers and shows favorable effects with clinical improvement in patients with Parkinson's disease, Alzheimer's, and spinocerebellar ataxia type 2.10,11,12,13,14 In addition, NeuroEPO has beneficial effects on glycemia and reproduction in diabetic rats.15,16
In previous studies, we reported that intranasal administration of NeuroEPO does not affect the structure of the respiratory mucosa or associated lymphoid tissue.17 However, there is insufficient evidence for the effect of NeuroEPO on the olfactory mucosa.
The objective of this research is to determine the effect of intranasal administration of NeuroEPO on the histological structure of the olfactory mucosa in Wistar rats.
Material and Methods
An experimental, prospective, and longitudinal study was carried out with the participation of the National Center for the Production of Laboratory Animals (CENPALAB) and the Institute of Basic and Preclinical Sciences “Victoria de Girón” (ICBP-UCMH). Nulliparous Wistar rats, three weeks old and weighing 150 to 175 g were used as animal models. They were maintained in the CENPALAB animal farm in suitable environmental conditions with a relative humidity of 40-70 %, with regulation of the light/dark cycle of 12/12 hours. The temperature ranged from 19 to 25 °C and they were offered access to food and water on demand.18,19
NeuroEPO formulation
The nasal formulation of NeuroEPO whose Active Pharmaceutical Ingredient (API) is rHu-EPO with low sialic acid content (with PCT / cu2006 / 000001 patents and 20050138 to CIDEM, Havana, Cuba)20 was developed by the Research Center for the Development of Medicines (CIDEM). Both the NeuroEPO and the vehicle were supplied by the CIM. The vehicle contains all substances (excipients) except API.
Animals were randomly distributed in two study groups of 5 rats each.
Control group, intranasal administration of vehicle (0.3μl/g/day)
Treated group, intranasal administration of NeuroEPO (300 μg/kg/day)
Intranasal application
Both groups of rats received intranasal (IN) treatment for 28 days. The animals were immobilized in a supine position. The administration of nasal formulation was applied slowly (approximately two minutes) with an automatic pipette.
Processing and staining of nasal structures for histological studies
The animals were euthanized on the 28th day and end of the experiment. The nasal structures were removed and washed with 0.9 % sodium chloride solution and fixed in neutral 4 % formalin for 24 hours. Subsequently, they were subjected to a process of bone decalcification with formic acid for a period of 14 days. Nasal tissue samples were obtained for histopathological examination of the third segment of the rats' nasal cavity (T3).21 The samples were processed using the paraffin inclusion technique.22 With a Histo-Line Laboratories MR 300 microtome, sections of 5 μm thickness were made to the nasal tissue included, and 5 slides were made per animal. The staining methods used were: the hematoxylin and eosin technique, the histochemical technique of PAS, and the Mallory trichrome technique.
Histological study of the olfactory mucosa
Histological slides were examined at various magnifications (100x, 400x, and 1000x) under a Motic BA 210 digital light microscope looking for histopathological changes in the olfactory epithelium and lamina propria. Ten randomly selected fields were observed in each slide. A global assessment was carried out in each field, analyzing aspects related to the shape, size, color, and location of the structures, also taking into account possible inflammatory or degenerative changes.23
Morphometric study of the olfactory mucosa
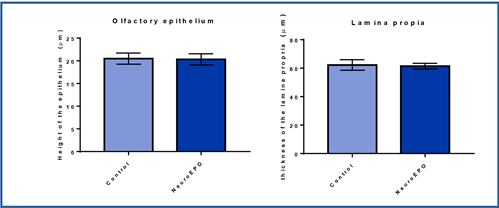
The olfactory mucosa was analyzed at the level of the dorsal meatus of the T3 segment. The height of the epithelium was measured from the basement membrane of the epithelium to its apical surface. The thickness of the lamina propria was measured by looking at the connective tissue underlying the epithelium.
Images randomly taken in 10 fields were digitized on each histological sheet. Motic Images Plus 2.0 software was used to photograph each field. A high-resolution digital camera model Moticam coupled to the microscope was used. The photographs were taken with 10x, 40x, and 100x lenses, a binocular 300 (F.N.20) Widefield projection tube, and 100/20.80 light distribution. Measurements were made on the scanned images using the Image Tool version 3.0 software for Windows.
Statistical analysis
Data on epithelium height and lamina propria thickness were collected on a Microsoft Excel sheet. The mean and standard deviation were determined as statisticians of central tendency and dispersion, respectively. The groups were compared using the Mann-Whitney U test because the data did not present a normal distribution (according to the Kolmogorov-Smirnov test). Statistically significant differences were considered for p<0.05. All statistical analyses were performed with the GraphPad Prism 7 software for Windows.
The primary data of this study are available from Mendeley Data as a principle of open access to information.24
Ethical issues
The experiment was carried out after approval by the Institutional Committee for the Care and Use of Laboratory Animals (CICUAL) of CENPALAB. All procedures with animals were performed by highly qualified personnel, following the indications of institutional and international guidelines for the use and care of animals in the laboratory.18,19
Results
Histological features of the olfactory mucosa
The epithelium of the olfactory mucosa presented a normal histological structure in the rats of both study groups. No inflammatory infiltration or signs of fibrosis were observed. (Figures 1 and 2).
The connective tissue of the lamina propria showed normal characteristics. The olfactory blood vessels and nerves did not present alterations. However, hyperplasia and hypertrophy of Bowman's glands were observed in all animals in the NeuroEPO-treated group (Figures 1B and 2B). The positivity of these glands was evidenced by the histochemical technique of SBP that impresses preserved secretion. (Figures 2C and 2D).
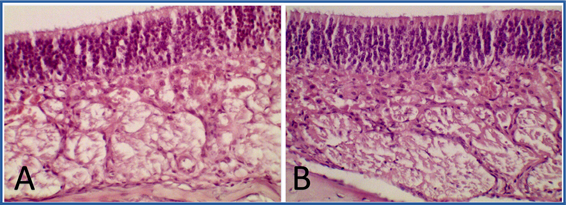
Fig. 1 Optical photomicrograph of the olfactory mucosa of Wistar rats. T3. Dorsal meatus. A: Control group. B: Group treated with NeuroEPO. OA, olfactory epithelium. LP: Lamina propia. Hematoxylin and eosin staining. 400X magnification.

Fig. 2 Optical photomicrograph of the olfactory mucosa of Wistar rats. T3. A and C: Control group. B and D: Group treated with NeuroEPO. OA, olfactory epithelium. GB: Bowman's gland, VS: blood vessels, NO: olfactory nerve. A and B: Mallory trichrome staining. Magnification 1000X. C and D: PAS staining. 400X magnification.
Morphometric study
Height of the olfactory epithelium and thickness of the lamina propria
The olfactory mucosa of the rats treated with NeuroEPO showed no differences in relation to the measurements made in the height of the epithelium or the thickness of the lamina propria with respect to that of the rats that received the vehicle. (Figure 3).
Discussion
Safety is a key issue when designing the formulation of a drug. During the development process, not only the safety of the drug itself but also the active ingredients and excipients within the formulation must be considered.25,26
The human nasal mucosa has an average physiological pH of 6.3 so it can be considered slightly acidic. Maintaining pH in nasal mucus ensures ciliary clearance function. That is why nasal formulations should have a pH within a range of 4.5 to 6.5 to avoid nasal irritation.25,26
Not only pH but also osmolarity can induce or favor the presence of toxicological or local effects. However, many substances influence mucociliary clearance through two fundamental mechanisms: by stimulation or by inhibition. Inhibition is the main cause of adverse side effects, such as nasal dryness, irritation, sneezing, nasal itching, rhinitis, and congestion.26
The present research demonstrated that treatment with intranasal NeuroEPO did not lead to significant histological changes in the olfactory mucosa of Wistar rats, corroborated by the morphometric study. It is a result that corresponds to a previous study on the respiratory mucosa under the same experimental conditions, in which no histological alterations were evidenced.17
In any of the cases, leukocyte infiltration, edema, vascular changes, tissue destruction, or connective tissue replacements with vessel proliferation or signs of fibrosis were not observed. Similarly, there were no histological alterations in the cellular or tissue structural characteristics of the olfactory epithelium. These results coincide with research, in which, the use of high doses of intranasal NeuroEPO for 14 days in Wistar rats did not cause signs of inflammatory response or cellular or tissue damage.27
The results of the current study contrast with several reports. The administration of high doses of substances such as benzalkonium chloride and dantrolene nasally has caused degeneration of the olfactory epithelium and loss of its cells (both sustaining and receptor cells). In addition, signs of inflammatory response indicative of tissue damage were described.28,29,30,31
In the present study, slight hyperplasia and hypertrophy of the Bowman's glands were observed in all animals in the treated group. However, it did not affect the thickness of the sheet itself. The positivity of these glands to the histochemical technique of PAS impressed preserved secretion. The hypertrophy and hyperplasia of the Bowman's glands described could likely be related to reversible adaptive changes of the cells of the glandular epithelium, which does not affect their function and does not translate pathological damage caused by the drug administered nasally.
These results differ from some research studies in which morphological changes in the lamina propria have been reported with the use of drugs at high doses through the nasal route. The absence of bundles of nerve fibers has been demonstrated, as well as reversible and irreversible signs of cell damage in the cells of the glandular epithelium.25,26,31
In the morphometric study of the olfactory mucosa, no significant differences were observed in the height of the olfactory epithelium and the thickness of the lamina propria in the study groups. In the literature reviewed, no morphometric data of the olfactory mucosa related to the use of NeuroEPO were found. However, a marked decrease in the height of the olfactory epithelium has been reported following the use of some drugs such as methyl bromide and methimazole.25,26,31 Concerning the thickness of the lamina propria, no reports were found in the literature consulted that could be compared with the present study.
This research was limited to the assessment of the structure of the olfactory mucosa at the level (T3) of the nasal cavity so we considered that further studies examining all three levels and including functional assessments could provide stronger evidence.
Conclusions
The administration of 300 μg/kg or 6900 IU/kg of NeuroEPO to Wistar rats, for 28 days, did not cause significant structural changes in the olfactory mucosa in our experimental conditions. These results suggest that NeuroEPO can be used intranasally without the risk of producing local adverse effects.