Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Biotecnología Aplicada
versión On-line ISSN 1027-2852
Biotecnol Apl vol.32 no.3 La Habana jul.-set. 2015
RESEARCH
Esters biotransformation by immobilized interfacial esterases from the Caribbean Sea anemone Stichodactyla helianthus
Biotransformación de ésteres por esterasas interfaciales inmovilizadas a partir de la anémona marina Stichodactyla helianthus
Alberto del Monte-Martínez1, Jorge González-Bacerio1, Bessy Cutiño-Avila1, Ritsie Ruiz2, Rizette Avila2, Miguel Ramos-Leal3, Héctor Nolasco4, Joaquín Díaz1, José M. Guisán5
1Laboratorio de Tecnología Enzimática, Centro de Estudio de Proteínas, Facultad de Biología, Universidad de La Habana. Calle 25 #455 entre I y J, Vedado, CP 10400, La Habana, Cuba.
2Departamento de Síntesis Química, Centro de Química Biomolecular, CQB. Calle 200 y 21, Playa, CP 11600, La Habana, Cuba.
3Laboratorio de Biotecnología, Departamento de Microbiología y Virología, Facultad de Biología, Universidad de La Habana, Cuba.
4Centro de Investigaciones Biológicas del Noroeste, Consejo Nacional de Ciencia y Tecnología (CONACyT). La Paz, Baja California Sur, México.
5Departamento de Biocatálisis, Instituto de Catálisis, Consejo Superior de Investigaciones Científicas (CSIC) Campus Cantoblanco, Código Postal 28049, Madrid, España.
ABSTRACT
An immobilized biocatalyst (ShIE-Octyl) was obtained by interfacial adsorption on Octyl-Sepharose CL 4B support of all interfacial esterases from the aqueous extract of the sea anemone Stichodactyla helianthus. ShIE-Octyl, synthesized by this simple method, contains semipurified interfacial esterases, including the isotoxins StI and StII. The immobilized esterases are maximally stable at pH 7.0 for p-nitrophenylacetate hydrolysis (determined spec-trophotometrically at 348 nm) during 6 days, although immobilization does not enhance the stability of the soluble enzymes toward pH. In contrast, immobilization appreciably increases the stability toward temperature and organic solvents. ShIE-Octyl shows 90 % residual activity after 6 days at 30 °C, and maintains at least 85 % initial activity in presence of 10 % methanol or acetonitrile. The immobilized derivative catalyzes the hydrolysis of the pharmaco-logically relevant esters: naproxen methyl ester, 2-oxyranylmethyl acetate (OMAc), methyl-prostaglandin A2 and ethyl-2-hydroxy-4-phenyl butanoate (HPBEt) (determined by RP-HPLC or HPTLC) with 95-100 % conversion in 6240 min, and tolerates 20 % organic solvents. The immobilized biocatalyst is selective for esters with simple alcoholic and complex acid structures, but showing the infrequent ability to hydrolyze esters with heteroatomic or aromatic alcoholic substituents, such as phenylethyl butyrate and 7-aminocephalosporanic acid. ShIE-Octyl is S-stereoselective in the bioconversion of chiral HPBEt and OMAc, and R-stereoselective in the hydrolysis of naproxen methyl ester in presence of 10 % methanol or acetonitrile. The selectivity by (S)-OMAc is favored at low temperature (4 °C) and buffer ionic strength (10 mM sodium phosphate). The enantioselectivities toward naproxen methyl ester and OMAc are unusual; then are particularly relevant.
Keywords: enantioselective hydrolysis, enzymatic bioconversion, interfacial adsorption, marine invertebrates, sticholysins.
RESUMEN
Se obtuvo un biocatalizador inmovilizado (ShIE-Octyl), mediante la adsorción interfacial en el soporte Octyl-Sepharose CL 4B de todas las esterasas interfaciales del extracto acuoso de la anémona marina Stichodactyla helianthus. El ShIE-Octyl sintetizado mediante este método simple contiene esterasas interfaciales semipurificadas, incluidas las isotoxinas StI y StII. Las esterasas inmovilizadas tienen estabilidad máxima a pH 7.0 para la hidrólisis de p-nitrofenil acetato (determinada espectrofotométricamente a 348 nm) durante 6 días, 90 % de la actividad residual a 30 ºC posteriormente, y conservan el 85 % de la actividad inicial en presencia de metanol o acetonitrilo 10 %. La inmovilización no incrementa la estabilidad de las enzimas solubles frente al pH y sí frente a la temperatura y los solventes orgánicos. El biocatalizador hidroliza los ésteres farmacológicamente relevantes: éster metílico del naproxeno, acetato de 2-oxiranilmetilo (OMAc), metil-prostaglandina A2 y 2-hidroxi-4-fenil-butirato de etilo (HPBEt), determinada mediante RP-HPLC o HPTLC, con 95-100 % de conversión en 6240 min, y tolera solventes orgánicos al 20 %. La enantioselectividad frente a los dos primeros es inusual y particularmente relevante. Además, es selectivo por ésteres con estructuras ácidas complejas y alcohólicas simples, aunque hidroliza aquellos con sustituyentes alcohólicos aromáticos o heteroatómicos, como el butirato de feniletilo y el ácido 7-aminocefalosporánico. Es S-estéreoselectivo en la bioconversión de HPBEt y OMAc quirales, y R-estéreoselectivo en la hidrólisis del éster metílico del naproxeno en presencia de metanol o acetonitrilo 10 %. La selectividad por (S)-OMAc se favorece a baja temperatura (4 °C) y fuerza iónica del tampón fosfato de sodio (10 mM).
Palabras clave: adsorción interfacial, bioconversión enzimática, hidrólisis enantioselectiva, invertebrados marinos, sticholisinas.
INTRODUCTION
Interfacial esterases, as lipases, are enzymes that require lipid/water interfaces to develop maximal catalytic activity (an effect called interfacial activation) [1]. These proteins are widely spread in nature, can catalyze distinct reactions, such as ester hydrolysis, esterification and transesterification [2], and show variable and complex substrate selectivities, including stereoselectivity [3]. They have been extensively used on bioconversion processes and racemic mixtures resolution in the food, detergent, chemical, agrochemical, pharmaceutical and fine chemistry industries [4].
Many drugs, herbicides and pesticides are commercialized as racemic mixtures, where one enantiomer prevails as biologically active whereas the other is inactive, toxic or antagonistic [5]. Due to strict pharmaceutical regulations, there is a growing demand of economically feasible methods to synthesize pure enantiomers [6]. This has been performed by stereoselective crystallization [7], chemical asymmetric synthesis [8], and preparative chiral chromatography [9]. Since these procedures are time-consuming or expensive, optimized biocatalytic processes, including those that use interfacial esterases, have emerged as a popular option [10]. In this sense, enzyme immobilization is advantageous for industrial applications, since it allows the biocatalyst recovery, avoids product contamination, and can increase enzyme stability [11, 12]. On the basis of interfacial activation, interfacial esterases can be selectively immobilized on hydrophobic solid supports at low ionic strength (a methodology called interfacial adsorption) [13, 14].
Considering the enzyme properties, those from marine invertebrates (or microorganisms associated with them) show unusual substrate specificities and molecular properties, making of them good candidates for biotechnological applications [15-19]. Previously, we described the immobilization by interfacial adsorption on Octyl-Sepharose CL 4B support of interfacial esterases from the aqueous whole extract of the Caribbean Sea anemone Stichodactyla helianthus [20]. Two of the immobilized enzymes were Sticholysins I and II (StI and StII), 19 kDa-isotoxins with esterase activity [20, 21]. The derived immobilized biocatalyst, named ShIE-Octyl, has kinetic characteristics toward p-nitrophenylacetate (p-NPA), suggesting its potential use on enzymatic biotransformation [20]. In this work is char-acterized the stereoselective application of ShIE-Octyl on ester hydrolysis, using relevant pharmacological active principles. The obtained conversion percentages (C) and enantiomeric ratios (E), as well as the stability properties, suggest the possibility of further optimization of this biocatalyst for its use on large scale production processes.
MATERIALS AND METHODS
Materials
Octyl-Sepharose CL 4B support was purchased from Sigma Chemical (St. Louis, USA). Phenylethyl butyrate (BPEt), ethyl-2-hydroxy-4-phenyl butanoate (HPBEt) and 2-oxyranylmethyl acetate (glycidol acetate, OMAc) were kindly supplied by Instituto de Catálisis, CSIC, Madrid, Spain. 7-aminocephalosporanic acid (7-ACA), methyl-prostaglandin A2 (Me-PGA2) and methyl-6-metoxy-α-methyl-2-naphtalen-acetate (naproxen methyl ester) were supplied by Centro de Química Farmacéutica, Havana, Cuba. Plates (5 × 7.5 cm) precoated with silica gel F254 for High Performance Thin Layer Chromatography (HPTLC), LiChrosorb C-4 column (125 × 4 mm) and solvents for Reverse-Phase High Performance Liquid Chromatography (RP-HPLC) were obtained from Merck (Darmstadt, Germany). All aqueous solutions were prepared with deionized water (Milli-Q quality). All other reagents and organic solvents (Sigma Chemical, St. Louis, USA) were analytical grade and used without further purification.
Preparation of S. helianthus extract and protein immobilization
Collection of the sea anemone S. helianthus, preparation of the aqueous whole protein extract, and protein immobilization by interfacial adsorption on Octyl- Sepharose CL 4B support, were performed as described [20]. Briefly, the protein extract was diluted (1:10 v/v) with 10 mM sodium phosphate buffer, pH 7.0, incubated with the support during 5 h, at 4 °C and constant stirring, filtrated and washed with the same buffer. Protein and activity loads during immobilization were 0.54 mg protein and 8 esterase activity units (U; toward p-NPA), respectively, per milliliter of support.
Protein electrophoresis
Protein samples (10 mL soluble and immobilized extract) were analyzed by standard 15 % sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) [22], under reducing conditions, and visualized by Coomassie blue staining. The following molecular weight markers were used: bovine serum albumin (66 kDa), egg albumin (45 kDa), trypsinogen (24 kDa), lactoglobulin (18.4 kDa), lisozyme (14.3 kDa) and aprotinin (6.5 kDa).
Esterase activity assay
Esterase activity toward p-NPA substrate was assayed as described [20]. Briefly, p-NPA-hydrolyzing activity was assessed by continuous and spectrophotometric measurement of the p-nitrophenol releasing at a wavelength of 348 nm (ε348 nm of 5150 M-1 cm-1 [23]), during 10 min. Automatic determinations (Spekol, Germany) were performed under magnetic stirring, at 30 °C, in a 1 cm cuvette. The composition of the reaction mixtures were the following: 0.01 mg/mL final protein concentration for soluble extract or 1:182 dilution for the immobilized derivative, 1.185 mM p- NPA, 40 mM CaCl2, 25 mM Tris-HCl buffer, pH 8.0. U is defined as the amount of enzyme that hydrolyzes 1 mmol of substrate per minute under the assays conditions.
Study of esterases stability
For the assessment of stability toward pH, soluble (8 U/mL) and immobilized (12.7 U/mg protein) extracts were preincubated in sealed flasks for 2 h at 25 °C in various buffers (25 mM): sodium acetate pH 4.0 and 5.0, sodium phosphate pH 6.0 and 7.0, Tris-HCl pH 8.0, sodium bicarbonate pH 9.0, 10.0 and 11.0. Aliquots were taken at 0, 5, 10, 15, 30, 60 and 120 min, and esterase activity was measured as was described above. Additionally, the immobilized extract was preincubated for 6 days at pH 7.0, and esterase activity was determined at 0, 1, 2, 4, 5, 10, 24, 48, 72, 96, 120 and 144 h (6 days). The results are presented as residual activity percentages (mean for 3 replicates), taking as 100 % the activities measured without preincubation. The stability study toward temperature was similar to that above described, but preincubation was performed at pH 7.0 and 10, 20, 25, 30, 40, 50, 60 or 70 °C. The additional 6 days-preincubation of the immobilized extract was performed at pH 7.0 and 30 °C. The stability study toward organic solvents was similar to those above described, but preincubation was performed at pH 7.0, 30 °C and in presence of different organic solvents: 10 % (v/v) methanol, acetonitrile and dimethyl sulfoxide (DMSO), 20 % (v/v) acetone and ethanol. Preincubation with sodium phosphate pH 7.0 (with no solvent) was done as a control. The additional 6 days-preincubation of the immobilized extract was performed at pH 7.0 and 30 °C, and all solvents were tested.
Esters bioconversion assay
The hydrolysis of esters by ShIE-Octyl (12.7 U/mg protein) was assayed by mixing 1 mL (0.7 g) immobilized derivative with 4 mL of different substrate solutions (25 mM sodium phosphate pH 7.0): 5.09 mM HPBEt at 25 °C, 5.05 mM BPEt at 25 °C, 1.77 mM naproxen methyl ester at 30 °C in presence of 10 % (v/v) acetonitrile or methanol, 4.32 mM OMAc at 25 °C, 258 mM Me-PGA2 at 30 °C in presence of 20 % (v/v) acetone or ethanol, and 612 mM 7-ACA at 30 °C. The chiral substrates (HPBEt, naproxen methyl ester and OMAc) were racemic mixtures (equivalent amounts of the S and R isomers; the mixture was denoted as (R,S)), and the reaction temperatures depend on the solubility of each compound. The reaction mixtures (in triplicates) were stirred during 6240 min (~4 days), and filtered aliquots (to exclude the agarose pearls) were taken each 12 h for RP-HPLC or HPTLC analysis. Negative controls of hydrolysis or adsorption of the compounds by the Sepharose CL 4B support were also assayed.
Enantioselectivity assay
The enantioselectivity of ShIE-Octyl (12.7 U/mg protein) was assayed as is described above for the hydrolysis of esters, but using (R,S)- and (R)-HPBEt, (S)- and (R)-naproxen methyl ester, and (S)- and (R)- OMAc as substrates. The reactions with OMAc were performed at 25 °C (25 mM buffer) and 4 °C (10, 25 and 200 mM buffer).
Analysis of ester hydrolysis by RP-HPLC
The hydrolysis of BPEt, HPBEt, OMAc, and naproxen methyl ester by ShIE-Octyl was monitored applying the reaction mixture aliquots on a LiChrosorb C-4 column (125 × 4 mm) for RP-HPLC [24]. The mobile phases for isocratic elutions were the followings (10 mM sodium phosphate buffer, pH 3.0): acetonitrile/buffer (60:40 v/v) for BPEt, acetonitrile/buffer (55:45 v/v) for HPBEt, acetonitrile/buffer (90:10 v/v) for OMAc, acetonitrile/water/acetic acid (50:49:1 v/v/v) for naproxen methyl ester. The flow rates were 2.0 mL/min for BPEt, 1.5 mL/min for HPBEt and OMAc, and 1.2 mL/min for naproxen methyl ester. Elution was monitored at 215 nm by diode array detection for BPEt and OMAc, or 254 nm for HPBEt and naproxen methyl ester. Data were captured and analyzed using the computer-based technology Biocrom v3.1 (Center of Genetic Engineering and Biotechnology, Havana, Cuba).
Analysis of ester hydrolysis by HPTLC
The aliquots from the reactions of hydrolysis of 7-ACA and Me-PGA2 were analyzed by HPTLC [25]. Samples were applied onto the precoated silica gel F254 plates, using the CAMAG Linomat IV applicator. Mobile phases were n-butanol/acetic acid/ water (3:1:1 v/v/v) and ethyl acetate/isooctane/acetic acid/water (9:5:2:10 v/v/v/v), respectively. Runs were quantified at 254 nm using the densitometer CAMAG TLC Scanner III (Muttenz, Switzerland). The technique was calibrated with known concentrations of both esters. The data were analyzed using the software winCATS Chromatography Manager, supplied by the manufacturer.
Bioconversion parameters
C values were calculated by comparing the areas under the ester peaks (resulting from RP-HPLC and HPTLC analyses) at different reaction times with the initial area. The conversion rates (vconv) were calculated according to the equation:
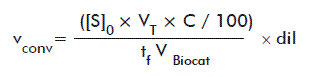
where:
[S]0: Initial substrate concentration in the assay (mM).
VT: Total assay volume (5 mL).
tf: Final time of the assay (6240 min).
VBiocat: Total volume of immobilized biocatalyst used in the assay (1 mL).
dil: Sample dilution.
For the chiral ester compounds (naproxen methyl ester, HPBEt and OMAc), E values were calculated as follows. When both pure isomers were available (for naproxen methyl ester and OMAc) E were calculated according to the equations:
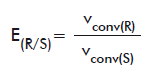 or
or 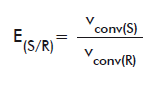
where:
E(R/S): E of the R isomer over the S form.
vconv(R): vconv of the R isomer.
vconv(S): vconv of the S isomer.
E(S/R): E of the S isomer over the R form.
When the racemic mixture and the pure R isomer were available (for HPBEt) E was calculated according to the equation:
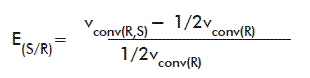
where: vconv(R,S): vconv of the racemic mixture.
Statistical analyses
Normal distribution and homogeneity of variances for the data were verified. Comparison of the means was performed by the Student’s t test. All these analyses were done using Statistica software (version 8.0; StatSoft Inc., available at: http://www.statsoft.com) with default parameters.
RESULTS AND DISCUSSION
Obtainment of ShIE-Octyl
As was previously corroborated [20], immobilization of the whole extract of S. helianthus by interfacial adsorption on the support Octyl-Sepharose CL 4B appreciably diminishes its protein heterogeneity (Figure 1). An immunoblotting with an antiserum anti-StII demonstrated that the most abundant proteins in the immobilized extract are StI and StII, which co-migrate in the same 19 kDa protein band [20, 21] (Figure 1). In addition, an esterase specific zymography with the substrate β-naphtylcaprilate showed that the immobilized extract contains, at least, a high molecular weight lipase [20] (> 30 kDa [26]). Other protein bands are unknown (Figure 1), although the presence of other esterases in ShIE-Octyl, non-detected in the zymography [20], should not be discarded. As was already demonstrated [20], all interfacial esterases from the protein extract (45 % of the total proteins) were immobilized, although ShIE-Octyl contains other non-esterase proteins, which may be bound to the octyl-covered surface by non-specific van der Waals interactions. Nevertheless, the simple, economical and selective immobilization procedure used here was useful not only to obtain an active biocatalyst, able to hydrolyze ester bonds (12.7 U/mg protein vs. p-NPA), but also to semipurify the interfacial esterases of S. helianthus aqueous whole extract (Figure 1). A similar result was obtained with commercial preparations of lipases [27]. Such semipurification effect by interfacial adsorption of interfacial esterases from a heterogeneous extract was previously reported for the lipases of Penicillium simplissicimum using the octyl agarose support [28].
Porous materials, such as the Octyl-Sepharose CL 4B, have been extensivelly used for enzyme immobilization [29]. Those supports are suitable for the full dispersal of protein molecules over their internal surfaces, preventing enzyme inactivation by contact with external hydrophobic interfaces. Different enzymes have been immobilized on porous supports by non-covalent attachments [11, 14, 30, 31]. For example, formaldehyde dehydrogenase was immobilized onto mesoporous zirconia material by electrostatic interactions [31]. Such binding was strong enough to retain about 80 % of the initial activity after 7 reaction cycles (operational stability). On the other hand, physical adsorption of lipases on different supports, with significant activity retention and reusability, has been reported too [32-36]. As was defined by Barbosa et al. [14], if the enzyme-support interactions are enough to maintain the enzyme immobilized during use (like ShIE-Octyl [20]), such system can be considered as an immobilized biocatalyst.
Stability of ShIE-Octyl
Immobilization is probable the most useful strategy to improve enzyme stability [12, 14, 37, 38]. Stabilization is associated with several factors. Immobilization may: i) induce some degree of rigidity in the enzyme (mainly after multipoint covalent attachment), preserving the native conformation; ii) prevent dissociation of the subunits of multimeric enzymes; iii) avoid aggregation, autolysis and proteolysis by proteases (in the case of protein extracts); iv) prevent enzyme contact with external hydrophobic interfaces, such as air bubbles or water/organic solvent interfaces (for porous supports); and v) generate a more suitable environment for catalysis, regarding the soluble enzyme [39, 40]. To verify a potential use of ShIE-Octyl in biotransformation processes we investigated the stability of the biocatalyst p-NPA-hydrolyzing activity (12.7 U/mg protein) toward pH, temperature, and organic solvents, comparing with the esterase activity of the soluble extract (8 U/mL).
As shown in Figure 2A-C, the immobilization did not exert a noticeable influence on stability of these enzymes toward pH, as the behaviors of soluble and immobilized extracts were similar until at least 2 h. In both cases, the highest activities (as a tendency) were achieved by preincubation at pH 7.0 (90 and 94 % residual activities for soluble extract and ShIE-Octyl, respectively), and stability was lower at the most extreme analyzed pHs (4.0, 5.0, 10.0 and 11.0). However, the residual activities were always maintained above 60 %. The stability of ShIE-Octyl at pH 7.0 was further studied enlarging the preincubation time to 6 days (144 h), which is more near to the real condi-tions in biotransformation processes. At this time, the immobilized derivative retained 90 % of the initial activity, and significant differences between 0 and 144 h were not detected (Figure 2D).
Other authors have reported an improvement of enzymes stability toward pH after immobilization [41, 42]. A multimeric β-galactosidase from Lactobacillus plantarum was stabilized by multipoint and multisubunit covalent attachment on glyoxyl-agarose [42]. The immobilized preparation was very stable in the pH range from 5 to 9, showing 10 % more activity than the soluble enzyme after incubation for 2 h at pH 10. An extracellular a-amylase from Bacillus acidocaldarius was significantly stabilized toward pH when was immobilized on glass beads (covalent binding) or cation exchange resin (ionic binding) [41]. Covalent immobilization of an Aspergillus aculeatus tannase on gelatin increased the pH range at which the enzyme retained at least 80 % residual activity (pH 4.5-8.0) as compared with free tannase (pH 5.0-6.0) [43]. It is relevant to notice that all these reports are related with purified enzymes, and not with heterogeneous extracts. It is possible that esterases present in the soluble extract of S. helianthus are stabilized against pH changes by some extract components, being the effect of immobilization negligible at this respect.
Additionally, the thermal stability of the esterases present in ShIE-Octyl was studied, maintaining pH at 7.0. For both samples, 25 °C was the highest temperature at which the esterase activity was not affected until 2 h preincubation (Figure 3A-C). In contrast to the result obtained in the pH study (Figure 2), the immobilization appreciably protected the esterase activity against the loss of stability caused by temperature in the 30-50 °C range (Figure 3C). Whereas the thermal stability of the esterases present in the soluble extract was very low, with the residual activity being reduced by 45 % after preincubation for 2 h at 30 °C, the immobilized enzymes exhibited 91 % residual activity in these conditions, and retained 50 % activity at 40 °C. Moreover, the incubation of ShIE-Octyl at 30 °C for 6 days (144 h) only diminished the esterase activity by 10 %, without significant differences regarding the initial activity (Figure 3D). As the soluble and immobilized extracts differ in their esterase composition [20], another explanation for the increase of stability caused by the immobilization is the selective adsorption of intrinsically more thermostable enzymes.
Thermal stability of immobilized enzymes is well documented [31, 41, 42, 44-46]. The β-galactosidase from L. plantarum, immobilized by multipoint covalent attachment on glyoxyl-agarose support, was 6-fold more stable than the soluble enzyme at 50 °C and pH 7.0 after 6 h [42]. Immobilization of formaldehyde dehydrogenase onto mesoporous zirconia material increased the denaturation temperature from 51.8 °C, for the soluble enzyme, to 78.9 °C, for the immobilized ones [31]. An organophosphate degrading enzyme, covalently immobilized on highly porous nonwoven polyester fabrics, was stable over 3 h at 55 °C [46]. In contrast, the soluble enzyme lost more than 60 % of its activity in the same conditions. Thermal stability of glucose-6-phosphate dehydrogenase, immobilized on silanized surfaces through coupling agents (cross-linkers) and incubated 1 h at 30 °C, augmented when the size of the enzyme aggregates increased [45]. The results of that work suggest that aggregation may enhance the thermostability of the multimeric enzyme, probably through a compaction of the tertiary structure of the monomers, but also by stabilizing the quaternary structure by the assembly of the different subunits. The activity of glucose oxidase, covalently immobilized on a polyamide film, was nearly 30 % higher than the activity of the free enzyme after incubation at 40, 50 and 60 °C for 5 h [44]. In another example, covalent or ionic immobilization protected a B. acidocaldarius a-amylase from heat inactivation [41]. The half-life times at 60 °C were 83 and 61 min, respectively, against 47 min for the free enzyme. Thermal stabilization of lipases due to their immobilization by interfacial adsorption has been also reported [28]. That is the case of a lipase from Bacillus thermocatenulatus adsorbed on Octadecyl-Sepabeads [47], and a lipase fraction from P. simplissicimum immobilized on octyl agarose [28].
Although 30 °C was not the best temperature for stability in the 2 h-study (Figure 3A-C), it was selected to perform the followings experiments, as it is suitable for biotransformation processes due to substrates/products solubility and conversion rate issues [48]. The esterase activity in the S. helianthus soluble extract was sensitive to all tested organic solvents, being methanol the most tolerated, with the retention of 63 % activity after 2 h incubation (Figure 4A and C).
On the contrary, the esterase stability in presence of organic solvents was dramatically enhanced by the immobilization (Figure 4A-C). The residual activities after 2 h toward DMSO, acetonitrile, ethanol, methanol and acetone were improved from 8 to 71 %, 43 to 90 %, 21 to 56 %, 63 to 96 %, and 32 to 50 %, respectively, with significant differences in all cases (Figure 4C). The maximal stability of the enzymes present in ShIE-Octyl was reached with methanol and acetonitrile, showing in both cases more than 90 or 85 % residual activities after 2 h or 6 days (144 h) of incubation, respectively, without significant differences regarding the initial activities (Figure 4D and E). Notably, the residual activities were higher than 50 % in all tested organic solvents after 6 days of incubation.
It has been reported that immobilization may improve greatly the enzymatic activity in organic solvents [29]. For example, a lipase from Pseudomonas sp., adsorbed on amberlite XAD-7, maintained 100 % activity after 30 h of incubation in n-hexane, heptanes or isooctane [49]. Nevertheless, free lipase showed too low or no activity in those conditions. In another study, the activity of Candida rugosa lipase, covalently immobilized on ultrafiltration hollow fiber membranes, was improved significantly in the organic medium (heptane) [50]. A lipase from B. thermocatenulatus, immobilized on Octadecyl-Sepabeads by interfacial adsorption, was considerably stabilized in presence of 30 % dioxane [47].
In summary, the stability data suggest that ShIE-Octyl has properties that may be useful for the use of the biocatalyst in the biotransformation of esters. In addition, the strategy used in this work to simultaneously semipurify, immobilize, and stabilize the interfacial esterases from S. helianthus, represents a contribution to the knowledge in the field of enzyme technology [14].
Bioconversion of ester compounds by ShIE-Octyl
To investigate the potential application of ShIE-Octyl on bioconversion processes, the immobilized biocatalyst was tested on the hydrolysis of the pharmacologically relevant esters: HPBEt, BPEt, naproxen methyl ester, OMAc, Me-PGA2 and 7-ACA [51-53]. Since the atoms involved in a carboxyl ester bond cannot act as hydrogen-bond donors, ester compounds are more hydrophobic than their respective alcohols and carboxylic acids. Then, the presence of organic solvents in the reaction media to dissolve some compounds was necessary. Total or very high conversions at 6240 min (about 4 days) were achieved for naproxen methyl ester, OMAc, Me-PGA2 and HPBEt (Table 1). Further-more, the interfacial esterases present in ShIE-Octyl tolerate organic solvents, at least until 20 %, without impairment of their bioconversion ability. Such tolerance is a desired characteristic in order to a potential application of the biocatalyst in the obtainment of relevant products [54].
The obtainment of the lowest C toward the ester compounds with the highest chemical complexity in their alcohol components (7-ACA and BPEt; table 1) suggests that immobilized S. helianthus interfacial esterases show a certain preference for esters with simple alcoholic substituents. On the contrary, complex and large acid parts yield high C. Our data suggest that these structural characteristics are critical for ShIE-Octyl activity. The results obtained for C with 7-ACA and BPEt (68 and 38 %, respectively; table 1) are important too, considering the small amount of commercial enzymes able to hydrolyze esters with heteroatomic condensed rings or aromatic groups in their alcohol substituents [55]. The ability of ShIE-Octyl to catalyze the deacetylation of 7-ACA and the hydrolysis of the PGA2 methyl ester is relevant for obtaining semi-synthetic cephalosporins and more potent prostaglandins [11, 56].
A number of biotechnological products with dissimilar applications have been obtained using immobilized enzymes [11]. A biocatalyst obtained by multipoint covalent immobilization of a β-galactosidase from L. plantarum on glyoxyl-agarose, allowed produce high yields of prebiotic oligosaccharides (30- 35 %) from the hydrolysis and transglycosylation of lactose and lactulose [42]. An organophosphate degrading enzyme, immobilized on polyester textiles, is useful in environmental remediation of organophosphate compounds [46]. Other works have been focused on interfacial esterases. For instance, a membrane bioreactor based on an immobilized lipase allowed obtaining 95 % esterification degree during the synthesis of 2-ethylhexyl palmitate [57]. Moreover, a lipase from thermophilic Bacillus sp., adsorbed and cross-linked on HP-20 beads, was found suitable for the esterification of oleic acid and methanol to methyl oleate in hexane [58].
Enantioselectivity of ShIE-Octyl toward chiral esters
Enzymes generally present high specificities for their natural substrates, but not toward non-natural ones. Among the kinds of substrate selectivities, several enzymes show stereoselectivity, which may be improved by immobilization (including physical adsorption). This effect may be due to the distortion of the protein molecule as a consequence of the interaction with the support, or due to the generation of a particular microenvironment around the enzyme, provided by the physical properties of the support [14, 29, 39]. Such strategies have been previously employed to modulate the enantiopreference of lipases [53, 59, 60]. Since directed vs. random immobilization is a powerful tool to optimize the catalytic properties of enzymes [14, 29], and the interfacial esterases in ShIE-Octyl were immobilized in an oriented form by interfacial adsorption [20], we anticipated the obtainment of interesting enantioselectivities toward ester substrates. This is relevant, since the development of novel procedures to obtain chiral compounds remains as a major challenge in biotechnology.
The S isomer of naproxen, an extensively used non-steroidal anti-inflammatory drug, is 28-fold more active than the R enantiomer. Therefore, the production of optically pure (S)-naproxen is a major research area [53]. Interestingly, the hydrolytic activity of ShIE-Octyl toward naproxen methyl ester is stereoselective for the R isomer (Table 2). The importance of this result is indicated by the prevalence of S-stereoselectivity for most lipase-based biocatalysts used in naproxen methyl ester hydrolysis [61]. Biocatalysis in non-aqueous media has received growing attention due to it increases the solubility of hydrophobic substrates, aids processes which are thermodynamically unfavorable in water (e.g., ester synthesis), and facilitates enzyme and products recovery [62]. It is also known that organic solvents can affect the stereoselectivity of lipases [63]. In this sense, the R-stereoselectivity of ShIE-Octyl toward naproxen methyl ester is higher in presence of 10 % acetonitrile than 10 % methanol (Table 2). Sahin et al. [53] reported the hydrolysis of racemic naproxen methyl ester in a water/isooctane biphasic system by encapsulated C. rugosa lipase.
Additionally, HPBEt derivatives constitute essential intermediates in the synthesis of inhibitors for angiotensin converting enzyme, which is involved in heart diseases [64]. ShIE-Octyl is slightly S-stereoselective in the hydrolysis of HPBEt (Table 2), result that is qualitatively similar to other reports on HPBEt bioconversion [27, 51]. Nevertheless, reaction conditions, such as biocatalyst load, temperature, pH and substrate concentration, should be further investigated in order to increase the enantioselectivity of ShIE-Octyl for HPBEt hydrolysis.
Glycidyl esters, as OMAc, are main intermediaries in the synthesis of various reagents for the pharmaceutical industry [10]. ShIE-Octyl is selective for the OMAc S isomer, and this enantioselectivity was increased in some extent when the temperature was diminished from 25 to 4 °C in 25 mM sodium phosphate pH 7.0 (Table 2). This result is consistent with the increment of E(R/S) from 5.2 to 9.3 observed for immobilized lipases from porcine pancreas when temperature was shifted from 25 to 4 °C [10]. Buffer molarity influences too the stereoselectivity of ShIE-Octyl by OMAc, as the highest E(S/R) (2.67) at 4 °C was obtained at the lowest buffer concentration tested (10 mM). This influence of the ionic strength over the enantioselectivity of lipases has been previously documented [65]. The S-stereoselectivity of ShIE-Octyl by OMAc hydrolysis is an attractive characteristic, because most reported glycidol acetate esterases are selective for the R enantiomer. In this direction, commercial lipases from Candida antarctica and Mucor miehei are R-stereoselective, exhibiting E values of 2.9 and 2.7, respectively, for the hydrolysis of glycidyl esters [52]. In agreement with our results, it has been reported that a lipase from Rhizopus oryzae, immobilized on Octyl-agarose, showed an E(S/R) of 2 with a 25 % enantiomeric excess (calculated at 50 % C) toward the structurally related substrate glycidyl butyrate at pH 7.0 and 25 °C [66].
Other authors have reported the enantioselective properties of various immobilized lipases/esterases [59, 60, 67-69]. For example, lipase B from C. antarctica, immobilized by hydrophobic interactions on octyl agarose, hydrolyzed racemic 2-O-butyryl-2-phenylacetic acid with 95 % enantiomeric excess (R) [59]. The same substrate was resolved with the opposite enantioselectivity (E(S/R) 85; > 95 % enantiomeric excess at 10-20 % C) by a C. rugosa lipase immobilized on octyl agarose [67]. An encapsulated lipase from C. rugosa showed higher C and E than the free enzyme [60]. The esterase of Klebsiella oxytoca, immobilized on Eupergit C 250 L, was enantioselective for the hydrolysis of (R,S)-ethyl mandelate [69]. A membrane bioreactor based on lipase B from C. antarctica was used for the enantioseparation of (R,S)-ketoprofen [68].
CONCLUSIONS
This is the first report concerning the utilization of immobilized interfacial esterases of S. helianthus for biotransformation processes. The stability, bioconversion and enantioselectivity data suggest the possibility of using
ShIE-Octyl for catalytic processes based on thermodynamically or kinetically controlled reactions [70]. Further work should be directed to evaluate on biotransformation different interfacial esterases fractions from S. helianthus ex-tract, in order to improve the functional properties of the biocatalyst.
ACKNOWLEDGEMENTS
The authors want to thank INFORMATICA ddmm., Bergamo, Italy, for kindly provided financial support to del Monte A. This work was partially supported by grants from Havana University, Cuba; Catalysis Institute (CI), CSIC, Madrid, Spain and CONACyT, Mexico. The authors are also grateful to CI and Pharmaceutical Chemistry Center, Havana, Cuba, for supplying the ester compounds used in this study.
REFERENCES
1. Reis P, Holmberg K, Watzke H, Leser ME, Miller R. Lipases at interfaces: a review. Adv Colloid Interface Sci. 2009;147-148:237-50.
2. Goujard L, Villeneuve P, Barea B, Lecomte J, Pina M, Claude S, et al. A spectrophotometric transesterification-based assay for lipases in organic solvent. Anal Biochem. 2009;385(1):161-7.
3. Ma J, Wu L, Guo F, Gu J, Tang X, Jiang L, et al. Enhanced enantioselectivity of a carboxyl esterase from Rhodobacter sphaeroides by directed evolution. Appl Microbiol Biotechnol. 2013;97(11):4897-906.
4. Zarevucka M, Wimmer Z. Plant products for pharmacology: application of enzymes in their transformations. Int J Mol Sci. 2008;9(12):2447-73.
5. Gottemukkala VV, Saripella KK, Kadari AK, Neau SH. Effect of methyl branching of C8H18 alkanes and water activity on lipase catalyzed enantioselective esterification of ibuprofen. Electron J Biotechnol. 2008;11:1-12.
6. Hernández-Fernández FJ, de los Ríos AP, Tomás-Alonso F, Gómez D, Víllora G. Kinetic resolution of 1-phenylethanol integrated with separation of substrates and products by a supported ionic liquid membrane. J Chem Technol Biotechnol. 2009;84(3):337-42.
7. Sarasua JR, López N, López A, Meaurio E. Stereoselective crystallization and specific interactions in polylactides. Macromolecules. 2005;38(20):8362-71.
8. Wan X, Meng Q, Zhang H, Sun Y, Fan W, Zhang Z. An efficient synthesis of chiral beta-hydroxy sulfones via ru-catalyzed enantioselective hydrogenation in the presence of iodine. Org Lett. 2007;9(26):5613-6.
9. Ali I, Kumerer K, Aboul-Enein HY. Mechanistic principles in chiral separations using liquid chromatography and capillary electrophoresis. Chromatographia. 2006;63(7):295-307.
10. Pregnolato M, Terreni M, de Fuentes IE, Alcántara AR, Sabuquillo P, Fernández- Lafuente R, et al. Enantioselective enzymatic hydrolysis of racemic glycidyl esters by using immobilized porcine pancreas lipase with improved catalytic properties. J Mol Catal B Enzymatic. 2001;11(4-6):757-63.
11. Brena B, González-Pombo P, Batista- Viera F. Immobilization of enzymes: a literature survey. In: Guisán JM, editor. Immobilization of enzymes and cells. 3rd ed. New York: Springer Science+Business Media; 2013. p. 15-31.
12. Nunes de Lima L, Aragon CC, Mateo C, Palomo JM, Giordano RLC, Tardioli PW, et al. Immobilization and stabilization of a bimolecular aggregate of the lipase from Pseudomonas fluorescens by multipoint covalent attachment. Proc Biochem. 2013;48(1):118-23.
13. Fernandez-Lafuente R, Armisen P, Sabuquillo P, Fernandez-Lorente G, Guisan JM. Immobilization of lipases by selective adsorption on hydrophobic supports. Chem Phys Lipids. 1998;93(1-2):185-97.
14. Barbosa O, Torres R, Ortiz C, Berenguer-Murcia A, Rodrigues RC, Fernandez- Lafuente R. Heterofunctional supports in enzyme immobilization: from traditional immobilization protocols to opportunities in tuning enzyme properties. Biomacromolecules. 2013;14(8):2433-62.
15. Kennedy J, Marchesi JR, Dobson AD. Marine metagenomics: strategies for the discovery of novel enzymes with biotechnological applications from marine environments. Microb Cell Fact. 2008;7:27.
16. Selvin J, Kennedy J, Lejon DP, Kiran GS, Dobson AD. Isolation identification and biochemical characterization of a novel halo-tolerant lipase from the metagenome of the marine sponge Haliclona simulans. Microb Cell Fact. 2012;11:72.
17. Pozzolini M, Scarfi S, Mussino F, Ferrando S, Gallus L, Giovine M. Molecular cloning, characterization, and expression analysis of a prolyl 4-hydroxylase from the marine sponge Chondrosia reniformis. Mar Biotechnol (NY). 2015;17(4):393-407.
18. Su J, Zhang F, Sun W, Karuppiah V, Zhang G, Li Z, et al. A new alkaline lipase obtained from the metagenome of marine sponge Ircinia sp. World J Microbiol Biotechnol. World J Microbiol Biotechnol. 2015;31(7):1093-102.
19. Debashish G, Malay S, Barindra S, Joydeep M. Marine enzymes. Adv Biochem Eng Biotechnol. 2005;96:189-218.
20. del Monte-Martínez A, González- Bacerio J, Aragón-Abreu C, Palomo- Carmona JM, Guisán-Seijas JM, Díaz-Brito J. Selective and oriented immobilization of (phospho)lipases from the Caribbean Sea anemone Stichodactyla helianthus (Ellis, 1768) by interfacial adsorption. Rev CENIC Cienc Biol. 2012;43:3-8.
21. del Monte-Martinez A, Gonzalez- Bacerio J, Romero L, Aragon C, Martinez D, Chávez M de los A, et al. Improved purification and enzymatic properties of a mixture of Sticholysin I and II: isotoxins with hemolytic and phospholipase A(2) activities from the sea anemone Stichodactyla helianthus. Protein Expr Purif. 2014;95:57-66.
22. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680-5.
23. Baker JE, Fabrick JA, Zhu KY. Characterization of esterases in malathion-resistant and susceptible strains of the pteromalid parasitoid Anisopteromalus calandrae. Insect Biochem Molec Biol. 1998;28(12):1039-50.
24. Jain DK, Jain N, Charde R, Jain N. The RP-HPLC method for simultaneous estimation of esomeprazole and naproxen in binary combination. Pharm Methods. 2011;2(3):167-72.
25. Patel TL, Patel BC, Kadam AA, Tipre DR, Dave SR. Application of novel consortium TSR for treatment of industrial dye manufacturing effluent with concurrent removal of ADMI, COD, heavy metals and toxicity. Water Sci Technol. 2015;71(9):1293-300.
26. del Monte A. Obtención de un nuevo material para ser utilizado como biocatalizador inmovilizado en Química Fina a partir de la anémona marina Stichodactyla helianthus [dissertation]. Havana: University of Havana; 2000.
27. Sabuquillo P, Reina J, Fernandez- Lorente G, Guisan JM, Fernandez-Lafuente R. ‘Interfacial affinity chromatography’ of lipases: separation of different fractions by selective adsorption on supports activated with hydrophobic groups. Biochim Biophys Acta. 1998;1388(2):337-48.
28. Cunha AG, Fernandez-Lorente G, Gutarra ML, Bevilaqua JV, Almeida RV, Paiva LM, et al. Separation and immobilization of lipase from Penicillium simplicissimum by selective adsorption on hydrophobic supports. Appl Biochem Biotechnol. 2009;156(1-3):133-45.
29. Singh RK, Tiwari MK, Singh R, Lee JK. From protein engineering to immobilization: promising strategies for the upgrade of industrial enzymes. Int J Mol Sci. 2013;14(1):1232-77.
30. Fang Y, Huang X-J, Chen P-C, Xu Z-K. Polymer materials for enzyme immobilization and their application in bioreactors. BMB Rep. 2011;44:87-95.
31. Masuda Y, Kugimiya S, Kato K. Improvement of thermal-stability of enzyme immobilized onto mesoporous zirconia. J As Ceram Soc. 2014;2(1):11-9.
32. Tutar H, Yilmaz E, Pehlivan E, Yilmaz M. Immobilization of Candida rugosa lipase on sporopollenin from Lycopodium clavatum. Int J Biol Macromol. 2009;45(3):315-20.
33. Yi SS, Noh JM, Lee YS. Amino acid modified chitosan beads: improved polymer supports for immobilization of lipase from Candida rugosa. J Mol Catal B: Enzym. 2009;57(1-4):123-9.
34. Deng HT, Wang JJ, Liu ZY, Ma M. Influence of varying surface hydrophobicity of chitosan membranes on the adsorption and activity of lipase. J Appl Polym Sci. 2010;115(2):1168-75.
35. Orrego CE, Salgado N, Valencia JS, Giraldo GI, Giraldo OH, Cardona CA. Novel chitosan membranes as support for lipases immobilization: characterization aspects. Carbohydr Polym. 2010;79(1):9-16.
36. Uygun DA, Çorman ME, Öztürk N, Akgöl S, Denizli A. Poly(hydroxyethyl methacrylate-co-methacryloylamidotryptophane) nanospheres and their utilization as affinity adsorbents for porcine pancreas lipase adsorption. Mater Sci Eng C. 2010;30:1285-90.
37. Bolivar JM, Eisl I, Nidetzky B. Advanced characterization of immobilized enzymes as heterogeneous biocatalysts. Catal Today. Forthcoming. doi: 10.1016/j.cattod.2015.05.004.
38. Fernández-Lorente G, Lopez-Gallego F, Bolivar JM, Rocha-Martin J, Moreno- Perez S, Guisán JM. Immobilization of proteins on glyoxyl activated supports: dramatic stabilization of enzymes by multipoint covalent attachment on pre-existing supports. Curr Org Chem 2015;19:1-13.
39. Mateo C, Palomo JM, Fernandez- Lorente G, Guisan JM, Fernandez-Lafuente R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzyme Microb Technol 2007;40(6):1451-63.
40. Aissaoui N, Landoulsi J, Bergaoui L, Boujday S, Lambert JF. Catalytic activity and thermostability of enzymes immobilized on silanized surface: influence of the crosslinking agent. Enzyme Microb Technol. 2013;52(6-7):336-43.
41. Singh P, Gupta P, Singh R, Sharma R. Activity and stability of immobilized alpha-amylase produced by Bacillus acidocaldarius. Int J Pharm Life Sci. 2012;3(12) :2247-53.
42. Benavente R, Pessela BC, Curiel JA, de las Rivas B, Munoz R, Guisan JM, et al. Improving properties of a novel beta-galactosidase from Lactobacillus plantarum by covalent immobilization. Molecules. 2015;20(5):7874-89.
43. EL-Tanash AB, Sherief AA, Nour A. Catalytic properties of immobilized tannase produced from Aspergillus aculeatus compared with the free enzyme. Braz J Chem Eng. 2011;28(3):381-91.
44. Hanušová K, Vápenka L, Dobiáš J, Mišková L. Development of antimicrobial packaging materials with immobilized glucose oxidase and lysozyme. Cent Eur J Chem. 2013;11(7):1066-78.
45. Aissaoui N, Bergaoui L, Boujday S, Lambert J-F, Méthivier C, Landoulsi J. Enzyme immobilization on silane-modified surface through short linkers: fate of interfacial phases and impact on catalytic activity. Langmuir. 2014;30(14):4066-77.
46. Gao Y, Truong YB, Cacioli P, Butler P, Kyratzis IL. Bioremediation of pesticide contaminated water using an organophosphate degrading enzyme immobilized on nonwoven polyester textiles. Enzyme Microb Technol. 2014;54:38-44.
47. Palomo JM, Segura RL, Fernandez- Lorente G, Pernas M, Rua ML, Guisan JM, et al. Purification, immobilization, and stabilization of a lipase from Bacillus thermocatenulatus by interfacial adsorption on hydrophobic supports. Biotechnol Prog. 2004;20(2):630-5.
48. Wang B, Tang X, Liu J, Yu H. Escherichia coli BioH: a highly enantioselective and organic solvent tolerant esterase for kinetic resolution of sec-alcohols. Tetrahedron Lett. 2010;51(48):6360-4.
49. Lo YS, Ibrahim CO. Some characteristics of amberlite XAD-7-adsorbed lipase from Pseudomonas sp. AK. Malays J Microbiol. 2005;1:53-6.
50. Ye P, Xu ZK, Wang ZG, Wu J, Deng HT, Seta P. Comparison of hydrolytic activities in aqueous and organic media for lipases immobilized on poly(acrylonitrile-co-maleic acid) ultrafiltration hollow fiber membrane. J Mol Catal B: Enzym. 2005;32(4):115-21.
51. Fernandez-Lorente G, Terreni M, Mateo C, Bastida A, Fernandez-Lafuente R, Dalmases P, et al. Modulation of lipase properties in macro-aqueous systems by controlled enzyme immobilization: enantioselective hydrolysis of a chiral ester by immobilized Pseudomonas lipase. Enzyme Microb Technol. 2001;28(4-5):389-96.
52. Lozano P, De Diego T, Carrié D, Vaultier M, Iborra JL. Synthesis of glycidyl esters catalyzed by lipases in ionic liquids and supercritical carbon dioxide. J Mol Catal A 2004;214(1):113-9.
53. Sahin O, Erdemir S, Uyanik A, Yilmaz M. Enantioselective hydrolysis of (R/S)-Naproxen methyl ester with sol-gel encapsulated lipase in presence of calix[n]arene derivatives. Appl Catal A. 2009;369(1-2):36-41.
54. Gupta A, Khare SK. Enzymes from solvent-tolerant microbes: useful biocatalysts for non-aqueous enzymology. Crit Rev Biotechnol. 2009;29(1):44-54.
55. Andrade LH, Barcellos T. Lipase-catalyzed highly enantioselective kinetic resolution of boron-containing chiral alcohols. Org Lett. 2009;11(14):3052-5.
56. Jústiz OH, Fernández-Lafuente R, Guisán JM, Negri P, Pagani G, Pregnolato M, et al. One-pot chemoenzymatic synthesis of 3’-functionalized cephalosporines (cefazolin) by three consecutive biotransformations in fully aqueous medium. J Org Chem. 1997;62(26):9099-106.
57. Tan T, Chen B, Ye H. Enzymatic synthesis of 2-ethylhexyl palmitate by lipase immobilized on fabric membranes in the batch reactor. Biochem Eng J 2006;29(1-2):41-5.
58. Nawani N, Singh R, Kaur J. Immobilization and stability studies of a lipase from thermophilic Bacillus sp: The effect of process parameters on immobilization of enzyme. Electron J Biotechnol 2006;9(5):559-65.
59. Cabrera Z, Fernandez-Lorente G, Fernandez-Lafuente R, Palomo JM, Guisan JM. Novozym 435 displays very different selectivity compared to lipase from Candida antarctica B adsorbed on other hydrophobic supports. J Mol Catal B Enzym. 2009;57(1-4):171-6.
60. Yilmaz E, Can K, Sezgin M, Yilmaz M. Immobilization of Candida rugosa lipase on glass beads for enantioselective hydrolysis of racemic naproxen methyl ester. Bioresour Technol. 2011;102(2):499-506.
61. Yilmaz E, Sezgin M, Yilmaz M. Enantioselective hydrolysis of racemic naproxen methyl ester with sol-gel encapsulated lipase in the presence of sporopollenin. J Mol Catal B Enzym. 2010;62(2):162-8.
62. Wang B, Jiang L, Wang J, Ma J, Liu M, Yu H. A tandem and fully enzymatic procedure for the green resolution of chiral alcohols: acylation and deacylation in non-aqueous media. Tetrahedron. 2011;22(9):980-5.
63. Bornscheuer UT. Methods to increase enantioselectivity of lipases and esterases. Curr Opin Biotechnol. 2002;13(6):543-7.
64. Segura RL, Palomo JM, Mateo C, Cortes A, Terreni M, Fernandez-Lafuente R, et al. Different properties of the lipases contained in porcine pancreatic lipase extracts as enantioselective biocatalysts. Biotechnol Prog. 2004;20(3):825-9.
65. Yu HW, Chen H, Yang YY, Ching CB. Effect of salts on activity, stability and enantioselectivity of Candida rugosa lipase in isooctane. J Mol Catal B Enzym. 2005;35(1-3):28-32.
66. Palomo JM, Segura RL, Fernández-Lorente G, Guisán JM, Fernández-Lafuente R. Enzymatic resolution of (±)-glycidyl butyrate in aqueous media. Strong modulation of the properties of the lipase from Rhizopus oryzae via immobilization techniques. Tetrahedron: Asymmetry. 2004;15(7):1157-61.
67. Palomo JM, Fernández-Lorente G, Mateo C, Ortiz C, Fernández-Lafuente R, Guisán JM. Modulation of the enantioselectivity of lipases via controlled immobilization and medium engineering: hydrolytic resolution of Mandelic acid esters. Enzyme Microb Technol. 2002;31(6):775-83.
68. Ong AL, Kamaruddin AH, Bhatia S, Aboul-Enein HY. Enantioseparation of (R,S)-ketoprofen using Candida antarctica lipase B in an enzymatic membrane reactor. J Sep Sci. 2008;31(13):2476-85.
69. Wang PY, Tsai SW, Chen TL. Improvements of enzyme activity and enantioselectivity via combined substrate engineering and covalent immobilization. Biotechnol Bioeng. 2008;101(3):460-9.
70. Kasche V, Haufler U, Riechmann L. Equilibrium and kinetically controlled synthesis with enzymes: semisynthesis of penicillins and peptides. Methods Enzymol. 1987;136:280-92.
Received in June 2015.
Accepted in October 2015.
Alberto del Monte-Martínez. Laboratorio de Tecnología Enzimática, Centro de Estudio de Proteínas, Facultad de Biología, Universidad de La Habana, Calle 25 #455 entre I y J, Vedado, CP 10400, La Habana, Cuba. E-mail: adelmonte@fbio.uh.cu.














