INTRODUCTION
Cenchrus purpureus cv. Cuba CT-115 is widely used in Cuba for its favorable growth characteristics, such as lower resistance to cutting, greater amount of leaves, less height than King grass and shorter internode distance as age progresses. For this reason, it offers better possibilities for harvesting as a biomass bank, including grazing (Crespo and Martínez 2016).
Mineral composition of grass is very important from the physiological point of view since it is essential to establish efficient systems of nutrition and plant management (Bloom and Smith 2014 and Rogóz and Tabak 2017). It also constitutes an essential source for the basic supply of mineral elements, which are necessary for animal feed (French 2017 and Villalobos and Sánchez 2018).
Taking these aspects into account, the objective of this study was to analyze the composition of some mineral nutrients of C. purpureus cv. Cuba CT-115 when used as a biomass bank.
MATERIALS AND METHODS
Location, weather and soil. The research began with the determination of homogeneity of the area and the way to perform sampling that would allow to obtain reliable, accurate, repeatable and representative results (Fortes et al. 2007). The study was conducted in the dairy B of the Institute of Animal Science (ICA, in Spanish), San José de las Lajas, Mayabeque, located between 22º 53 N and 82º 02 W, at 80 m.o.s.l. (Anon 1989), in a previously established paddock of Cenchrus purpureus cv. Cuba CT-115.
Soil was classified as fluffy gray brown according to Hernández et al. (2015). Before starting the experimental stage, 10 soil samples were taken in the diagonals of the paddock length, between 0 and 15 cm deep, with a helicoidal auger. They were air dried, homogenized in a mortar and passed through a sieve with 0.5 mm mesh. They were packed in glass flasks with hermetic seal and stored at room temperature until analysis. Organic matter (Walkley and Black, cited by Jackson 1970), nitrogen (AOAC 2016), phosphorus (Oniani 1964), calcium and magnesium (Maslova, cited by Paneque 1965) were determined.
Chemical composition of soil appears in table 1, data presented are the means of composite samples that were taken. The pH was slightly acid and N and K contents were relatively low (Fortes 2013).
Table 1 Chemical composition of soil of the experimental area
| % | mg.100g -1 | pH | ||||
|---|---|---|---|---|---|---|
| N | OM | Ca | Mg | P | K | |
| 0.19 | 3.20 | 2.53 | 0.26 | 2.15 | 5.44 | 5.87 |
Figure 1 shows the accumulated rainfall and mean, minimum and maximum monthly temperature, during the year of experimentation, data taken from the ICA Meteorological Station.
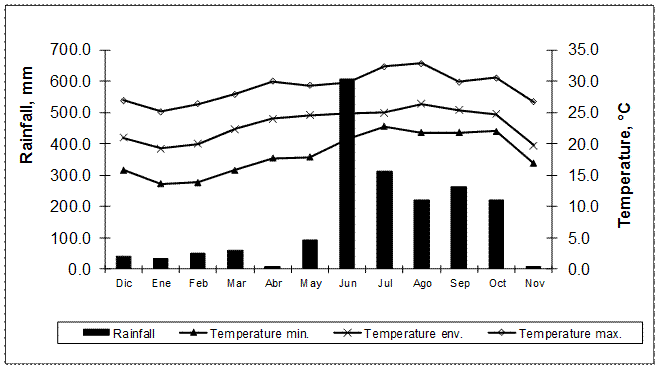
Figure 1 Performance of the accumulate of precipitations and temperatures during the experimental period
Treatments and design. Treatments consisted on regrowth ages or grass rest from the time of departure of the animals from the paddock (time zero), as follows: 0, 15, 30, 45, 60, 75 and 90 days of regrowth of grazing cycle one; 0, 15, 30, 45 and 60 days of regrowth of cycle two; 0, 15, 30 and 45 days of regrowth of grazing cycle three and 0, 15, 30, 45, 60, 75, 90 and 105 days of regrowth of grazing cycle four (table 2); and according to the biomass bank technology (Martínez and Herrera 2006). For sample taking, a homogeneity study of the selected area was carried out and the appropriate sample size was determined for the morphophysiological study of Cenchrus purpureus cv. Cuba CT-115 in grazing. Results showed that the area turned out to be homogeneous, and it was determined that 15 samples (tillers as experimental units) are sufficient to accurately indicate the performance of the indicators of the studied population (Fortes et al. 2007).
Table 2 Distribution of rest cycles during the experiment
| Rest cycles | Date | Duration, days |
|---|---|---|
| Cycle 1 | December 2006- March 2007 | 90 |
| Cycle 2 | March-May 2007 | 60 |
| Cycle 3 | May-July 2007 | 45 |
| Cycle 4 | August- November 2007 | 105 |
Procedure. A paddock of 0.75 ha was integrated to the technology of biomass banks (Martínez and Herrera 2006). Fifteen samples (tillers as experimental units) were selected, at the previously mentioned regrowth ages for each grazing cycle. Tillers were always divided into residue (rejection with their new growth) and regrowth (tiller). Then, they were separated into leaf, stem and dead material (DM) of the residue, and leaf and stem of the regrowth.
For chemical analysis, samples were dried at 60 °C in a Memmert air circulation oven and ground in a hammer mill (Culatte typs MFC) with a 1 mm diameter sieve, packed in hermetically sealed glass flasks and stored at room temperature until their processing.
Contents of ash, nitrogen (N), calcium (Ca), phosphorus (P) and magnesium (Mg) were determined according to the methodology described by the AOAC (2016).
Statistical analysis. Analysis of variance was carried out according to completely randomized design and Duncan (1955) test was used for the comparison of means in the necessary cases. All analyzes were performed by duplicate per treatment. For data processing, the statistical package IBM-SPSS, version 22 (2013) was used.
RESULTS AND DISCUSSION
Table 3 shows ash contents for all grazing cycles during the evaluation year. Differences (P<0.01) were observed among regrowth ages for leaves and stems of tillers and residue, except in the leaves of tillers of grazing cycle three. Ashes of leaves were generally higher than stems, and the values ranged between 3.89% and 15.41%. Herrera et al. (2002), when studying this indicator in CT-115 under grazing conditions, found higher ash contents in leaves than in stems and varied between 8-9% and from 4.5 to 7%, respectively. On the other hand, Valenciaga et al. (2001) reported lower values of 4.01% in leaves and higher, in stem, of 7.04%, which could be due to the different management conditions.
Table 3 Contents of ash (%) in Cenchrus purpureus cv. Cuba CT-115
| Cycle 1 (December-March) | ||||
|---|---|---|---|---|
| Age, Days | Tillers | Residue | ||
| Ash leaf | Ash stem | Ash leaf | Ash stem | |
| 0 | - | - | 12.97ab | 4.02ab |
| 15 | 9.57ª | 5.10ª | 12.34ª | 3.89ª |
| 30 | 8.94ab | 8.62bc | 12.29ª | 4.94ab |
| 45 | 8.76ab | 8.16bc | 12.37ª | 4.45ab |
| 60 | 8.44b | 8.94c | 12.50ª | 4.89ab |
| 75 | 8.56b | 8.30c | 14.21b | 5.25b |
| 90 | 8.52b | 6.95ab | 12.77ª | 4.69ab |
| SE±/Sign. | 0.23** | 0.28*** | 0.35*** | 0.21*** |
| Cycle 2 (March-May) | ||||
| 0 | - | - | 11.33ª | 6.35ª |
| 15 | - | - | 11.83ab | 6.32ª |
| 30 | 10.52ª | 5.02ª | 12.49bc | 5.43b |
| 45 | 10.25ab | 9.34b | 12.78c | 5.50b |
| 60 | 8.96b | 10.34b | 11.96abc | 5.67ab |
| SE±/Sign. | 0.14*** | 0.20*** | 0.18** | 0.17** |
| Cycle 3 (May-July) | ||||
| 0 | - | - | - | 4.47ª |
| 15 | 12.05 | 5.37ª | 13.77ª | 5.00ab |
| 30 | 12.33 | 6.17ª | 14.03ab | 6.11bc |
| 45 | 11.89 | 8.27b | 15.12bc | 6.25c |
| 60 | 11.25 | 7.65b | 15.35c | 6.53c |
| SE±/Sign. | 0.34 | 0.30** | 0.23** | 0.25** |
| Cycle 4 (August-November) | ||||
| 0 | - | - | 15.41ª | 5.48abc |
| 15 | 11.52ab | 5.85ab | 14.27ab | 6.71ª |
| 30 | 12.08b | 8.13bc | 13.85ab | 6.16ab |
| 45 | 11.53ab | 9.25c | 13.13b | 6.64ª |
| 60 | 12.30b | 7.07abc | 14.67ab | 4.58bc |
| 75 | 10.17ª | 4.34ª | 13.91ab | 4.11c |
| 90 | 11.15ab | 5.65ab | 12.91b | 4.91abc |
| 105 | 11.16ab | 5.32ab | 13.50ab | 4.65bc |
| EE±/Sign. | 0.25*** | 0.43*** | 0.32*** | 0.26*** |
abc Values with different letters per column differ at P <0.05 (Duncan 1955)
***P <0.001 ** P <0.01
On the other hand, Valenciaga et al. (2009) found ash values in C. purpureus cv. Cuba CT-115 that increased with age, up to 12.64% for 140 days of regrowth. However, in this study, the contents were higher than those reported by these authors and the performance with age was variable. On the other hand, Correa et al. (2016) obtained higher ash content in Kikuyo grass at 45 days than at 80 days of regrowth; while Kozloski et al. (2005), in samples of C. purpureus cv. Mott harvested between 30 and 90 days of regrowth, did not find large differences in this indicator.
Figure 2 shows nitrogen contents for all studied grazing cycles. Nitrogen had higher value in leaves than in stems in tillers and in the residue. Nitrogen content had a tendency of reduction with age in the plant.
This occurs because this nutrient has greater importance in the younger parts. Its absorption gradually decreases when the plant physiologically ages or reaches maturity. Gándara et al. (2017) also found reductions in nitrogen and protein with grass age. In this sense, Pérez et al. (2004) state that nitrogen content in plants decreases during growth, and has a high correlation with the accumulation of dry matter, more than other indicators and for any growth stage and regrowth age. These authors found reductions in N contents in Brachiaria hibrido leaves as the age of the plant progressed.
Table 4 shows calcium contents in the four grazing cycles of the technology. In all cases, differences (P <0.05) were observed with regrowth age, in leaves and stems, except in the leaf of residue of grazing cycle one, the values ranged between 0.30 and 0.83% of DM. Despite the significant differences found among ages, their variation was in a narrow range. In this sense, Valenciaga et al. (2009) found no differences among regrowth ages for this indicator in Cuba CT-115. Calcium contents were always superior in sheets at 0.30% of DM, level established by Cunha and Mc Dowell (2012) as critical for the animal feeding. Similar results were reported by Clavero et al. (1994).
Table 4 Calcium content (%) in leaves and stems of Cenchrus purpureus cv. Cuba CT-115
| Cycle 1 (December-March) | ||||
|---|---|---|---|---|
| Age, days | Tillers | Residue | ||
| Ca Leaf | Ca Stem | Ca Leaf | Ca Stem | |
| 0 | - | - | 0.79 | 0.50ª |
| 15 | 0.65ab | 0.47ª | 0.82 | 0.54ab |
| 30 | 0.69bc | 0.57b | 0.82 | 0.62c |
| 45 | 0.70c | 0.54b | 0.83 | 0.58bc |
| 60 | 0.64ª | 0.58b | 0.82 | 0.60bc |
| 75 | 0.65ab | 0.53b | 0.79 | 0.64c |
| 90 | 0.64a | 0.57b | 0.82 | 0.65c |
| SE±/Sign. | 0.01*** | 0.01*** | 0.01 | 0.01*** |
| Cycle 2 (March-May) | ||||
| 0 | - | - | 0.41ª | 0.34ª |
| 15 | - | - | 0.42ª | 0.40c |
| 30 | 0.50ª | 0.32ab | 0.43ª | 0.36ab |
| 45 | 0.63ab | 0.29ª | 0.47b | 0.34ª |
| 60 | 0.62b | 0.38b | 0.47b | 0.371bc |
| SE±/Sign. | 0.01*** | 0.01*** | 0.004*** | 0.01** |
| Cycle 3 (May-July) | ||||
| 0 | - | - | - | 0.37ab |
| 15 | 0.63ª | 0.45ª | 0.48ª | 0.39b |
| 30 | 0.63ª | 0.30b | 0.46ª | 0.34ab |
| 45 | 0.57b | 0.31b | 0.35b | 0.31ª |
| 60 | 0.49c | 0.35b | 0.40b | 0.36ab |
| SE±/Sign. | 0.01*** | 0.01** | 0.01** | 0.01*** |
| Cycle 4 (August-November) | ||||
| 0 | - | - | 0.60ab | 0.38ª |
| 15 | 0.52ª | 0.35ab | 0.62b | 0.36ab |
| 30 | 0.54ª | 0.37b | 0.62b | 0.37ª |
| 45 | 0.55ab | 0.37b | 0.63b | 0.36ab |
| 60 | 0.51ª | 0.35b | 0.58ª | 0.30bc |
| 75 | 0.61b | 0.31ª | 0.63b | 0.30c |
| 90 | 0.54ª | 0.32ab | 0.63b | 0.33abc |
| 105 | 0.52a | 0.33ab | 0.62b | 0.31bc |
| SE±/Sign. | 0.01*** | 0.01** | 0.01* | 0.01** |
abc Values with different letters per column differ at P <0.05 (Duncan 1955)
***P <0.001 **P <0.01 * P <0.05
Values of calcium contents found in the leaf were always higher than in the stems. Similar results to these were reported by Herrera et al. (2008) for all the studied Cenchrus varieties including that of this study. This performance is contradictory, taking into account that calcium plays an important role as a cementing element in the cell wall and it is precisely in the leaves, where the lowest cell wall content is found.
For all grazing cycles, phosphorus contents (table 5) showed differences (P <0.05) of this indicator with regrowth age. In the leaves, phosphorus contents showed a reduction (P<0.01) with the age of the plant, except for grazing cycle one, values ranged between 0.17 and 0.43% of DM. In the case of stems, there was no fixed tendency to increase or decrease with age, but in all cases the contents were variable and ranged between 0.15 and 0.26% of DM. In the literature, reductions of this element are reported with the age of the plant (Santiago et al. 2016) However, in this study, the performance was variable.
Table 5 Phosphorus content (%) in leaves and stems of Cenchrus purpureus cv. Cuba CT-115
| Cycle 1 (December-March) | ||||
|---|---|---|---|---|
| Age, days | Tillers | Residue | ||
| P Leaf | P Stem | P Leaf | P Stem | |
| 0 | - | - | 0.24abc | 0.19ª |
| 15 | 0.24ab | 0.19ª | 0.26c | 0.23ab |
| 30 | 0.21ª | 0.19ª | 0.21ª | 0.21ab |
| 45 | 0.21ª | 0.21ab | 0.23ab | 0.21ab |
| 60 | 0.26b | 0.25c | 0.25bcd | 0.24b |
| 75 | 0.23ª | 0.25bc | 0.24abc | 0.22ab |
| 90 | 0.23ab | 0.24bc | 0.25bc | 0.21ab |
| SE±/Sign. | 0.01*** | 0.01*** | 0.01*** | 0.01*** |
| Cycle 2 (March-May) | ||||
| 0 | - | - | 0.33ª | 0.29ª |
| 15 | - | - | 0.28c | 0.17b |
| 30 | 0.35ª | 0.25ª | 0.29c | 0.24ab |
| 45 | 0.27b | 0.26ª | 0.32ab | 0.23ab |
| 60 | 0.25b | 0.22b | 0.30bc | 0.24b |
| SE±/Sign. | 0.01*** | 0.01* | 0.01** | 0.01** |
| Cycle 3 (May-July) | ||||
| 0 | - | - | - | 0.19ab |
| 15 | 0.32ª | 0.20ª | 0.43ª | 0.19ab |
| 30 | 0.33ª | 0.22ª | 0.34ab | 0.21b |
| 45 | 0.27b | 0.20ª | 0.26bc | 0.18ª |
| 60 | 0.24b | 0.24b | 0.24c | 0.20ab |
| SE±/Sign. | 0.01** | 0.01** | 0.01*** | 0.01** |
| Cycle 4 (August-November) | ||||
| 0 | - | - | 0.23ab | 0.20ab |
| 15 | 0.24ª | 0.21ª | 0.25ª | 0.15ª |
| 30 | 0.23ab | 0.18ab | 0.23ab | 0.18ab |
| 45 | 0.23ab | 0.16b | 0.21abc | 0.19ab |
| 60 | 0.23ab | 0.20ª | 0.22abc | 0.21b |
| 75 | 0.20b | 0.16b | 0.19bc | 0.17ab |
| 90 | 0.21ab | 0.19b | 0.17c | 0.18ab |
| 105 | 0.20b | 0.19ab | 0.19bc | 0.20ab |
| SE±/Sign. | 0.01** | 0.01** | 0.01*** | 0.01*** |
abcValues with different letters per column differ at P <0.05 (Duncan 1955)
***P <0.001 **P <0.01 * P <0.05
Leaves had higher phosphorus values than stems in general. This performance is logical taking into account the multiple functions that this element develops in plant metabolism, since it is part of a wide range of molecules such as phosphate sugars, nucleic acids, coenzymes, and some others, in addition to controlling different metabolic processes that occur primarily in the leaves (Gardner et al. 2017 and Malhotra et al. 2018).
Phosphorus requirements for optimal plant growth are in the range of 0.3 to 0.5% of dry matter for the period of plant growth according to Vistoso et al. (2017). However, Epstein and Bloom (2005), as well as Dominguez et al. (2012) consider that a content superior to 0.2% in the dry mass constitutes an adequate tissue level of phosphorus in plants. Taking this into consideration for some regrowth ages, phosphorus was below the appropriate levels for the metabolism so it could be suggested the use of phosphorus fertilization to the soil to fulfill this deficiency in the plant. For animal feed, values lower than 0.25% of DM are considered as critical level (Cunha and Mc Dowell 2012). In some grazing cycles, values below this were found, so it is possible that some type of supplementation is needed in animals to cover this deficit.
Magnesium contents were always higher in leaves than in stems (table 6). Chloroplasts of plants are rich in Mg because the main function of this element is its role as a central atom of chlorophyll molecule present mainly in leaves (Basantes 2016 and Chen et al. 2018). Values were between 0.25 and 0.52% for the leaves and 0.16 and 0.30% for the stems. According to Torres (1999), contents of 0.2% of magnesium in dry matter are considered acceptable concentrations of this element for the good physiological functioning of the plant. For this reason, concentrations found in this study, in some cases, are below those required by the plant, although the difference between the requirements and the found Mg contents is very low.
Table 6 Magnesium content (%) in leaves and stems of C. purpureus cv. Cuba CT-115
| Cycle 1 (December-March) | ||||
|---|---|---|---|---|
| Age, days | Tillers | Residue | ||
| Mg Leaf | Mg Stem | Mg Leaf | Mg Stem | |
| 0 | - | - | 0.40ª | 0.24ª |
| 15 | 0.33bc | 0.30b | 0.47b | 0.22ª |
| 30 | 0.35c | 0.36c | 0.52b | 0.26ab |
| 45 | 0.37c | 0.36c | 0.48b | 0.30b |
| 60 | 0.26ª | 0.29ab | 0.47b | 0.25ª |
| 75 | 0.27ab | 0.25ab | 0.47b | 0.25ª |
| 90 | 0.25a | 0.25a | 0.38a | 0.26ab |
| SE±/Sign. | 0.01*** | 0.01*** | 0.01*** | 0.01*** |
| Cycle 2 (March-May) | ||||
| 0 | - | - | 0.28ab | 0.26bc |
| 15 | - | - | 0.29abc | 0.28c |
| 30 | 0.32ª | 0.28ª | 0.28ª | 0.28c |
| 45 | 0.42ab | 0.22b | 0.30bc | 0.20ª |
| 60 | 0.49b | 0.23b | 0.31c | 0.22ab |
| SE±/Sign. | 0.04* | 0.01** | 0.01* | 0.01** |
| Cycle 3 (May-July) | ||||
| 0 | - | - | - | 0.24ª |
| 15 | 0.50ª | 0.27ab | 0.38ª | 0.25ab |
| 30 | 0.48ª | 0.29b | 0.39ª | 0.26ab |
| 45 | 0.29b | 0.29b | 0.29b | 0.26b |
| 60 | 0.30b | 0.25a | 0.30b | 0.24b |
| SE±/Sign. | 0.01*** | 0.01** | 0.01*** | 0.01* |
| Cycle 4 (August-November) | ||||
| 0 | - | - | 0.29a | 0.23abc |
| 15 | 0.30 a | 0.19ab | 0.36ab | 0.24bc |
| 30 | 0.33 ab | 0.23ab | 0.36ab | 0.22abc |
| 45 | 0.34 ab | 0.25b | 0.37b | 0.25c |
| 60 | 0.32 ab | 0.23ab | 0.34ab | 0.21abc |
| 75 | 0.34 ab | 0.25b | 0.36ab | 0.22abc |
| 90 | 0.35 ab | 0.16a | 0.37b | 0.17ab |
| 105 | 0.37 b | 0.16a | 0.38b | 0.16a |
| SE±/Sign. | 0.01*** | 0.01*** | 0.01*** | 0.02** |
abc Values with different letters per column differ at P <0.05 (Duncan 1955)
***P <0.001 **P <0.01 *P <0.05
On the other hand, Cunha and Mc Dowell (2012) considered 0.20% of DM as a critical level for animal feed, so that for some ages of the plant, the results were below this value.
It is concluded that contents of ash, calcium, phosphorus and magnesium had a variable performance with the regrowth age for all grazing cycles of biomass bank technology. Nitrogen content was reduced with the age of regrowth in all the fractions of the plant studied. The studied indicators had higher values in leaves than in stems for tillers and residue with their new growth. Phosphorus and magnesium contents found in leaves and stems for some regrowth ages were lower than those required for proper growth and development of the grass, so the use of maintenance fertilization might be necessary. It is recommended to use these results to design other management options.











 texto en
texto en 



