Introduction
Endo-periodontal lesions are clinical conditions that affect both the pulp and periodontal tissues and are categorized as acute or chronic. Associated etiological factors include live and dead pathogens and other contributing factors; the affected regions include dentinal tubules, accessory ducts, lateral canals, and the apical foramen. The most common signs and symptoms of endo-periodontal lesions are deep periodontal pockets in or near the root apex, negative or altered response in pulp vitality testing, bone resorption in the root and furcation areas, spontaneous pain or tenderness on palpation and percussion, purulent exudate, tooth mobility, discharging fistula, and changes in the color of the crown and/or gums. The classification system mainly used in dental consultations in the past few years was published in 1972 by Simon and others1 but has been criticized because the categories are based on the primary source of infection (root canal or periodontal pocket), In most of the cases, a complete history is not available. Therefore, a simplified classification relying on the current disease status and prognosis of the affected tooth was proposed.2
The management of endo-periodontal lesions involves the endodontic and periodontal treatment, which ranges from the very evaluation of the preoperative prognosis up to proving treatment with conservative approaches such as root canal treatment combined with basic periodontal therapy, including surgical treatment with or without periodontal regeneration (PR) and even tooth extraction.3
Given the wide range of treatments, the objective of this case report is to demonstrate that surgical periodontal therapy aided by gelatine sponges containing colloidal silver nanoparticles restores lost bone in endo-periodontal lesions.
Case presentation
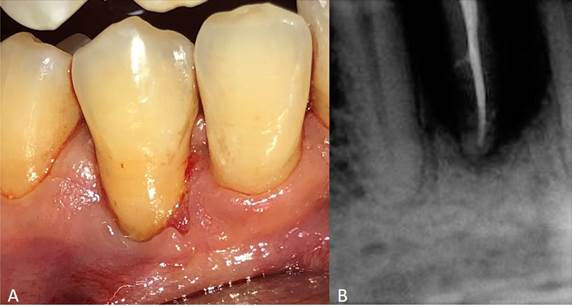
The patient was a 55-year-old man without systemic alterations. In February 2017, he was treated with scaling and root planning. This patient came back 11 months after for periodontal re-evaluation. He was presented with inflammation in the lower right region without pain. Clinical examination showed a purulent discharge in the gingival sulcus in tooth 44. Periodontal probing in the vestibular area indicated a periodontal pocket of 13 mm in the middle aspect and 5 mm on the mesial and distal aspects of this tooth. However, tooth mobility, occlusal trauma, and periodontal pockets on the lingual aspect of this tooth were not observed.
A periapical radiograph showed vertical bone loss of > 80% on the mesial and distal aspects; sensitivity test results indicated that the tooth was insensitive to cold stimulation.
The patient was referred to the endodontic service, which confirmed that the tooth was insensitive to percussion. Exploratory surgery was recommended to rule out a cracked root. The exploratory surgery ruled out fracture or crack of the root and showed calcified plaques and granulomatous tissue. The tissue flaps were repositioned and sutured.
Periodontal lesions were diagnosed with grade 3 endo-periodontal lesions following Herrera and others2 The prognosis was classified as class D according to Samet and others.4) with bone loss > 80%, pulp necrosis, and a high likelihood of losing the affected tooth. However; the surgical team together with the patient decided to keep the tooth. Besides the patient’s approval, the following clinical characteristics were considered: 1) adequate height of the crestal bone on the adjacent teeth, 2) lack of tooth mobility, and 3) lack of periodontal pockets on the lingual aspect.
The root canal treatment was performed in February 2018 (fig. 1). It still presented purulent discharge in the gingival sulcus and a periodontal probe checked a subgingival biofilm eight days after the intervention.
Hence, periodontal surgery using ultrasonic and hand instrumentation was performed in the area to remove the granulomatous tissue in the affected region. An examination showed a bone defect of > 80%. No tooth mobility was evidence either. Gelatine sponges containing colloidal silver nanoparticles were used to fill the intrabony defect and stabilize the blood clot; the tissue flaps were repositioned and sutured (fig. 2).
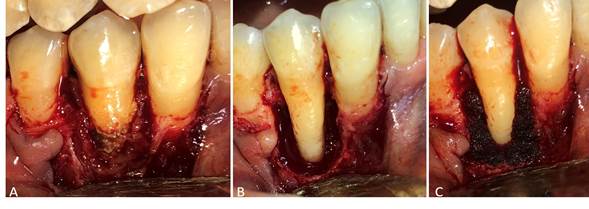
Fig. 2 A. Granulomatous tissue and open-flap debridement, B. Intrabony defect, C. Placement of gelatine sponges containing colloidal silver nanoparticles on the intrabony defect.
Treatment with 500 mg of amoxicillin every 8 hours for 8 days was then initiated. A clinical check-up was conducted 8 days after and then every 3 months. Radiography control was performed at 3, 9, and 24 months after treatment (fig. 3).
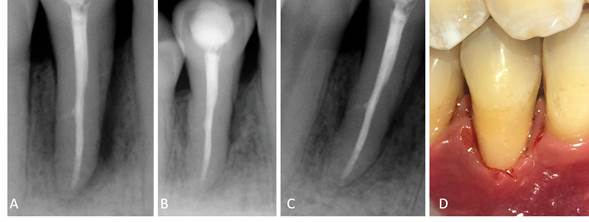
Fig. 3 A. Control radiograph after 3 months, B. After 9 months, C. After 24, D. Clinical aspect after 24 months of treatment.
The rate of bone regeneration was > 70%, no periodontal pockets were observed, and the probing depth was reduced from 13 to 3 mm on the vestibular medial aspect and from 5 mm to 2 mm on the mesial and distal aspects. The average clinical attachment level gain on the medial aspect was 10 mm, and there was no tooth mobility.
Discussion
The initial treatment of teeth with a prognosis class D -as in our patient grade 3 endo-periodontal lesion, characterized by vertical bone loss > 80% in the mesial and distal aspects-is tooth extraction and implant use.5 However, this strategy has increased the number of radical treatments and decreased the number of conservative approaches.6 A conservative approach was used in our patient, and the affected tooth survived.
The treatment of periodontal lesions depends on timely and accurate diagnosis. The initial approach involves root canal disinfection and periodontal therapy. However, root canal treatment alone promotes PR by improving all clinical parameters a few weeks after treatment. Therefore, periodontal therapy with or without surgery is recommended only after assessing the results of endodontic treatment.2
Periodontal disease may cause a total loss of surrounding tissues and damage the pulp. In such cases, root canal disinfection does not achieve complete PR, and periodontal therapy is recommended.7 In our patient, surgery was performed because periodontitis damaged the pulp, and infection continued 8 days after root canal treatment with purulent discharge in the gingival sulcus and subgingival calculus near the apical foramen.
Periodontal lesions lead to intrabony defect formation in the periodontium; therefore, the flap design is vital for the correct treatment and may vary according to the defect. Cortellini and Tonneti8 recommend using a large flap in cases where the defect affects three-quarters of the dental tissue for better visualization. Such a claim matches our results, with bone loss of > 80%. Cortellini and Tonneti8 also found a positive correlation between the success of PR and the number of bone walls. A factor that improved the prognosis of surgical treatment was the presence of two remaining walls formed by the crestal bone on the mesial aspect of tooth 45, the distal aspect of tooth 43, and the lingual surface of these teeth.
Intrabony defects can be treated using various approaches, including surgery alone and guided tissue regeneration (GTR) with barrier membranes, bone grafts, enamel proteins, and platelet-rich fibrin. GTR produces favorable results by preventing the migration of epithelial cells to the affected area, and bone grafts stimulate pathways related to osteoinduction and osteoconduction. The consensus on the GTR of intrabony defects corroborated the clinical effectiveness of different types of treatment. Moreover, surgical debridement alone resulted in significant clinical improvement.8,9
Surgery was performed in this case study since the patient could not afford the regenerative materials. Alternatively, gelatine sponges containing colloidal silver nanoparticles were used to fill the intrabony defects on the mesial and distal aspects of the affected teeth. These sponges were homogeneously distributed throughout the tissue to promote rapid hemostasis, stabilize blood clots, and prevent infections given their long-term antimicrobial activity.10,11,12 Additionally, hydrogels were used to prevent tooth-extraction complications.13Liao and others14 used this material in the alveolar bone of anterior teeth and dental implants after 4 weeks and observed that treatment improved hemostasis and the stability of soft tissues and prevented infections. An animal study focused on the effect on bone defects infected with Staphylococcus aureus and found that treatment with gelatine sponges containing colloidal silver nanoparticles decreased the bacterial count and proinflammatory cytokines such as interleukin-6 and tumor necrosis factor-α. Furthermore, the gelatine sponges containing colloidal silver nanoparticles significantly reduced infections in cranial defects and accelerated bone healing compared to hemostatic sponges without silver nanoparticles.15 However, no studies assessed the effect of hemostatic sponges containing colloid silver nanoparticles on spontaneous bone regeneration in intrabony defects or compared this treatment with other regenerative techniques. The approach adopted in the present study resulted in significant clinical and radiographic improvement and decreased the cost of GTR materials.
Conclusion
This case report suggests the importance of retaining teeth with a diagnosed grade 3 endo-periodontal lesion with a poor prognosis. A conservative approach with a protocol that involved excluding associated etiological factors, thoroughly disinfecting the root canal, assessing the potential of periodontal regeneration of intrabony defects, and gelatine sponges containing colloidal silver nanoparticles to stabilize the clot and sterilize the tissue; resulted in a marked clinical and radiographic improvement after a 24-month follow-up. However, additional scientific evidence is needed to support the use of this treatment.