Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de Protección Vegetal
versión impresa ISSN 1010-2752
Rev. Protección Veg. vol.29 no.2 La Habana Mayo.-ago. 2014
ORIGINAL ARTICLE
Nesidiocoris tenuis Reuter (Hemiptera: Miridae) and Cycloneda sanguinea limbifer (Casey) (Coleoptera: Coccinellidae): behaviour and predatory activity on Myzus persicae Zulzer (Hemiptera: Aphididae)
Nesidiocoris tenuis Reuter (Hemiptera: Miridae) y Cycloneda sanguinea limbifer (Casey) (Coleoptera: Coccinellidae): comportamiento y actividad predadora sobre Myzus persicae Zulzer (Hemiptera: Aphididae)
Leticia DuarteI, Ronal PachecoI, Madelaine QuiñonesI, María de los Ángeles MartínezI, Vanda Helena Paes BuenoII
IDirección de Sanidad Vegetal. Centro Nacional de Sanidad Agropecuaria (CENSA), Apartado 10, San José de las Lajas, Mayabeque, Cuba. Correo electrónico: leticia@censa.edu.cu.
IILaboratorio de Control Biologico. Departamento do Entomologia, Universidade Federal de Lavras, Lavras, Minas Gerais, Brazil.
ABSTRACT
The aim of this work was to evaluate the predation and behavior activities of the predatory bug Nesidiocoris tenuis Reuter and the lady beetle Cycloneda sanguinea limbifer (Casey) on the aphid. Myzus persicae Zulzer, as well as their effect on this prey dispersal. The experiments were carried out in an acclimatized room at 23.7±1, 8oC, RH 62,6±5% and 12 h photofase. The number of aphids on the infested and non-infested plants was recorded after 24 hours. Behavioral activities such as moving, searching, resting, encountering aphids, feeding, encountering aphid exuviae, and cleaning, and the time spent in each one were recorded. C. sanguinea limbifer was more voracious than N. tenuis on M. persicae. The ladybeetle led to more aphid dispersion than N. tenuis; however, the highest aphid dispersion occurred when a higher number of both predatory species was used. Significant differences were not observed in the time spent by both predators in moving, encountering and feeding activities, but the time spent in feeding by N. tenuis was longer than that spent by C. sanguinea limbifer. N. tenuis spent longer time in resting activity than C. sanguinea limbifer (p=0,0088). The lady beetle spent longer time searching for the prey than the predatory bug (p=0,0007), but the time needed by C. sanguinea limbifer for cleaning its mouthparts with its front legs was longer than that used by N. tenuis for cleaning its whole body (p=0,0218). The results showed that M. persicae dispersion was an effect of its antipredator behavior in the presence of the predator.
Key words: biological control, bug, lady beetle, aphid, pepper.
RESUMEN
El objetivo del trabajo fue evaluar las actividades de conducta y depredación de la chinche Nesidiocoris tenuis Reuter y la cotorrita Cycloneda sanguinea limbifer (Casey) sobre el áfido Myzus persicae Zulzer, así como su efecto sobre la dispersión de la presa. Los experiementos se realizaron en una sala climatizada a 23,7±1,8oC, 62,6±5% de HR y fotoperíodo de 12 horas. El número de áfidos encontrado sobre las plantas sanas e infestadas fue registrado a las 24 horas. Se registró la duración del tiempo dedicado en actividades de conducta como: moverse, buscar, descansar, encuentro con áfidos y exuvias y limpieza. C. sanguinea limbifer fue más voraz que N. tenuis sobre M. persicae. La cotorrita permitió mayor dispersión de áfidos que la chince; sin embargo, la mayor dispersión ocurrió cuando un mayor número de depredadores fue usado en ambas especies. El tiempo dedicado en actividades como moverse, encuentros y aliementación en ambos predadores no mostró diferencias significativas, pero el tiempo dedicado por N. tenuis en alimentarse fue mayor al empleado por C. sanguinea limbifer. N. tenuis gasta más tiempo en descansar que C. sanguinea limbifer (p=0,0088). La cotorrita emplea más tiempo en buscar la presa que la chinche (p=0,0007), pero el tiempo necesitado por C. sanguinea limbifer para limpiar sus piezas bucales con sus patas delanteras fue mayor que el usado por N. tenuis para limpiar todo su cuerpo (p=0,0218). Los resultados mostraron que la dispersión de M. persicae es un efecto de su conducta antipredadora, en presencia de los predadores.
Palabras clave: control biológico, chinche, cotorrita, áfido, pimiento.
INTRODUCTION
The family Miridae (plant bugs) represents nearly one-third of the described species within the Heteroptera, with at least one third of them estimated to exhibit predatory habits (1). Nesidiocoris tenuis (Reuter) is one of the representative species of this group, and currently of great interest for being released in augmentative control programs and conservative biological control (2, 3). Several informations concerning its biology, mass rearing, and use as a biological control agent to control Tuta absoluta and Bemisia tabaci in greenhouses are reported so far (2, 4), Also N. tenuis is object of much research because of its duality of presenting a zoophytophagous behavior (5, 6) and also being associated with several pests like whiteflies, thrips, aphids in several crops (7). This predatory bug is common in tomato (Solanum lycopersicum L.), and most of vegetable crops in the Mediterranean area and others with similar climate, such as the Canary Islands (8,9,10). Natural populations of this insect also occur in Cuba (11).
Predatory beetles (Coleoptera: Coccinellidae) have been used in the biological control of several agricultural pests in the world (12). Many aspects of their biology, their use as a biological control agent, the conservation of native species, and pests suppression, such as aphids, scales, eggs of various insects, spider mites, and other pests have been reported (13).
Lady beetles are aphidophagous and highly polyphagous. They consume most (if not all) of aphid species they encounter. However, some results evidence that not every aphid species is equally suitable for every lady beetle species (14). The beetle Cycloneda sanguinea limbifer (Coleoptera: Coccinellidae) is a Cuban native species associated with aphid species of vegetables and other crops (15).
Aphids are one of the major pests of most crops in Cuba. They were reported to limit the development of crops by causing direct damages and transmitting virus diseases (16). Myzus persicae Zulzer is the most important pest associated with many botanical families in Cuba. It is reported on several species of Solanaceae (Solanum tuberosum L., S. lycopersicum, Capsicum annuum L., Solanum melongena L., Nicotiana tabacum L.), Crucciferae (Brassica oleraceae L. var. capitata, Brassica oleracea L. var. botrytis and Brassica oleraceae L. var. italica plenck), Cucurbitaceae (Cucumis melo L. and Cucumis sativus L.): Umbeliferae (Daucus carota L.) and others (17).
The literature on the effects of biological control of aphids on plant virus dispersal suggests that the effect of the natural enemies depends on the mode of the virus transmission, since it determines the time required for virus acquisition and inoculation, and on the level of disturbance (i.e. antipredator behaviour) induced by natural enemies on aphid colonies (18). As C. sanguinea limbifer and N. tenuis are present together in Cuban agroecosystems, and due to the importance of M. persicae as a pest in vegetables crops, the aim of this work was to study the behavior and predator activities of both natural enemies on M. persicae, and also their effect on the prey dispersal. These elements will allow to determine their potential as biological control agents in Cuba under natural or released conditions, and also to evaluate their influence in the pest dispersal.
MATERIALS AND METHODS
All the experiments were carried out at the Laboratory of Entomology of the Plant Health Division in the National Centre for Animal and Plant Health (CENSA), Cuba.
Plant and Insects obtaining
Capsicum annuum (pepper) and S. lycopersicum (tomatoe) plants (California Wonder and Vita varieties, respectively) were used in the tests. The plants were obtained from balled root trays (50 x 30 x 15 cm) placed in a room with natural conditions of temperature and relative humidity. Plants with 30 days of age were transferred to plastic pots (11cm high; diameter 14cm) with sterilized soil and used in the experiments.
Aphids: M. persicae was collected from Solanum melongena (aubergine) in the field from Alamar organic garden in Havana, Cuba. A colony of aphids was maintained on pepper plants in wooden and glass cages (75×90×95 cm) covered with a fine mesh. The aphids used in the experiments came from the second-generation of a stock obtained in the laboratory.
Predators: Adults of N. tenuis, originally collected from tomato plants from Las Papas farm (Mayabeque Province, Cuba), were reared in plastic cages (45×45×70 cm). The insect feeding was supplied with bee pollen from flowers of Bidens alba L. and tomato plants infested with Bemisia tabaci Gennadius (whitefly). The bug predator adults obtained from the second generation of 24 to 48 hours of emergence were transferred to another cage (45×45×70 cm) and fed with aphids of M. persicae on pepper plants 72 hours before of the experiment. Adults of C. sanguinea limbifer were collected from corn plants in Alamar organic garden (Havana, Cuba) and maintained on infested pepper plants with M. persicae in similar plastic cages. All the insects were kept in an acclimatized room at 24±1, 6oC RH 63, 5±5% and photophase of 12 hours to establish a stock colony at the laboratory. Individuals from the second-generation were used in the experiments.
Predation and Aphid dispersal Test
The aphid consumption and effect of predators on dispersal of M. persicae were evaluated as follows: a pepper plant infested with an aphid colony of M. persicae with aproximately 160 aphids (nymphs plus apterous adults) was placed in the center of the wooden and mesh cage (120×60×60 cm). Then, four non-infested pepper plants were placed around this infested plant in circle at a distance of 15cm from the infested plant. After, N. tenuis and C. sanguinea limbifer adults were released manually by opening a plastic tube with the predators on the infested plant.
Five treatments were used with adult predators, which consisted in releasing one N. tenuis, two N. tenuis, one C. sanguinea limbifer, two C. limbifer sanguinea and a control treatment, without predators. For obtaining starving insects at the beginning of the test, the predators were maintained without food for the previous 24 hours. The experimental cages were maintained in an acclimatized room at 23, 7±1, 8æ%C RH 62,6±5% and photophase of 12 hours.
After 24h, the number of aphids on the infested and non-infested plants, and the location of both predators on the plants were recorded. The aphid dispersal was measured by the number of aphids found on the non-infested plants. The predator consumption was determined by the number of aphids that survived on the infested and non-infested plants. Six replications per each treatment (n=6) were included. To know the normal distribution of the data in the experiment, a Shapiro-Wilks test was used. Predator consumption between treatments by ANOVA (p<0.05) was compared and the dispersal aphids was related by KruskalWallis test (p<0.05).
Behaviour of predators. The behaviour of predators feeding on aphids was observed in the laboratory. Both predatory species without food were maintained separately inside of tube glasses for 24h before the test. Only female adults with less than seven days of age were used in the test. The observation arena consisted in a cylindrical glass (7cm high; diameter 9cm) containing pepper leaves infested with approximately 100 mixed stages of M. persicae and covered with a Petri dish (Ø 9,5 cm). A single predator (predatory bug or coccinellid adult) was gently placed in the aphid colony. Subsequently, the behaviour of the predators on the aphids was continuously observed during 60 and 20 minutes for N. tenuis and C. sanguinea limbifer, respectively. For these observations, a photographic camera (Canon) installed on a stereomicroscope (Zeiss Stemi 2000-C) was used, with a magnification of 1x10 and 1,25x10, respectively. Behavioural observations were recorded with the program Etholog 2.2 version (19).
The following behaviour activities of the predators and the duration of each one were recorded: moving (i.e. walking), searching (i.e. pivoting moving and without moving from the same place), resting, encountering aphids (i.e. touching aphids), feeding, encountering aphid exuviae, and cleaning (i.e. cleaning mouthparts with forelegs and/or cleaning another parts of the body). The number of aphids remaining after the observation of the predator behaviour was counted. The methodology was adapted from Belliure et al. (18).
Three treatments were used: predatory bug, coccinellid and control (without predator). Replicates where predation occurred were used for further analysis (10-12 per treatment). A control treatment without predators was used to compare the number of aphids at the beginning of the experiment, after the observation with the predator. The time percentage spent by predators performing each behaviour was compared between treatments using Kruskal-Wallis ANOVA (p<0.05).
RESULTS
Predation and aphid dispersal test
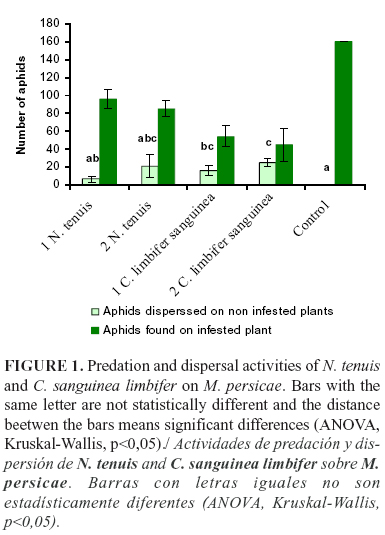
The total number of aphids on the infested plants differed significantly between treatments after 24h of the beginning of the experiment (Fig.1, ANOVA, F=14.99, d.f.=4, p<0.0001). The number of aphids per plant was higher in the control than in the treatments with predator. Among predator treatments, the number of aphids on the infested plant was higher in the presence of N. tenuis than in the presence of C. sanguinea limbifer. In addition, the highest quantity of aphids was found on the infested plant in the treatments where a single (one) predator was used.
Dispersal of M. persicae towards non-infested plants (i.e. total number of aphids on the non-infested plants) was higher with predators than without them (Fig.1, Kruskal-Wallis), with significant differences between the treatments. The coccinellid C. sanguinea limbifer allowed more dispersion of M. persicae than N. tenuis, but the highest aphid dispersion occurred when a higher predator quantity (2 predators/plant) was used for both species. This result showed an antipredator behaviour of the aphids with the predator presence, considering that M. persicae moved to another plant or evaded predator proximity for persisting, mainly when the two predators were present.
Behavior of predators
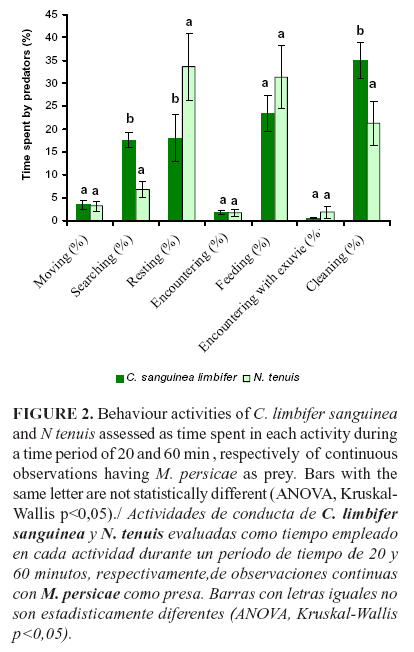
The number of aphids consumed by N. tenuis during 60 min was 1.43, whereas by C. limbifer sanguinea was 11.97 during 20 min. The behavior activities of C. limbifer and N. tenuis during 20 and 60 min, respectively, of continuous observations are shown in Fig. 2.
The time spent in moving, encountering (touching aphids), encountering with exuviae and feeding activities between the predators did not show significant differences (Figure 2). The time spent in feeding by N. tenuis was higher than that spent by C. limbifer sanguinea. N. tenuis needed longer time for resting than C. sanguinea limbifer (p=0,0088). The lady beetle spent longer time in searching preys than the predatory bug (p=0,0007). The time needed by C. sanguinea limbifer for cleaning the mouthparts with it forelegs was longer than that needed by N. tenuis for cleaning its whole body (p=0,0218).
In general, the predatory bug and the coccinellid spent an important part of the time in cleaning (41%) and resting (38%) activities when both were combined. However, C. sanguinea limbifer used longer time performing other activities directly associated with aphid predation (moving, searching, encountering and feeding) than N. tenuis did. The lady beetle started predation activity after 11 seconds of being released in the arena, and they were quicker and more voracious than the mirid. The encountering of C. sanguinea limbifer with M. persicae was more frequent and the time spent by it for taking the first prey was in average 25 seconds. C. sanguinea limbifer consumed approximately 5.4 aphids between cleaning and resting activities. With lady beetles, the first predation event and the first antipredator behavior of M. persicae occurred almost simultaneously, whereas with N. tenuis was not in this way. The stronger antipredator behavior of M. persicae occurred at least 30 minutes after the predator N. tenuis started the attack, and this behavior was mainly showed by aphid nymphs.
In the behavioral observations, N. tenuis started to prey on average 32 min after being released on the aphid colony. The time spent by bugs for taking the first prey was 17.22 min on average, and the previous time before predation were used for moving, cleaning and resting activities. In contrast, with C. sanguinea limbifer, N. tenuis preferred 1st - 3rd instar aphids as prey. The antipredator behavior of the 4 instar nymphs and the aphid adults after the attack of N. tenuis allowed them to disperse and evade predation (i.e. to run away).
DISCUSSION
The evidence on finding a high quantity of aphids on infested plants when N. tenuis was present and a low quantity when the two predators were present suggested that C. sanguinea limbifer was more voracious than N. tenuis when the prey was the aphid M. persicae, and that the quantity of them had an influence on reducing aphid populations. Both natural enemies promoted M. persicae dispersal towards non-infested plants. The predatory bug N. tenuis induced some antipredator behavior of M. persicae (i.e. running away) when it was released and entered into a colony, whereas the coccinellid C. limbifer sanguinea did not seem to be perceived by aphids until the first aphid was attacked. However, when the first predation event occurred, more aphids then dropped off the plant with coccinellids than with mirid bugs. Also the number of aphids performing antipredator behavior during a searching event was higher with coccinellids than with mirid bugs.
These results indicated that C. sanguinea limbifer adults induced antipredator behaviour in M. persicae only after the first attack, and predation by this coccinellid species induced stronger antipredator behavior than predation by mirid bug N. tenuis. The higher number of aphids showing antipredator behavior with coccinellids than with bugs could be due to the longer distance covered by C. sanguinea limbifer or to the faster escape of aphids. In turn, it could enhance an earlier infestation of surrounding non-infested plants and, therefore, increase transmission of the non-persistently transmitted virus by M. persicae.
Belliure et al. (18), reported the antipredator behavior of aphids starting only after the first coccinellid attack, an event associated with the alarm pheromone emission only when the aphids have been physically attacked, and not when they have detected a predator. This phenomenon occurs by (E)-beta-farnesene pheromone in several aphids species, including M. persicae. It is also released as a volatile substance by plant species of several families, including Solanaceae (20), together with other sesquiterpene hydrocarbons that inhibit pheromonal activity. This allows aphids to distinguish between the signal emitted by plants and that from aphids following attack by natural enemies (21).
Furthermore, aphids release a basal level of pheromone also in the absence of predator attack (22). The response of M. persicae to emission of alarm pheromone also results in a higher survival rate in the presence of a coccinellid predator (Hippodamia convergens) (23), and moreover, the alarm pheromone serves as an attractive cue for predators such as coccinellids and other beetles (24). Therefore, it is adaptively beneficial for aphids to emit the alarm pheromone only in case of actual danger (25). This might explain in this study why M. persicae did not react to C. sanguinea limbifer before any aphid was attacked. This behavioural response is consistent with the reported lack of attraction of Adalia bipunctata (L.) towards M. persicae unless aphids are stressed and therefore emitting the alarm pheromone (26,18).
Moreover, observations of natural aphid colonies revealed a collective twitching and kicking responses (CTKR) against natural enemies. In addition, laser vibrometry of the substrate revealed twitching-associated vibrations that formed a train of sharp acceleration peaks in the course of a CTKR. This suggests that visual signals in combination with twitching-related substrate vibrations may play an important role in synchronising defence among individuals of an aphid colony. This cooperative defence behaviour provides an example of a surprising sociality that can be found in some aphid species that are not expected to be social at all (27).
It is known that natural enemies affect aphids not only by causing direct mortality, but also may affect pest dynamics inducing its dispersion to another host as a response to escape from predation. Predators and parasitoids promote dispersal of pests, affect the feeding behavior of their prey, and reduce prey populations by inducing an antipredator behavior (18).
CONCLUSION
These results showed that a higher dispersion of the aphid M. persicae occurs in the presence of the coccinellid C. sanguinea limbifer than with the predatory bug N. tenuis, and that this might be due to the strongest antipredator behaviour induced by C. sanguinea limbifer in comparison with N. tenuis on colonies of M. persicae. However, the highest number of aphids consumed was by C. sanguinea limbifer. Overall, these results highlight the relevance of the studies on plantpest speciesnatural enemy interactions for understanding the epidemiology of vector-borne plant viruses, in the case of M. persicae. Other studies have also pointed out the importance of such studies with parasitoids (28,29). The use of both species in regulating populations of insect pests (30,31) is well known, for this reason, the potential practical implications of these results for the biological control of aphid vectors would be the use of appropriate natural enemies, preferably selecting those predators inducing weaker antipredator responses, when there is a high risk of spread of non-persistent viruses.
REFERENCES
1. Wheeler AG. Predacious plant bugs (Miridae). In Heteroptera of Economic Importance, 2000.
2. Calvo FJ, Lorente MJ, Stansly PA, Belda JE. Preplant release of Nesidiocoris tenuis and supplementary tactics for control of Tuta absoluta and Bemisa tabaci in greenhouse tomato. Entomologia Experimentalis et Applicata. 2012;143(2):111-119.
3. van Lenteren JC. IOBC Internet Book of Biological Control - Version 6. 2012. Available online: http://www.iobcglobal.org/publications_iobc_internet_ book_of_biological_control.html. Consulted 24/3/2014.
4. Bueno VH, van Lenteren JC. Insect Bioecology and Nutrition for Integrated Pest Management (Book). Chapter 22. Predatory Bugs (Heteroptera). 2012; 539-569.
5. Sanchez JA. Density thresholds for Nesidiocoris tenuis (Heteroptera: Miridae) in tomato crops. Biological Control. 2009;51:493-498.
6. Castañé C, Arnó J, Gabarra R, Alomar O. Plant damage to vegetable crops by zoophytophagous mirid predators. Biological Control. 2011;59(1):22-29.
7. Urbaneja A, Calvo J, Bolckmans K, Stansly PA. Predation by Nesidiocoris tenuis on Bemisia tabaci and injury to tomato. BioControl. 2009;54:237-246.
8. Arnó J, Castañé C, Riudavets J, Roig J, Gabarra R. Characterization of damage to tomato plants produced by the zoophytophagous predator Nesidiocoris tenuis. IOBC wprs Bulletin. 2006;29:249-254.
9. Sanchez JA, Martinez-Cascales JI, La casa A. Abundance and wild host plants of predator mirids (Heteroptera: Miridae) in horticultural crops in the Southeast of Spain. IOBC wprs Bulletin. 2003;26:147-151.
10.Trottin-Caudal Y, Fournier C, Leyre JM, Chabriere C. La tomate sous serre dans le Sud-Est de la France. Protection contre la punaise Nesidiocoris tenuis. Infos Ctifl. 2006;224:30-35.
11.Grillo H. Heteropteros de Cuba. Tesis presentada en opción al Título de Doctor en Ciencias. Universidad Central de Las Villas, Cuba. 2012.
12.Gullan PJ, Craston PC. The Insects: An Outline of Entomology. Chapter 16: Pest management. Fourth Edition. Wiley-Blackwell. 2010. 590p.
13.Van Driesche R, Hoddle M. Control of pests and weeds by natural enemies an introduction to biological control. Parte 2, Kids of natural enemies. Chapter 4. Predator diversity and ecology. Blackwell Publishing Ltd. 2008. 502p.
14.Finlayson C, Alyokhin A, Gross S, Porter E. Differential consumption of four aphid species by four lady beetle species. Journal of Insect Science. 2010;10:31. Available online: http://www.insectsicence.org/10.31.
15.Milán O, Cueto N, Hernández N, Ramos T, Pineda MD, Granda R, et al. Prospección de coccinélidos benefices asociados a plagas y cultivos en Cuba. Fitosanidad. 2008;12(2):71-78.
16.Duarte L, Martínez MA, Ceballos M. Aphelinus abdominalis Dalman (Hymenoptera: Aphelinidae): Parámetros biológicos, hospedantes y cultivos asociados. Rev Protección Veg. 2012;27(3):147-150.
17.Duarte L, Martínez MA, Ceballos M, Baños HL, Miranda I, Sánchez A. Biología y tabla de vida de Myzus persicae (Sulzer) (Hemiptera: Aphididae) en condiciones de laboratorio. Rev Protección Veg. 2011;26(1):1-4.
18.Belliure B, Amorós-Jiménez R, Fereres A, Marcos-García MA. Antipredator behaviour of Myzus persicae affects transmission efficiency of Broad bean wiltvirus 1. Virus Research. 2011;159:206-214.
19.Ottoni EB. EthoLog 2.2: A tool for the transcription and timing of behaviour observation sessions. Behav Res Methods Instrum Computers. 2000;32(3):446-449.
20.Agelopoulos NG, Hooper AM, Maniar SP, Pickett JA, Wadhams LJ. A novel approach for isolation of volatile chemicals released by individual leaves of a plant in situ. J Chem Ecol. 1999;25:1411-1425.
21.Dawson GW, Griffiths DC, Merritt LA, Mudd A, Pickett JA, et al. Aphid semiochemicals: a review, and recent advances on the sex pheromone. J Chem Ecol. 1990;16:3019-3030.
22.Almohamad R, Verheggen FJ, Francis F, Lognay G, Haubruge E. Emission of alarm pheromone by non-preyed aphid colonies. J Appl Entomol. 2008;132:601-604.
23.De Vos M, Cheng WY, Summers HE, Raguso RA, Jander G. Alarm pheromone habituation in Myzus persicae has fitness consequences and causes extensive gene expression changes. Proc Natl Acad Sci. U.S.A. 2010;107:14673-14678.
24.Verheggen FJ, Fagel Q, Heuskin S, Lognay G, Francis F, Haubruge E. Electrophysiological and behavioral responses of the multicolored Asian lady beetle, Harmonia axyridis Pallas, to sesquiterpene semiochemicals. J Chem Ecol. 2007;33:2148-2155.
25.Fievet V, Lhomme P, Outreman Y. Predation risk cues associated with killed conspecifics affect the behavior and reproduction of prey animals. Oikos. 2008;117:1380-1385.
26.Francis F, Lognay G, Haubruge E. Olfactory responses to aphid and host plant volatile releases: (E)-Farnesene an effective kairomone for the predator Adalia bipunctata. J Chem Ecol. 2004;30:741-755.
27.Hartbauer M. Collective Defense of Aphis nerii and Uroleucon hypochoeridis (Homoptera, Aphididae) against Natural Enemies. PLoS ONE. 2010;5(4):e10417.
28.Hodge S, Hardie J, Powell G. Parasitoid said dispersal of an on persistently transmitted plant virus by disturbing the aphid vector. Agric Forest Entomol. 2010;13:83-88.
29.Jeger MJ, Chen Z, Powell G, Hodge S, van den Bosch F. Interactions in a host plant-virus-vector-parasitoid system: modeling the consequences for virus transmission and disease dynamics. Virus Research. 2011;159:183-193.
30.Perdikis D, Alomar O. Heteropteran Predators and Their Role in Biological Control in Agroecosystems. Biological Control. 2011;59(1):1-68.
31.van Lenteren JC. The state of commercial augmentative biological control: plenty of natural enemies, but a frustrating lack of uptake. BioControl. 2012;57:1-20.
Recibido: 7-3-2014.
Aceptado: 6-5-2014.