Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Biotecnología Aplicada
versión On-line ISSN 1027-2852
Biotecnol Apl vol.31 no.1 La Habana ene.-mar. 2014
REVIEW
Uses of immunoglobulin A in the control of the infectious diseases
Usos de la Inmunoglobulina A en el control de las enfermedades infecciosas
Nadine Alvarez1, María E Sarmiento1, Norazmi Mohd-Nor2, Armando Acosta1
1 Vicepresidencia de Investigaciones y Desarrollo, Instituto Finlay. Ave. 27 No. 19805, La Lisa, PO Box 16017, CP11600, La Habana, Cuba.
2 School of Health Sciences, University Sains Malaysia. 16150 Kubang Kerian, Kelantan, Malaysia.
ABSTRACT
The main door of entrance for most of pathogens is the mucosal route. As known, one of the main immunological elements at this level is the immunoglobulins and within them, immunoglobulin A (IgA) is the most abundant. Its role in the protection against different pathogens has been demonstrated from observations in experimental models and humans, where its effect was evaluated from the prophylactic and therapeutic points of view. However, there are still many infectious diseases for which the role of IgA has not been studied so far and this field remains open for future research and potential applications to diseases for which there are no efficient methods of control. Here we focus on accumulative evidences of the uses of IgA in the control of infectious diseases, derived from experimental observations in animal models and humans. The main source of information was derived from papers published related with the subject, included in Pubmed and Google Scholar databases.
Keywords: immunoglobulin A, secretory IgA, infectious diseases, prophylaxis, treatment.
RESUMEN
La principal puerta de entrada de la mayoría de los microrganismos patógenos es la vía mucosal. Uno de los principales protagonistas inmunológicos en este nivel son las inmunoglobulinas, y entre ellas, prevalece la inmunoglobulina A (IgA). Su función en la protección contra varios patógenos se ha demostrado a partir de la evaluación de sus efectos profiláctico y terapéutico en modelos experimentales y en seres humanos. Sin embargo, aún existen muchas enfermedades infecciosas para las cuales no se ha estudiado la función de la IgA. Por tanto, el campo de aplicaciones de esta inmunoglobulina en enfermedades para las que no existen eficientes métodos de control, permanece abierto a futuras investigaciones. Este artículo trata acerca de las evidencias acumuladas sobre el uso de la IgA para el control de las enfermedades infecciosas, a partir de observaciones experimentales en modelos animales y seres humanos. La información se obtuvo tras la consulta de artículos científicos sobre la temática indizados en las bases de datos Pubmed y Google Académico.
Palabras clave: immunoglobulina A, IgA secretora, enfermedades infecciosas, profilaxis, tratamiento.
INTRODUCTION
Mucosal surfaces cover 400 m2 and one of their principal functions is to protect internal tissues from external influences, as inhaled and ingested antigens or pathogens, and maintaining homeostasis with the commensal microbiota [1]. The mucosal surface is the primary site of most infections and, therefore, the place of the initial immune defense. Mucosal secretory immunoglobulin A (S-IgA) is a relevant immunological barrier against pathogens that infect epithelial surfaces, with a broad cross-protection against different microorganisms [2]. Genetic sequence analysis and functional comparisons have shown that IgA is present in all mammals and birds [3].
The structure and functions of IgA, as well as their implications for the protection of mucosal surfaces have been previously reviewed [4-6]. Here we will focus on accumulative evidences of the uses of IgA in the control of infectious diseases, derived from experimental observations in animal models and humans. The main source of information was derived from papers published related with the subject, included in Pubmed and Google Scholar databases.
IgA STRUCTURE
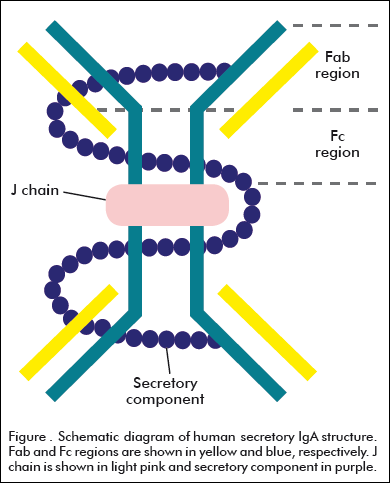
IgA exists in three monomeric variants (IgA1, IgA2m1 and IgA2m2), as dimeric form in serum (IgA1d) and S-IgA in external secretions and bile [7]. The most significant difference between the two isotypes of monomeric IgA lies in the presence of a region with 13 additional aminoacid residues present in the IgA1 isotype, containing carbohydrates attached to oxygen groups in this region [8]. The basic monomer of IgA is arranged into two identical Fab regions which bind antigen, linked through the hinge region to the Fc fragment. In dimeric IgA (dIgA), Fc regions of two monomers are linked end to end through disulfide bridges to the J chain [5] (Figure). The J chain itself is an extremely highly conserved polypeptide believed to adopt either a single β-barrel-like domain [5] or a two-domain structure, forming covalent links to the tailpiece through some Cys residues in dIgA [9].
The trans-epithelial transport of IgA onto the mucosal surfaces modifies its structure. This transport is mediated by the polymeric Ig receptor (pIgR), a receptor expressed basolaterally on glandular and mucosal epithelial cells that binds to IgA [10]. On binding, both receptor and ligand are internalized and transcytosed through a system of vesicular compartments to the apical plasma membrane. At this point, the extracellular portion of pIgR is proteolitically cleaved to form the secretory component (SC), which is covalently bound to polymeric IgA (pIgA) forming the S-IgA [11] (Figure). SC has an important role protecting S-IgA of proteolitic cleavage.
IgA IMMUNOPHYSIOLOGY
In the context of infection, S-IgA contributes to the protection of the mucosal-epithelium barrier by two primary protective functions. The first one, known as immune exclusion, is active in the epithelial stromal side where IgA can made complexes with the antigens. These immunocomplexes can be taken up by phagocytic cells, absorbed in the vascular system or transported through the epithelium into the lumen, using the pathway mediated by pIgR [12]. Thus, the antigens that cross the epithelial barrier can be clarified back into the lumen and transported forming immunocomplexes with IgA. This feature of the IgA immune elimination allows the maintenance of mucosal tissues free of immune antigen excess. The second function is based on intracellular neutralization [13]. During pIgR mediated transport, the pIgA can bind to de novo synthesized viral proteins within epithelial cells, preventing virion assembly and neutralizing viral replication [14]. Thus, it may interfere with the Ags ability (including viruses, bacteria, bacterial toxins and enzymes) to adhere and penetrate the mucosa.
In its secretory form, with the exception of vaginal fluids [15], IgA is the main immunoglobulin found in mucous secretions, including tears, saliva, colostrum and secretions from the genitourinary and gastrointestinal tracts, prostate and respiratory epithelium, and it is also found in small amounts in blood [16]. Additionally, S-IgA plays an important role in the oral homeostasis, by interacting with different microorganisms that affect the oral cavity [17]. The most abundant Ig in saliva, as in other human secretions, is dimeric S-lgA, produced by plasma cells located in the salivary glands. The proportion of subclass of IgA varies between the different mucosal sites. IgA1 represents 80-90 % of the total IgA in nasal and male genital secretions in contrast with saliva where it represents the 60 %. In colonic and female genital secretions, IgA2 is the most abundant subclass (60 %) [8]. S-IgA in the gut comes from two sources. Approximately 75 % is produced by B2 lymphocytes in organized germinal centers of mucosal lymphoid tissues such as Peyer’s patches, by a T lymphocyte-dependent mechanism [8]. The remaining 25 % of the S-IgA is produced by B1 lymphocytes that develop in the peritoneal cavity and are distributed diffusely in the intestinal lamina propria. This S-IgA may represent a primitive T lymphocyte independent source of IgA recognizing commensal bacteria [8].
Additionally, IgA is transported through the hepatobiliary pathway [7]. In certain animals (rats, mice, rabbits), pIgA is efficiently cleared by the liver and transported into bile by a receptor-mediated vesicular pathway across hepatocytes. In the rat hepatocyte, SC is synthesized as a transmembrane glycoprotein and is expressed preferentially on the sinusoidal plasma membrane. Circulating pIgA that binds to SC is internalized into endocytic vesicles and transported across the hepatocyte to the bile canalicular membrane, where pIgA is released into bile as a soluble complex with a portion of the SC, the complex being S-IgA. In some other animals (dog, guinea pig, and sheep) as well as humans, biliary epithelial cells, not hepatocytes, express SC and perform the transcytosis and secretion of pIgA into bile. The major biological functions ascribed to the secretion of IgA into bile are enhancement of immunological defense of the biliary and upper intestinal tracts and the clearance of harmful antigens from the circulation as IgA-antigen complexes [7].
SC, as an integral part of the IgA molecule, stabilizes its structure making it more resistant to the action of proteases [18]. Similarly, SC glycosylated residues enhance the anchoring of the mucosal S-IgA [19], which can potentiate the protective ability of this immunoglobulin against several pathogens that invade mucosal surfaces.
EVIDENCES ON THE ROLES OF IgA AGAINST INFECTIOUS DISEASES
Animal studies
The protective effect of IgA has been evaluated against infection with several microorganisms. For example, Ruggeri et al. developed a library of IgA monoclonal antibodies (mAbs) against different proteins of rhesus rotavirus [20]. As a result, they identified an IgA monoclonal antibody (IgA VP8 mAb) which protected newborn mice against diarrhea upon oral challenge with rotavirus, and neutralized the virus in vitro in the apical side of the filter-grown Madin-Darby canine kidney cells expressing the pIgR [20]. Nevertheless, other studies in developing countries indicated that rotavirus specific serum IgA levels are not an optimal correlate of protection following vaccination [21].
In another study, using an in vitro model, the effect of antibodies in the bovine colostrums (IgA and IgG1) was evaluated as pretreatment of necrotizing enterocolitis (NEC), an important disease in infants with low birth weight [22]. These authors reported that specific IgA and IgG1 antibodies against enterobacteria were present in bovine colostrums and also observed a marked reduction in the adhesion of different enterobacterial species to colon-derived HT-29 cells. More recently, Boullier et al. demonstrated that anti-Shigella lipopolysaccharide (LPS) S-IgA prevented Shigella-induced inflammation responsible for the destruction of the intestinal barrier, mainly due to both immune exclusion and neutralization of translocated bacteria [23].
Besides, Perryman et al. studied the roles of IgA during the infection of the mucosal pathogen Cryptosporidium parvum in mice [24]. They obtained dimeric S-IgA mAbs from hybridomas against the P23 antigen of C. parvum, which contains epitopes sensitive to neutralization, and evaluated its prophylactic and therapeutic efficacy. The authors concluded that IgA mAbs directed against the P23 antigen may be useful in passive immunization against infection by C. parvum [25], because the number of intestinal parasites was reduced when the mAbs were administered in a prophylactic scenario and the intestinal infection was reduced with the therapeutic administration of such mAbs.
Another group immunized male Mongolian gerbils (an animal model for amebiasis) by intranasal route with a vaccine based on Gal-lectin of the protozoan parasite Entamoeba histolytica and CpG-motif-containing oligodeoxynucleotides (CpG-ODN) as potent inducer of T helper type 1 immune responses [26]. Gal-lectin is a protein involved in the virulence and adhesion of E. histolytica to cause enteric amoebic colitis and abscesses of the liver in humans, and activates immune system cells. Sera from animals vaccinated with this formulation had detectable anti Gal-lectin IgG and IgA titers by immunoblotting, which were able to block parasite adhesion to target cells in vitro [26].
It has been also reported protection against enterotoxigenic Escherichia coli in weaned pigs fed with Arabidopsis thaliana seeds containing recombinant secretory IgA devoided of ligth chains containing the variable region of a llama heavy chain [27].
In another report, human polymeric IgA obtained from plasma associated with recombinant and colostrum derived SC, maintaining biochemical and functional characteristics, showing inhibitory activity upon colonization cytotoxity to human cell lines [28].
The role of IgA in protection against infection with human immunodeficiency virus (HIV) have been evaluated with recombinant IgA containing the variable region of IgG1 b12, a potent broadly neutralizing anti-gp 120 antibody which have been shown to protect macaques against vaginal simian/HIV challenge [28-31]. Using different variants of recombinant IgA containing the b12 variable region, the protection against HIV have been reported in cell lines, humanized mice and monkeys [29-32].
Several other studies had focused in the evaluation of the role of IgA in the protection against tuberculosis (TB). Mycobacterium tuberculosis (Mtb) infects by the aerogenic route and specific S-IgA could have a prominent role in the protection against the infection. Taking into consideration these antecedents the study of the protective role of S-IgA in protection against TB has a paramount importance by its potential application in the development of improved vaccines and new immunotherapeutic tools.
Currently, TB treatment is far from ideal because it requires the combination of multiple drugs, which needs to be administered by prolonged periods of time [33]. This means that a high percentage of patients often abandon the treatment which contributes to the appearance of multidrug-resistant strains. Vaccination with Mycobacterium bovis BCG is currently the only alternative to prevent tuberculosis. Nevertheless, this vaccine has the significant disadvantage that it only protects against severe forms of the disease in childhood. It has been shown that in endemic countries this vaccine does not protect against the adult pulmonary form, which is the most common manifestation of disease and responsible for its transmission [33].
Among the first studies in this line of research, a mouse monoclonal IgA against 16 kDa protein of Mtb was evaluated ex vivo demonstrating its binding to the galectin-3/Mac-2 lectin from mouse macrophage cell lines [34]. Gal-3 is accumulating only in those phagosomes that contained live Mtb, through the binding to phosphatidylinositol mannosides (PIM) and appeared to influence the clearance of late infection [34]. IgA antibodies recognizing mycobacterial surface components, as the one used in this study, could thus give an additional targeting opportunity to influence the course of the intracellular infection. Based on their preliminary data showing IgA binding more prominently to intracellular rather than the surface Gal-3 of the J774 macrophages, these authors suggested that it is possible that IgA-coated particles may be endocytosed via some other IgA receptor on the cell surface. Subsequently, once inside the cell, the IgA immune complexes may be targeted for Gal-3 mediated sequestration to phagosomes. Williams et al. reported that the same IgA mAb directed against the α-crystallin protein of Mtb when administered intranasally is protective against early TB infection in mice, by an isotype and epitope specific mechanism [35]. However, this effect was not significant after 9 days post-infection. Based on this, the same group further investigated the possibility of extending this protective effect by inoculating IFN-γ 3 days before and 2 and 7 days after infection with Mtb by aerosol [36]. Indeed, this new treatment extended the passive protection conferred by IgA, which was evidenced as reduced infection and granulomatous lung infiltration by 4 weeks, compared to independent administration of IgA or IFN-γ [36].
Our group studied the protective activity of two mAbs directed against Acr and PstS-1 proteins of Mtb, TBA 61 and TBA84 respectively, using an intratracheal model of pulmonary infection with Mtb H37Rv [37]. The study revealed a significant reduction in bacterial load and morphometric and histopathological changes in lungs of mice treated 21 days post-infection with TBA61, compared to those receiving TBA84 and control groups.
Balu et al. evaluated the properties of a new monoclonal IgA1 clone constructed using a single chain variable fragment (2E9IgA1), selected from an antibody phage library [6]. The intranasal co-inoculation of 2E9IgA1 with recombinant murine IFN-γ significantly inhibited lung infection in transgenic mice for human CD89 but not in the control group. This supported the assumption that CD89 binding is required for passive protection conferred by IgA. Previously, the same group had postulated that the IgA mAb administered by intranasal route induces pro-inflammatory cell responses, which may have enhanced protection by promoting apoptosis of macrophages infected with mycobacteria [38]. The potential role of CD89 receptor in the protective mechanism mediated by IgA is also supported by reports using therapeutic human monoclonal IgA antibodies against tumor antigens in C89 transgenic mice [39]. In that study, IgA monoclonal antibodies demonstrated a superior tumoricidal activity, compared to monoclonal antibodies of the IgG isotype with the same combining site.
Recently, we evaluated the effect of S-IgA obtained from human colostrum, administered by intranasal route to Balb/c mice, against infection with Mtb H37Rv, [4]. The results showed a reduction in pneumonic areas in mice that received S-IgA before challenge with mycobacteria. However, this protective effect was more evident when IgA was administered together with the mycobacteria, after pre-incubation with the microorganism, which was evidenced by a reduction in bacterial load and tissue damage in lungs, as well as increased production of iNOS, compared to the group receiving the IgA alone and with the control group [4]. These results were the first evidence of prophylactic effect of S-IgA derived from human colostrum against infection with Mtb. However, future research is required to determine whether S-IgA from human colostrum also has therapeutic effect related to infection with Mtb.
The role of IgA was also evaluated in the protection against intranasal infection with M. bovis BCG, using IgA deficient and wild type non-targeted littermate mice [40]. The animals were immunized with the mycobacterium surface antigen PstS-1 formulated with cholera toxin. The results demonstrated that IgA deficient mice were more susceptible to BCG infection compared to wild type mice, revealed by the higher bacterial load in lungs and broncho-alveolar lavage. Besides, IgA deficient mice showed a reduction in the IFN-γ and TNF-α level in lungs after the analysis of the cytokine response.
The results obtained related with the protective effect against mycobacteria in mice of murine and human S-IgA suggest that clinical applications of specific S-IgA in the control of human tuberculosis could be feasible. One possibility is to use S-IgA for infection prophylaxis on high risk groups, such as HIV infected individuals at risk of Mtb infection. Another important application could be as a therapeutic element combined with the conventional therapy, to shorten the treatment period, possibly decreasing the dose of the drugs and the related treatment side effects, thus favoring the compliance and lowering the risk of generation of drug resistant strains. The uses of S-IgA as adjunt in the treatment of Multidrug Resistant (MDR) strains as well as in the treatment of latent tuberculosis are exciting possibilities that deserves further insight. Based on the results of protection with S-IgA in mice, our group is currently exploring the possibility to develop experimental vaccine candidates able to elicit potent IgA responses to Mtb epitopes and antigens in the respiratory tract.
There are also studies evaluating IgA production and its role in protection in transgenic animals. Transgenic mice lines (BCBA, C57BL/6 CBA/J) were generated containing the complete murine pIgR gene under lactogenic control of a milk gene promoter, rather than under immunological control [41]. Mice over-expressing the pIgR protein in mammary gland epithelial cells, 60- and 270-fold above normal pIgR protein levels, showed 1.5- and 2-fold higher total IgA levels in milk, respectively, compared to the IgA levels in the milk of non-transgenic mice. The authors referred that this result indicates that the amount of pIgR produced was indeed a limiting factor in the transport of dIgA into the milk under normal non-inflammatory circumstances. Later, they explored the mechanism by which IgA can mediate a protective effect against malaria, using recombinant human IgA specific for the C-terminal 19 kDa region of Plasmodium falciparum merozoite surface protein 1 and transgenic Balb/c mice for the human Fcα receptor (FcαR1/CD89) [42]. In this study, the human IgA failed to protect against parasite challenge in vivo in transgenic mice, suggesting that this antibody class does not play a major role in control of infection. However, these authors did not exclude the possibility that protective capacity is compromised in this model due to a rapid clearance and inappropriate bio-distribution of IgA and differences in FcαR1 expression profile, between humans and transgenic mice [42]. Some of the results discussed in this section are summarized in the table.
Observations in humans
Some individuals with specific IgA deficiency show susceptibility to different infections. The most common are gastrointestinal infections such as Giardia lamblia, Campylobacter, Clostridium, Salmonella and rotavirus [8]. Commonly IgA deficiency is associated with an increased incidence of autoimmune diseases, in general related to gastrointestinal tract, as in the case of celiac disease [43]. Furthermore, healthy contacts of leprosy patients having high levels of S-IgA in saliva develop clinical symptoms of the disease at a low frequency and a high percentage of them develop protective immunity [44].
Taking into account these elements, and the accumulated experimental evidence, the protective and therapeutic potential of IgA have been evaluated in humans. One example of the clinical evaluation of formulations based on IgA against infections that affect humans is the case of S-IgA obtained from plants. Production of antibodies in plants through genetic engineering has as main advantages the low cost of production and the absence of pathogenic viruses or bacteria to humans, causing unneeded pathogen removal steps during purification [45]. The development of S-IgA Guy’s 13 plantibody technology began with the study of Ma et al. [46]. These authors sexually crossed four transgenic tobacco plants, expressing heavy and light chains of IgA, the J chain and SC respectively. The product CaroRx, an IgA/G chimeric secretory antibody produced in plants already completed its phase II clinical trial. Preliminary clinical studies with this antibody indicate that plant-derived IgA prevents oral colonization by Streptococcus mutans through passive immunization of mucosal surfaces by topical application, specifically by binding to the major adhesin SA I/II of the bacteria. They reported that by this mechanism, the antibody leads to replacement of this pathogen by endogenous harmless flora.
Additionally, there are evidences that specific S-IgA contribute to the efficacy of a live attenuated influenza vaccine, as concluded from randomized, placebo-controlled clinical trials in young children [47].
Several studies support the use of antibodies from colostrums for the treatment of some infections. That is the case of effective treatment of rotavirus-induced diarrhea in children using antibodies derived from colostrums of immunized cows [48]. However, this treatment only based on colostrums is not practical because the source is limited and for this reason some years ago was assessed another alternative to address the antibody-based therapy. This new method uses probiotic bacteria taking advantage of its ability to survive into the intestinal tract, its production of antimicrobial compounds and stimulation of the mucosal immune response leading to increased levels of S-IgA [49].
Other applications of bovine colostrum antibodies in different infections were comprehensively reviewed by Weiner et al. [50]. Moreover, a study conducted in healthy volunteers showed that the use of a mouthwash containing milk-derived antibodies against S. mutans reduced the amount of bacteria, which form small colonies [51].
Interestingly, studies designed to assess the role of IgA in protection against Helicobacter pylori infection have shown conflicting results. Some studies evidence the influence of IgA limiting H. pylori colonization in children, showing protection against this microorganism in infants consuming breast milk [52]. Otherwise, another group provided contradictory results of protection against that pathogen in mice deficient of mature B cells [46]. In this case, therapeutic immunization stimulates an immune response, which reduces H. pylori by an antibody independent mechanism [53].
The influence of IgA on protection against Vibrio cholerae infection has also been evaluated in several studies. One of the most recent demonstrated that levels of serum IgA specific to three V. cholerae antigens –the B subunit of cholera toxin, LPS, and TcpA, the major component of the toxin co-regulated pilus– predicted protection in household contacts of patients infected with V. cholerae O1. Circulating IgA antibodies to TcpA were also associated with protection from V. cholerae O139 infection [54].
On the other hand, Crooks et al. evaluated the effect of bovine colostrum supplementation on salivary IgA in distance runners [55], tacking as hypothesis that nutritional supplements may improve mucosal immunity and could be beneficial to athletes who are at increased risk of upper respiratory tract infection. They consumed a supplement of either bovine colostrum or placebo as negative control for 12 weeks. The results demonstrated increased IgA levels in saliva among a cohort of athletes following colostrum supplementation. Also in 2006, the efficacy and tolerability of colostrums in preventing recurrent episodes of infections of the upper respiratory tract and diarrhea in children was assessed [56]. As result, bovine colostrums were highly effective, not only in the prophylactic treatment of recurrent upper respiratory tract infections and diarrhea, but also to reduce hospitalization episodes caused by these conditions. It was previously reported that IgA is the major mediator of resistance against viral infections of the respiratory mucosa [57] and the concentration of IgA in saliva is increased after 2 weeks of consumption of bovine colostrum [58].
Human IgA has been also used to develop a product, IgAbulin, which was evaluated in previous studies as nasal treatment, with the aim of preventing infection of the upper respiratory tract in athletes. In one study, athletes did not develop apparent infection after 17 days of treatment twice daily with IgAbulin [59]. Instead, other results showed no significant reduction in respiratory tract infection after treatment with nose drops of the product [60]. Besides, two studies evaluating the effect of this product as intranasal passive immunization in children against respiratory tract infections [61, 62] showed that the prophylactic nasal effect with antibody is practical and effective.
In another development, there was obtained a murine monoclonal IgA (HNK20) against the F glycoprotein of the respiratory syncytial virus (RSV), specifically developed for passive intranasal immunization [63]. When administered intranasally and daily to healthy adults and children, and to children at high risk of infection, there were no reports of adverse effects on small-scale treatments [63]. The prophylactic effect was evaluated in adult volunteers challenged intranasally with the wild strain of RSV subgroup A, 1 hour after receiving the first dose of mAb. During the acute phase of infection (5-8 days after challenge), mean daily virus shedding was lower in subjects treated with HNK20 than those receiving placebo.
According to its physiological role, the more obvious potential use of IgA is at mucosal level as a prophylactic and potentially therapeutic tool against infectious agents that penetrates or establishes infections in mucosal tissues. It remains to be demonstrated in future studies the superiority of IgA administration over the use of IgG by mucosal route. In our experience, similar results have been obtained with the use of human IgG and IgA formulations in models of mycobacterial infection, particularly in the prophylaxis of progressive tuberculosis in mice [64, 65, 4].
Up to now, the main limitation for the generalized evaluation and use of IgA is the lack of adequate natural sources for large scale production. This is being solved with the use of new production methods for recombinant and plasma derived IgA of intact functional capacity, which offers a promisory outlook of future clinical application [27-32, 39]. Another aspect to be taken into account is the possibility of adverse reactions to the administration of IgA in individuals with congenital deficits of this immunoglobulin Another aspect to be taken into account is the possibility of adverse reactions to the administration of IgA in individuals with congenital deficits of this immunoglobulin
Despite the epidemiological and experimental evidences of the role of IgA in protection against infections and the successful attempts to use it in the prophylaxis and treatment of infectious diseases, its potential as treatment against the wide range of mucosal infections should be further assessed.
CONCLUSIONS
To date, several groups have demonstrated the involvement of IgA in the prophylaxis and treatment of various infectious diseases in animals and humans. Hence, there is an increasing interest to unravel the function of this immunoglobulin for the control of a wide number of disorders, some of them neglected, and also in autoimmune diseases. In this sense, the potential of IgA should be explored in the future, particularly in poor infection control scenarios. The availability of methods for production of humanized therapeutic antibodies which are available in the field of cancer makes us envisage the future use of S-IgA, as a real alternative for the treatment and prevention of infectious diseases.
ACKNOWLEDGEMENTS
This work was jointly supported by the Ministry of Higher Education, Malaysia LRGS Grant (203.PSK.6722001).
REFERENCES
1. Pamer EG. Immune responses to commensal and environmental microbes. Nat Immunol. 2007;8(11):1173-8.
2. Corthesy B. Multi-faceted functions of secretory IgA at mucosal surfaces. Front Immunol. 2013;4:18.
3. Kawamura S, Saitou N, Ueda S. Concerted evolution of the primate immunoglobulin alpha-gene through gene conversion. J Biol Chem. 1992;267(11):7359-67.
4. Alvarez N, Otero O, Camacho F, Borrero R, Tirado Y, Puig A, et al. Passive administration of purified secretory IgA from human colostrum induces protection against Mycobacterium tuberculosis in a murine model of progressive pulmonary infection. BMC Immunol. 2013;14 Suppl 1:S3.
5. Woof JM, Russell MW. Structure and function relationships in IgA. Mucosal Immunol. 2011;4(6):590-7.
6. Balu S, Reljic R, Lewis MJ, Pleass RJ, McIntosh R, van Kooten C, et al. A novel human IgA monoclonal antibody protects against tuberculosis. J Immunol. 2011;186(5):3113-9.
7. Brown WR, Kloppel TM. The liver and IgA: immunological, cell biological and clinical implications. Hepatology. 1989;9(5):763-84.
8. Macpherson AJ, Gatto D, Sainsbury E, Harriman GR, Hengartner H, Zinkernagel RM. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science. 2000;288(5474):2222-6.
9. Johansen FE, Braathen R, Brandtzaeg P. The J chain is essential for polymeric Ig receptor-mediated epithelial transport of IgA. J Immunol. 2001;167(9):5185-92.
10. Wieland WH, Orzaez D, Lammers A, Parmentier HK, Verstegen MW, Schots A. A functional polymeric immunoglobulin receptor in chicken (Gallus gallus) indicates ancient role of secretory IgA in mucosal immunity. Biochem J. 2004;380(Pt 3):669-76.
11. Kaetzel CS, Mostov K. Immunoglobulin transport and the polymeric immunoglobulin receptor. In: Mestecky J, Lamm ME, Strober W, Bienenstock J, McGhee JR, Mayer L, editors. Mucosal Immunology. 3rd ed. Amsterdam: Elsevier/Academic Press; 2005. p. 211-50.
12. Robinson JK, Blanchard TG, Levine AD, Emancipator SN, Lamm ME. A mucosal IgA-mediated excretory immune system in vivo. J Immunol. 2001;166(6):3688-92.
13. Strugnell RA, Wijburg OL. The role of secretory antibodies in infection immunity. Nat Rev Microbiol. 2010;8(9):656-67.
14. Yan H, Lamm ME, Bjorling E, Huang YT. Multiple functions of immunoglobulin A in mucosal defense against viruses: an in vitro measles virus model. J Virol. 2002;76(21):10972-9.
15. Wright PF. Inductive/effector mechanisms for humoral immunity at mucosal sites. Am J Reprod Immunol. 2011;65(3):248-52.
16. Junqueira LC, Carneiro J. Basic Histology. 10th ed. New York: McGraw-Hill; 2003.
17. Rashkova M, Baleva M, Toneva N, Jegova G. Secretory immunoglobulin A (S-IgA) and periodontal status in children with diseases and conditions influencing the oral environment. J IMAB Annu Proc. 2009:36-40.
18. Crottet P, Corthesy B. Secretory component delays the conversion of secretory IgA into antigen-binding competent F(ab’)2: a possible implication for mucosal defense. J Immunol. 1998;161(10):5445-53.
19. Phalipon A, Cardona A, Kraehenbuhl JP, Edelman L, Sansonetti PJ, Corthesy B. Secretory component: a new role in secretory IgA-mediated immune exclusion in vivo. Immunity. 2002;17(1):107-15.
20. Ruggeri FM, Johansen K, Basile G, Kraehenbuhl JP, Svensson L. Antirotavirus immunoglobulin A neutralizes virus in vitro after transcytosis through epithelial cells and protects infant mice from diarrhea. J Virol. 1998;72(4):2708-14.
21. Angel J, Franco MA, Greenberg HB. Rotavirus immune responses and correlates of protection. Curr Opin Virol. 2012;2(4):419-25.
22. Brooks HJ, McConnell MA, Corbett J, Buchan GS, Fitzpatrick CE, Broadbent RS. Potential prophylactic value of bovine colostrum in necrotizing enterocolitis in neonates: an in vitro study on bacterial attachment, antibody levels and cytokine production. FEMS Immunol Med Microbiol. 2006;48(3):347-54.
23. Boullier S, Tanguy M, Kadaoui KA, Caubet C, Sansonetti P, Corthesy B, et al. Secretory IgA-mediated neutralization of Shigella flexneri prevents intestinal tissue destruction by down-regulating inflammatory circuits. J Immunol. 2009;183(9):5879-85.
24. Perryman LE, Jasmer DP, Riggs MW, Bohnet SG, McGuire TC, Arrowood MJ. A cloned gene of Cryptosporidium parvum encodes neutralization-sensitive epitopes. Mol Biochem Parasitol. 1996;80(2):137-47.
25. Enriquez FJ, Riggs MW. Role of immunoglobulin A monoclonal antibodies against P23 in controlling murine Cryptosporidium parvum infection. Infect Immun. 1998;66(9):4469-73.
26. Ivory CP, Chadee K. Intranasal immunization with Gal-inhibitable lectin plus an adjuvant of CpG oligodeoxynucleotides protects against Entamoeba histolytica challenge. Infect Immun. 2007;75(10):4917-22.
27. Virdi V, Coddens A, De Buck S, Millet S, Goddeeris BM, Cox E, et al. Orally fed seeds producing designer IgAs protect weaned piglets against enterotoxigenic Escherichia coli infection. Proc Natl Acad Sci USA. 2013;110(29):11809-14.
28. Longet S, Miled S, Lotscher M, Miescher SM, Zuercher AW, Corthesy B. Human plasma-derived polymeric IgA and IgM antibodies associate with secretory component to yield biologically active secretory-like antibodies. J Biol Chem. 2013;288(6):4085-94.
29. Mantis NJ, Palaia J, Hessell AJ, Mehta S, Zhu Z, Corthesy B, et al. Inhibition of HIV-1 infectivity and epithelial cell transfer by human monoclonal IgG and IgA antibodies carrying the b12 V region. J Immunol. 2007;179(5):3144-52.
30. Moldt B, Saye-Francisco K, Schultz N, Burton DR, Hessell AJ. Simplifying the synthesis of SIgA: Combination of dIgA and rhSC using affinity chromatography. Methods. 2014;65(1):127-32.
31. Watkins JD, Sholukh AM, Mukhtar MM, Siddappa NB, Lakhashe SK, Kim M, et al. Anti-HIV IgA isotypes: differential virion capture and inhibition of transcytosis are linked to prevention of mucosal R5 SHIV transmission. AIDS. 2013;27(9):F13-20.
32. Hur EM, Patel SN, Shimizu S, Rao DS, Gnanapragasam PN, An DS, et al. Inhibitory effect of HIV-specific neutralizing IgA on mucosal transmission of HIV in humanized mice. Blood. 2012;120(23):4571-82.
33. World Health Organization. Global Tuberculosis Report 2012. 2012 [cited 2013 Aug 18]. Available from: http://apps.who.int/iris/bitstream/10665/75938/1/9789241564502_eng.pdf
34. Reljic R, Crawford C, Challacombe S, Ivanyi J. Mouse monoclonal IgA binds to the galectin-3/Mac-2 lectin from mouse macrophage cell lines. Immunol Lett. 2004;93(1):51-6.
35. Williams A, Reljic R, Naylor I, Clark SO, Falero-Diaz G, Singh M, et al. Passive protection with immunoglobulin A antibodies against tuberculous early infection of the lungs. Immunology. 2004;111(3):328-33.
36. Reljic R, Clark SO, Williams A, Falero-Diaz G, Singh M, Challacombe S, et al. Intranasal IFNgamma extends passive IgA antibody protection of mice against Mycobacterium tuberculosis lung infection. Clin Exp Immunol. 2006;143(3):467-73.
37. Lopez Y, Yero D, Falero-Diaz G, Olivares N, Sarmiento ME, Sifontes S, et al. Induction of a protective response with an IgA monoclonal antibody against Mycobacterium tuberculosis 16kDa protein in a model of progressive pulmonary infection. Int J Med Microbiol. 2009;299(6):447-52.
38. Reljic R, Williams A, Ivanyi J. Mucosal immunotherapy of tuberculosis: is there a value in passive IgA? Tuberculosis (Edinb). 2006;86(3-4):179-90.
39. Boross P, Lohse S, Nederend M, Jansen JH, van Tetering G, Dechant M, et al. IgA EGFR antibodies mediate tumour killing in vivo. EMBO Mol Med. 2013;5(8):1213-26.
40. Rodriguez A, Tjarnlund A, Ivanji J, Singh M, Garcia I, Williams A, et al. Role of IgA in the defense against respiratory infections IgA deficient mice exhibited increased susceptibility to intranasal infection with Mycobacterium bovis BCG. Vaccine. 2005;23(20):2565-72.
41. De Groot N, Van Kuik-Romeijn P, Lee SH, De Boer HA. Increased immunoglobulin A levels in milk by over-expressing the murine polymeric immunoglobulin receptor gene in the mammary gland epithelial cells of transgenic mice. Immunology. 2000;101(2):218-24.
42. Shi J, McIntosh RS, Adame-Gallegos J, Dehal PK, van Egmond M, van de Winkel J, et al. The generation and evaluation of recombinant human IgA specific for Plasmodium falciparum merozoite surface protein 1-19 (PfMSP1 19). BMC Biotechnol. 2011;11:77.
43. Robins G, Howdle PD. Advances in celiac disease. Curr Opin Gastroenterol. 2005;21(2):152-61.
44. Ramaprasad P, Fernando A, Madhale S, Rao JR, Edward VK, Samson PD, et al. Transmission and protection in leprosy: indications of the role of mucosal immunity. Leprosy Rev. 1997;68(4):301-15.
45. Wycoff KL. Secretory IgA antibodies from plants. Curr Pharm Des. 2005;11(19):2429-37.
46. Ma JK, Hikmat BY, Wycoff K, Vine ND, Chargelegue D, Yu L, et al. Characterization of a recombinant plant monoclonal secretory antibody and preventive immunotherapy in humans. Nat Med. 1998;4(5):601-6.
47. Ambrose CS, Wu X, Jones T, Mallory RM. The role of nasal IgA in children vaccinated with live attenuated influenza vaccine. Vaccine. 2012;30(48):6794-801.
48. Harris JB, LaRocque RC, Chowdhury F, Khan AI, Logvinenko T, Faruque AS, et al. Susceptibility to Vibrio cholerae infection in a cohort of household contacts of patients with cholera in Bangladesh. PLoS Negl Trop Dis. 2008;2(4):e221
49. Sarker SA, Casswall TH, Mahalanabis D, Alam NH, Albert MJ, Brussow H, et al. Successful treatment of rotavirus diarrhea in children with immunoglobulin from immunized bovine colostrum. Pediatr Infect Dis J. 1998;17(12):1149-54.
50. Weiner C, Pan Q, Hurtig M, Boren T, Bostwick E, Hammarstrom L. Passive immunity against human pathogens using bovine antibodies. Clin Exp Immunol. 1999;116(2):193-205.
51. Zhang Z, Xiang Y, Li N, Wang B, Ai H, Wang X, et al. Protective effects of Lactobacillus rhamnosus GG against human rotavirus-induced diarrhoea in a neonatal mouse model. Pathog Dis. 2013;67(3):184-91.
52. Filler SJ, Gregory RL, Michalek SM, Katz J, McGhee JR. Effect of immune bovine milk on Streptococcus mutans in human dental plaque. Arch Oral Biol. 1991;36(1):41-7.
53. Thomas JE, Austin S, Dale A, McClean P, Harding M, Coward WA, et al. Protection by human milk IgA against Helicobacter pylori infection in infancy. Lancet. 1993;342(8863):121.
54. Ermak TH, Giannasca PJ, Nichols R, Myers GA, Nedrud J, Weltzin R, et al. Immunization of mice with urease vaccine affords protection against Helicobacter pylori infection in the absence of antibodies and is mediated by MHC class II-restricted responses. J Exp Med. 1998;188(12):2277-88.
55. Crooks CV, Wall CR, Cross ML, Rutherfurd-Markwick KJ. The effect of bovine colostrum supplementation on salivary IgA in distance runners. Int J Sport Nutr Exerc Metab. 2006;16(1):47-64.
56. Patel K, Rana R. Pedimune in recurrent respiratory infection and diarrhoea -the Indian experience- the pride study. Indian J Pediatr. 2006;73(7):585-91.
57. Murphy BR. Mucosal immunity to viruses. In: Ogra PL, Mestecky J, Lamm ME, Strober W, McGhee JR, Bienenstock J, editors. Handbook of Mucosal Immunology. New York: Academic Press. 1994; p. 333-43.
58. Mero A, Kahkonen J, Nykanen T, Parviainen T, Jokinen I, Takala T, et al. IGF-I, IgA, and IgG responses to bovine colostrum supplementation during training. J Appl Physiol (1985). 2002;93(2):732-9.
59. Hemmingsson P, Hammarstrom L. Nasal administration of immunoglobulin as effective prophylaxis against infections in elite cross-country skiers. Scand J Infect Dis. 1993;25(6):783-5.
60. Lindberg K, Berglund B. Effect of treatment with nasal IgA on the incidence of infectious disease in world-class canoeists. Int J Sports Med. 1996;17(3):235-8.
61. Giraudi V, Riganti C, Torales MR, Sedola H, Gaddi E. Upper respiratory infections in children: response to endorsal administration of IgA. Int J Pediatr Otorhinolaryngol. 1997;39:103-10.
62. Heikkinen T, Ruohola A, Ruuskanen O, Waris M, Uhari M, Hammarstrom L. Intranasally administered immunoglobulin for the prevention of rhinitis in children. Pediatr Infect Dis J. 1998;17(5):367-72.
63. Weltzin R, Hsu SA, Mittler ES, Georgakopoulos K, Monath TP. Intranasal monoclonal immunoglobulin A against respiratory syncytial virus protects against upper and lower respiratory tract infections in mice. Antimicrob Agents Chemother. 1994;38(12):2785-91.
64. Olivares N, Leon A, Lopez Y, Puig A, Cadiz A, Falero G, et al. The effect of the administration of human gamma globulins in a model of BCG infection in mice. Tuberculosis (Edinb). 2006;86(3-4):268-72.
65. Olivares N, Puig A, Aguilar D, Moya A, Cadiz A, Otero O, et al. Prophylactic effect of administration of human gamma globulins in a mouse model of tuberculosis. Tuberculosis (Edinb). 2009;89(3):218-20.
66. Rachid R, Bonilla FA. The role of anti-IgA antibodies in causing adverse reactions to gamma globulin infusion in immunodeficient patients: a comprehensive review of the literature. J Allergy Clin Immunol. 2012;129(3):628-34.
Received in September, 2013.
Accepted in January, 2014.
Nadine Alvarez. Vicepresidencia de Investigaciones y Desarrollo, Instituto Finlay. Ave. 27 No. 19805, La Lisa, PO Box 16017, CP 11600, La Habana, Cuba. E-mail: nalvarez@finlay.edu.cu.