Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Biotecnología Aplicada
versión On-line ISSN 1027-2852
Biotecnol Apl vol.32 no.1 La Habana ene.-mar. 2015
RESEARCH
Evaluation in vitro of the cytotoxicity and genotoxicity of silica nanoparticle aggregates synthesized by the sol-gel method
Evaluación in vitro de la citoxicidad y genotoxicidad de agregados de nanopartículas de sílice sintetizadas por el método sol-gel
Diana M Gómez-Gallego1, Claudia E Espinal-Correa2, Ana C Ossa-Giraldo1
1Grupo de investigación Infettare. Universidad Cooperativa de Colombia, Sede Medellín. Calle 48 # 28-00, Medellín, Antioquia, Colombia.
2Grupo de investigación Olistica. Universidad Cooperativa de Colombia, Sede Medellín, Colombia.
ABSTRACT
Amorphous silica nanoparticles (NPs) has become attractive for biomedical applications. In the present study, the toxicity of different concentrations of amorphous silica NPs of 27 nm was evaluated, such NPs synthesized by the sol-gel method assisted by reverse micelle microemulsion. Characterization of nanoparticles was done using Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM) and Dynamic Light Scattering (DLS). Cytotoxicity was assessed in CHO-K1 and Cal27 cell cultures by the reduction bromide 3-(4,5-dimethylthiazol-2-yl)-2,5-difeniltetrazol (MTT) test. Cellular viability and density tests were assessed in CHO-K1 cells with Trypan Blue, while genotoxicity was evaluated in the CHO-K1 cell line and in primary cultures of human lymphocytes using the Comet assay. Results showed that silica NPs produced cytotoxicity in both cell models in a dose dependent manner (12.5 µg/mL or higher), while genotoxicity was observed in CHO-K1 cells at all the concentrations tested (1.5625-12.5 µg/mL), but not in the primary cultures of human lymphocytes. The variability of the toxic effects of amorphous silica NPs, as dependent on NPs concentration and the cell line tested, among others, indicates that their applications could be determined by their use at non-toxic doses or through functionalization by chemical modifications to reduce their toxicity. These parameters, together with their unique chemical and physical properties could make NPs a feasible option for biomedical applications.
Keywords: silica nanoparticles, cytotoxicity, genotoxicity, nanotechnology, biomedicine.
RESUMEN
Las nanopartículas (NP) amorfas de sílice son atractivas para aplicaciones futuras en la biomedicina. En este estudio, se evaluó la toxicidad de diferentes concentraciones de agregados de NP amorfas de sílice de 27 nm, sintetizadas mediante el método sol-gel asistido por microemulsión de micelas inversas. Estas NP se caracterizaron mediante Microscopía Electrónica de Barrido, Microscopía Electrónica de Transmisión y Dispersión Dinámica de Luz. La citotoxicidad se evaluó en células CHO-K1 y Cal27 en cultivo mediante el test de reducción de Bromuro de 3-(4,5-dimetiltiazol-2-ilo)-2,5-difeniltetrazol (MTT) y en CHO-K1 se hicieron pruebas Viabilidad y Densidad Celular con Azul de Tripano. La genotoxicidad se evaluó en la línea celular CHO-K1 y en cultivos primarios de linfocitos humanos usando el Ensayo Cometa. Las NP indujeron citoxicidad dependiente de la dosis en ambos modelos celulares, a concentraciones iguales o superiores a 12.5 µg/mL, mientras que se observó genotoxicidad en las células CHO-K1 a todas las concentraciones evaluadas (1.5625-12.5 µg/mL), pero no en los cultivos primarios de linfocitos humanos. La variabilidad de los efectos citotóxicos de las NP amorfas de sílice en función de la concentración y línea celular, entre otros, indican que su posible uso puede estar condicionado por el empleo a dosis no tóxicas, o a su disminución mediante la funcionalización de las NP mediante modificaciones químicas que disminuyan su toxicidad. Tales estrategias, unidas a sus características químicas y físicas exclusivas, las convierten en una opción a considerar para diversas aplicaciones en la biomedicina.
Palabras clave: nanopartículas de silica, citotoxicidad, genotoxicidad, nanotecnología, biomedicina.
INTRODUCTION
In recent decades, the increase and progress of nanotechnology research [1] has produced a wide span of medical applications ranging from diagnostics, monitoring and biological control to its therapeutic use in the treatment of different pathologies, for example, in cancer [2, 3].
Notably, the invention of nanoparticles (NPs) has been focused on their use as vehicles for the delivery and controlled release of drugs to their target cells and organs [1, 4], in order to decrease the potential toxicity or side effects caused by the interaction of the drugs with other cells or tissues toward which the treatment is not aimed. Significantly, silica NPs have emerged as a potential option [5], due to their exclusive properties, such as their chemical and thermal stability, their large surface area, the high stability in aqueous suspensions and their innocuousness to the environment of their inert material composition [6]. They have been also demonstrated as being hardly appealing for microorganisms and acting as a barrier that limits the effect of the external environment on the nucleus of the particle [6, 7]. Therefore, research is being carried out at a worldwide level in relation to the synthesis of silica NPs that may be used for encapsulating and controlling the drug delivery.
However, it is necessary to evaluate the toxicity that may be produced by NPs in a biological system to ensure their good use in the biomedical field. Therefore, it is important to make a toxicological characterization through in vitro and in vivo assays that offer information on the interaction of the nano-material with cells or tissues, since the physical characteristics of the NP (form, size, adsorption capacity, hydrophobicity, surface charge, optics and catalytic activity), as well as the exposure time and concentration of NPs, can have an essential role in evoking deleterious effects on cell functions [8-10], including cell growth, apoptosis, adhesion, migration and damage to the genetic material, among others [11].
The aim of this study was to assess the cytotoxicity and genotoxicity potential of amorphous silica NPs synthesized by the sol-gel method assisted by micro-emulsion of inverse micelles, thus contributing to the research in nano-medicine in a country where the study in this field is just starting.
MATERIALS AND METHODS
Synthesis of silica nanoparticles
NPs were synthesized in the Ceramics and Vitreous Laboratory of the National University of Colombia at Medellin, through the sol-gel method assisted by the micro-emulsion of inverse micelles, using tetra-ethyl-ortho-sylicate (TEOS) (Sigma Aldrich) as a precursor of silica, Triton X-100 (Sigma Aldrich) as surfactant, 98 % pure methanol (Carlo Erba) as the co-surfactant and 99.5 % pure cyclo-hexane (Sigma Aldrich) at as the oily phase. In order to ensure that the particles obtained have nanometric sizes, molar ratios h, p and R of 59.1, 4.5 and 9.2, respectively, were used. These parameters were calculated by equations: h = [H2O]/[TEOS], p = [Metanol]/[Triton X-100], and R = [H2O]/[Triton X-100].
The surfactant, co-surfactant and oil phase were initially mixed at 700 rpm and 25 °C, to form the micro-emulsion. The order and amount of reagents used determined the inversion of the micelles. After forming the micro-emulsion of inverse micelles, the medium was adjusted to pH 8.0, with a solution of aqueous ammonium hydroxide, to catalyze the polymerization reactions needed for the growth of silica particles that are formed in the following step. Finally, the silica precursor, TEOS, was added to enable the hydrolysis and condensation of the silica molecules within the micelles. The mixture was then shaken for two hours, and then the micro-emulsion was disrupted by adding 5 mL of ethanol. They were centrifuged at 1500 rpm, and washed with ethanol and water to eliminate the excess surfactant and co-surfactant, to recover the NPs obtained.
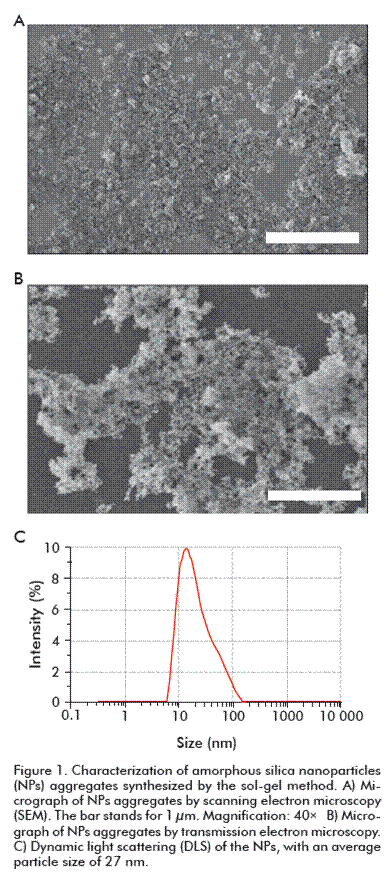
NPs were characterized by the size of the particles and their morphology through high resolution Scanning Electron Microscopy (SEM) Hitachi S4800 SEM-FEG, Transmission Electronic Microscopy (TEM) with a Philips CM200 microscope and Dynamic Light Scattering (DLS) through a Zetasizer Nano S Malvern Instruments device.
Cytotoxicity tests
The experiments were carried out using a culture at the exponential growth stage of the CHO-K1 cell line that was propagated in a routine manner using the RPMI 1640 (Gibco) medium supplemented with 10 % fetal bovine serum (Gibco) and 1 % Glutamine (Gibco). The cells were incubated at 37 ºC, under a 5 % CO2 atmosphere and a relative humidity of 95 %. Subcultures were made by growing 2 × 105 cells in T25 culture flasks, under the same conditions described above. All experiments were carried out with viability of or above 95 % and with the cultures at the exponential growth phase.
Viability and cellular density by Trypan blue
For counting, a cell concentration of a culture treated with the different concentrations of the NP aggregates (100, 50, 25, 12.5, 6.25 and 3.125 µg/mL) was submitted to a 1:1 proportion of 0.4 % Trypan blue (Gibco). From the mixture, 10 μL were taken to the Neubauer plate (hemocytometer) and the viable cells (transparent cells) were counted, as well as the dead cells (blue cells) under an optic microscope in the four quadrants. A negative control (untreated cells) and a positive control (cells treated with 10 µL of Dimethyl Sulfoxide (DMSO; Amresco)) were made. The experiment was carried out by triplicate for each treatment and the average results of the two separate experiments are shown. The percentage of viability and density were measured with equations 1 and 2, respectively:
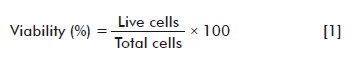
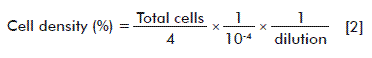
Cellular proliferation
A total of 1.0 × 104 cells/mL were grown in 96-well culture plates (final volume 100 µL) in an RPMI 1640 medium supplemented with 10 % SBF. They were incubated for 24 hours at 37 ºC, under 5 % CO2 and at a relative humidity of 95 %. The culture medium was eliminated and 100 µL of the culture medium with the different aggregate concentrations that were to be evaluated: 50, 25, 12.5, 6.25, 3.125 and 1.5625 µg/mL, were added to each well. A negative control (untreated cells) and a positive control (cells treated with 10 µL of DMSO) were made. After 24, 36 and 48 h of treatment, the culture medium with the NPs was removed and two washings were made with the phosphate buffer solution (PBS; JT Baker). Later on, the cells were detached with 0.25 % trypsin and diluted in a 1:1 proportion with 0.4 % Trypan blue. Samples of 10 μL of the mixture were placed on the Neubauer plate (hemocytometer) and viable cells (transparent cells) and dead cells (blue cells) were counted under the optic microscope. The experiment was done in triplicates for each treatment at each time point. Finally, the cell concentration was calculated by the formula of equation [2].
Mitochondrial metabolic activity by 3-(4,5-dimethyltiazol-2-ilo)-2,5-diphenyltetrazol bromide (MTT)
These assays were made in cell lines CHO-K1 and CAL27. A total of 2 × 104 cells/mL were grown in 96-well culture plates (final volume 100 µL). They were incubated for 24 h at 37 ºC, under a 5 % CO2 atmosphere and 95 % relative humidity. The culture medium was eliminated and 100 µL of the culture medium with the different concentrations of nanoparticles that were to be evaluated: 50, 25, 12.5, 6.25, 3.125 and 1.5625 µg/mL, were added to each well. A negative control (untreated cells) and a positive control (cells treated with 10 µL of DMSO) were made. After 24 h, the culture medium with the NPs was eliminated and two washings with PBS were made and a new culture medium with 10 µL of MTT (Alfa Aesar) at a final concentration of 0.5 µL, were added to each well. This was incubated for 4 h in the dark, the medium was eliminated and 100 µL of DMSO were added.
Then it was shaken for 10 min at 6 rpm and the absorbance value was read at 570 nm, in a plate reader. The experiment was carried out by triplicate for each treatment and the average of the results of the two separate experiments is shown. The percentage of metabolic viability for each one of the concentrations was obtained using the equation:

The statistical analysis is based on the comparison of the averages of the treatments in relation to the negative control, with the viability percentage obtained as a response variable.
Comet assay genotoxicity test
The assessment for genotoxicity of the NPs aggregates was made through a contract with the Genetic, Regeneration and Cancer Group of the School of Exact and Natural Sciences, University of Antioquia. This assessment was made through the alkaline electrophoresis of individual cells assay, also called Comet Test or Assay, according to the laboratory protocols. The assay was made in primary human lymphocyte cultures and in the CHO-K1 cell line, treated for 24 h with aggregate concentrations: 12.5, 6.25, 3.125 and 1.5625 µg/mL.
Statistical analysis
The results were processed with the IBM SPSS program (IBM SPSS Statistics for Windows, Version 20.0; IBM Corp.). Data were expressed as median, following the Central Theorem of Limit. For the bivariate analysis, the Kruskal-Wallis test was carried out and the Mann Whitney U test was made with Bonferroni’s corrections as post-hoc analyses for comparisons between groups. In all cases, statistical significance was considered for p < 0.05.
RESULTS
Nanoparticle synthesis
As shown in SEM and TEM micrographs, NPs were effectively synthesized through the sol-gel method and assisted by reverse micelle micro-emulsion, taking into account the synthesis parameters affecting the size and morphology of the particles (Figure 1A and 1B). NPs were aggregated, which is explained by the interaction forces between the particles that naturally tend to cluster together due to their small size. NPs aggregates were approximately 1 µm in size, being formed by an important number of NPs with sizes ranging from 6 nm to approximately 200 nm (Figure 1C). Distribution behavior was monomodal, with a maximum peak at approximately 27 nm, the average size of the particles obtained.
Cytotoxicity assessment
Viability and cellular density
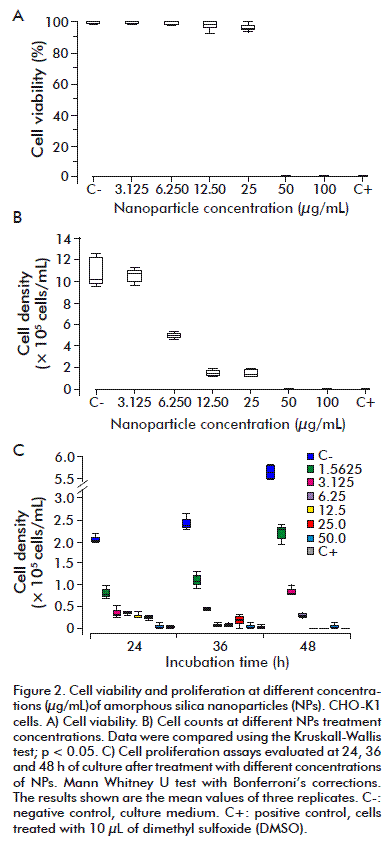
After the treatment with the aggregated NPs for 24 h, cell viability was observed to be over 90 % in the CHO K-1 line for concentrations ranging from 3.125 to 25 µg/mL. No effect on cell viability was observed in the wells with the treatments of 3.125 and 6.25 µg/mL on comparing them to the negative control (p ≥ 0.05). Furthermore, density was found to be equally affected by 12.5 µg/mL and 25 µg/mL or by 50 and 100 µg/mL of aggregates, where the latter concentrations had the same behavior as the positive control with a cellular viability of zero (p ≥ 0.1) (Figure 2A). In spite of the high percentage of viability at lower concentrations, on counting the cells it was interesting to see that cell number was less than those initially grown in the wells with the treatments starting with 12.5 µg/mL NPs and above, and that the cells treated with 6.25 µg/mL had a lower count than the negative control, indicating that cell density is affected by the NP aggregates. This was confirmed with the cell density test, where the number of cells decreased in more than 50 %, starting from the 6.25 µg/mL treatment, on comparing them with the negative control and with the well of 3.125 µg/mL. It was observed that this decrease was dose dependent, where the concentrations of 12.5 µg/mL or higher, affected cell density more strongly. It is relevant to point out that differences between the concentrations of 12.5 and 25 µg/mL, and 50, 100 µg/mL and the positive control were statistically non-significant (p = 0.0590 and p ≥ 0.01, respectively) (Figure 2B).
A large number of NPs aggregates were observed at the concentration of 100 µg/mL, which was so high that the cells could not be seen. In view of this, and the previous results of viability and density, we decided to eliminate that concentration and include a dilution that would be inferior to the lowest one previously used (1.5625 µg/mL) to carry out the subsequent experiments.
Cell proliferation
According to the cell density observed, it was essential to know if the aggregated NPs affect cell proliferation; for this the cultures with the treatments at the described concentrations were followed for 24, 36 and 48 h of growth. The number of cells was doubled after 24 h in the negative control, where we observed this trend up to 48 h, as was expected in a culture that is in an exponential phase. The proliferation of cells treated with the NPs was, however, affected at all concentrations. In the highest concentrations the behavior was the same as in the positive control, while in the lowest concentrations there was a decrease in proliferation when compared to the negative control (Figure 2C). No statistically significant differences were observed between the negative control and the treated cells in regard to time periods. When the cultures were observed at the different time points, they showed variations in the number of cells, their morphology and the NPs aggregates, between the wells with different concentrations. It was found that at higher NPs concentrations there are more NPs aggregates and less cell density, even at 50 µg/mL the NPs aggregates made it impossible to observe the cells. Figure 3 shows images of the cultures taken at 36 h.
Mitochondrial metabolic activity by MTT
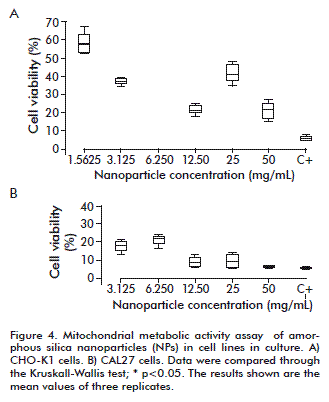
On assessing the mitochondrial activity in cells exposed to NPs, it was observed that in the CAL27 cell line, cellular viability was lower than 50 % at all NPs concentrations, compared to the negative control. Meanwhile, the viability of the CHO K1 line was of about 60 % when treated with 1.5625 µg/mL of NP and lower than 50 % for the rest of the concentrations (Figure 4). In the CHO-K1 line, statistically significant differences were found between all NPs concentrations (p ≤ 0.05) with the exception of 3.125 and 25 µg/mL, with a similar behavior at 50 µg/mL. Treatments with 1.5625, 3.125 and 6.25 µg/mL were statistically different among them and also from the positive control (p = 0.0006). Overall, the dependence of mitochondrial metabolic activity was demonstrated to be a function of the NPs concentration.
The best cell viability was above 50 %, being obtained at 1.5625 µg/mL, Although all NPs concentrations decreased cell viability in 40 % or more, they were all less cytotoxic than the positive control treatment (10 µL DMSO, p = 0.0006). In the CAL27cell line, all NPs concentrations had a marked cytotoxic effect, with a decrease in cell viability that was greater than 60 % in all cases. On analyzing the differences between the concentrations studied, however, a dose dependent effect of viability was observed (p ≤ 0.05). There were differences between all concentrations with the exception of 12.5 and 25, which were the same (p = 0.0958).
Genotoxicity assessment by the Comet assay
Considering the results obtained in the cytotoxicity tests, we decided to assess the genotoxic effect of the NP only at lower concentrations and in two different cellular model systems: the CHO-K1 line and primary cultures of human lymphocytes. A different behavior was found on comparing the genotoxicity of NPs in both models, their genotoxicity being higher for the CHO-K1 cells than for the human lymphocytes. In the CHO-K1 cell line, all concentrations assessed showed greater genotoxicity than the negative control (p ≤ 0.05), while in the primary cultures of human lymphocytes there were no significant differences between the negative control and the treated cells. Micrographs of the Comet assay tests are shown in figure 5.
DISCUSSION
The amorphous silica NPs analyzed in this study remained as aggregates in the culture media in spite of the application of a dispersion protocol. This has been reported by different groups, demonstrating that NPs form aggregates when they are in contact with physiological solutions (culture media, sera and buffer solutions, among others) [12-14]. This may affect the toxicity produced, as demonstrated by Mu et al. [12]. It is possible that under our conditions the formation of aggregates may have affected cytotoxicity, in spite of having carried out the dispersion protocols of the NPs before their inclusion in the culture medium. This may be assumed when considering that both the formation of the aggregates and cytotoxicity depend on NPs concentrations.
The viability and cell density results may be explained by the lysis produced by the interaction between the NP and the cells. Mu et al. observed the detritus of the A549 cells (human lung epithelial cells) accompanied by the NP through transmission electron microscopy, when they were incubated with 100 µg/mL of amorphous silica NPs for 24 h [12]. These results may be compared with our own findings in the wells of 50 and 100 µg/mL, but they contrast with the wells treated with 3.125 µg/mL that behaved like the negative control, indicating that the cytotoxicity of the NPs was concentration-dependent. Nevertheless, comparisons must be cautiously considered between experimental settings, particularly the differences in cell line lineage and culture conditions. Although it has generally been considered that the silica NPs are non-toxic [15], various in vitro studies using the MTT technique have demonstrated that cytotoxicity induced by NPs depends on the NPs concentration, as we have detected in our experimental setting. For example, Yang et al. [16] found that silica NPs of 15 and 30 nm were able to induce cytotoxicity and apoptosis in a dose-dependent manner in HaCaT cells (human keratinocytes), using concentrations in the range 2.5-80 µg/mL [16]. At the same time, a direct dependence was found between NPs concentration and cytotoxicity induced by both ROS and the depletion of anti-oxidant mechanisms in liver HepG2 cells, at 1-200 µg/mL NPs concentrations [17].
Once analyzing the cellular proliferation results, a decrease in treated cells as a function of the NPs concentrations was also detected, but not depending on the exposure time. Notably, the time of exposure dependence of NPs cytotoxicity have been previously reported in several works. For instance, Ye et al. found a time-dependent reduction of cellular viability in human liver cells (L-02) exposed to silica NPs of 21 nm [18]. Viability of the A549 cells was demonstrated to progressively decrease over time (24 h, 48 h, 72 h) in the presence of silica NPs of 15 and 46 nm [9]. Nevertheless, in our study, no dependence of cytotoxicity was found on the exposure time, with a decrease in cellular viability compared to the start at all the time points tested (24, 36 and 48 h).
It has been demonstrated through in vitro and in vivo studies with silica NPs that they can produce cytotoxicity either directly or indirectly; for example this may be derived from its internalization by cells and the effector responses against them, triggering cellular stress mechanisms such as ROS production [6, 7, 12, 17] and attacking the DNA structure with single or double strands breaks among other damages, which may be detected using the Comet assay [19]. Our results differ from previous studies, since we observe genotoxicity in the CHO K-1 cell line and according to findings in human lymphocytes, there does not seem to be an increase in basal DNA damage levels produced by the exposure to NPs. In other words, they do not produce an increase in the simple or double stranded DNA breaks as expressed in the tail of the comet assay.
In our study we have observed that the toxic effects induced by the NPs not only depend on the concentration, but also on the cell model used, since both cytotoxicity and genotoxicity results showed differences between cell types; this is in agreement with previous reports in the literature [15, 20, 21]. As mentioned above, these toxic effects have been explained by different mechanisms derived from the interaction between the NPs and the cells. These may include the damage to the cell membrane, inflammatory reactions, mitochondrial damage, the production of ROS and NOS, apoptosis and necrosis, among others [12, 17, 21-24], which could mediate some of the effects seen in our study. Nevertheless, our experimental setting does not supports further explanation of the cytotoxicity and genotoxicity results based on such mechanisms.
Hence, these results should be interpreted cautiously, since the demonstrated toxicity the variability found in relation to the NPs concentration and cell type does not necessarily indicate that the amorphous silica NPs should not be used, but that the toxic effects can be controlled or avoided. For instance, when it is used at non-toxic concentrations or when these NPs work are functionalized with specific chemical groups, the deleterious damages in cells may be decreased as previously demonstrated [10, 25]. Such strategies, together with the exclusive chemical and physical properties of the amorphous silica NPs, could make such particles a feasible option for its use as vehicles for drug delivery or in other biomedical applications.
CONCLUSIONS
Variable in vitro effects of cytotoxicity and genotoxicity of amorphous silica NPs aggregates on cell lines and primary human lymphocyte cultures were found in this study. Nevertheless, the variability seen on its effects as a function of the concentration and cell line, in a time-independent behavior, are indicators of the possible use of these NPs at nontoxic concentrations or with chemical modifications that could decrease their toxicity. This could result in successful applications in biomedicine, taking advantage of their particular chemical and physical properties.
ACKNOWLEDGEMENTS
The authors thank to the National University of Colombia and the Cooperative University of Colombia at Medellin, for the funds to carry out this study, and to Dr. Claudia Patricia García for providing the nanoparticles, the invitation to carry out this study and her advisory. Thanks also to the Cellular and Molecular Physiology group of the School of Medicine of the National University of Colombia at Bogota, and to Victor Alonso Solarte David for the technical advice in the MTT technique. The authors are also grateful to Jaiberth Antonio Cardona Arias, Juan Carlos Hernández López and Wildeman Zapata Builes, for their constant technical advice in carrying out the assays and for manuscript review.
REFERENCES
1. Manish G, Vimukta S. Targeted drug delivery systems: A Review. Res J Chem Sci. 2011;1(2):135-8.
2. Moghimi SM, Hunter AC, Murray JC. Nanomedicine: current status and future prospects. FASEB J. 2005;19(3):311-30.
3. Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, Price RE, et al. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc Natl Acad Sci USA. 2003;100(23):13549-54.
4. Rahimi M, Wadajkar A, Subramanian K, Yousef M, Cui W, Hsieh JT, et al. In vitro evaluation of novel polymer-coated magnetic nanoparticles for controlled drug delivery. Nanomedicine. 2010;6(5):672-80.
5. He Q, Gao Y, Zhang L, Zhang Z, Gao F, Ji X, et al. A pH-responsive mesoporous silica nanoparticles-based multi-drug delivery system for overcoming multi-drug resistance. Biomaterials. 2011;32(30):7711-20.
6. Gary-Bobo M, Hocine O, Brevet D, Maynadier M, Raehm L, Richeter S, et al. Cancer therapy improvement with mesoporous silica nanoparticles combining targeting, drug delivery and PDT. Int J Pharm. 2012;423(2):509-15.
7. Costa CAR, Leite CAP, Galembeck F. Size Dependence of Stöber silica nanoparticle microchemistry. J Phys Chem B. 2003;107(20):4747-55.
8. Jin Y, Kannan S, Wu M, Zhao JX. Toxicity of luminescent silica nanoparticles to living cells. Chem Res Toxicol. 2007;20(8):1126-33.
9. Lin W, Huang YW, Zhou XD, Ma Y. In vitro toxicity of silica nanoparticles in human lung cancer cells. Toxicol Appl Pharmacol. 2006;217(3):252-9.
10. Slowing, II, Vivero-Escoto JL, Wu CW, Lin VS. Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv Drug Deliv Rev. 2008;60(11):1278-88.
11. Huang X, Teng X, Chen D, Tang F, He J. The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials. 2010;31(3):438-48
12. Mu Q, Hondow NS, Krzeminski L, Brown AP, Jeuken LJ, Routledge MN. Mechanism of cellular uptake of genotoxic silica nanoparticles. Part Fibre Toxicol. 2012;9:29.
13. Orts-Gil G, Natte K, Drescher D, Bresch H, Mantion A, Kneipp J, et al. Characterisation of silica nanoparticles prior to in vitro studies: from primary particles to agglomerates. J Nanopart Res. 2011;13:1593-604.
14. Chen M, von Mikecz A. Formation of nucleoplasmic protein aggregates impairs nuclear function in response to SiO2 nanoparticles. Exp Cell Res. 2005;305(1):51-62.
15. Uboldi C, Giudetti G, Broggi F, Gilliland D, Ponti J, Rossi F. Amorphous silica nanoparticles do not induce cytotoxicity, cell transformation or genotoxicity in Balb/3T3 mouse fibroblasts. Mutat Res. 2012;745(1-2):11-20.
16. Yang X, Liu J, He H, Zhou L, Gong C, Wang X, et al. SiO2 nanoparticles induce cytotoxicity and protein expression alteration in HaCaT cells. Part Fibre Toxicol. 2010;7:1-12.
17. Ahmad J, Ahamed M, Akhtar MJ, Alrokayan SA, Siddiqui MA, Musarrat J, et al. Apoptosis induction by silica nanoparticles mediated through reactive oxygen species in human liver cell line HepG2. Toxicol Appl Pharmacol. 2012;259(2):160-8.
18. Ye Y, Liu J, Xu J, Sun L, Chen M, Lan M. Nano-SiO2 induces apoptosis via activation of p53 and Bax mediated by oxidative stress in human hepatic cell line. Toxicol In Vitro. 2010;24(3):751-8.
19. Petersen EJ, Nelson BC. Mechanisms and measurements of nanomaterial-induced oxidative damage to DNA. Anal Bioanal Chem. 2010;398(2):613-50.
20. Rabolli V, Thomassen LC, Princen C, Napierska D, Gonzalez L, Kirsch-Volders M, et al. Influence of size, surface area and microporosity on the in vitro cytotoxic activity of amorphous silica nanoparticles in different cell types. Nanotoxicology. 2010;4(3):307-18.
21. Julien DC, Richardson CC, Beaux MF, 2nd, McIlroy DN, Hill RA. In vitro proliferating cell models to study cytotoxicity of silica nanowires. Nanomedicine. 2010;6(1):84-92.
22. Corbalan JJ, Medina C, Jacoby A, Malinski T, Radomski MW. Amorphous silica nanoparticles trigger nitric oxide/peroxynitrite imbalance in human endothelial cells: inflammatory and cytotoxic effects. Int J Nanomedicine. 2011;6:2821-35.
23. Chang JS, Chang KL, Hwang DF, Kong ZL. In vitro cytotoxicitiy of silica nanoparticles at high concentrations strongly depends on the metabolic activity type of the cell line. Environ Sci Technol. 2007;41(6):2064-8
24. Zhang XQ, Yin LH, Tang M, Pu YP. ZnO, TiO(2), SiO(2,) and Al(2)O(3) nanoparticles-induced toxic effects on human fetal lung fibroblasts. Biomed Environ Sci. 2011;24(6):661-9.
25. Liu Y, Zhang Z, Zhang Q, Baker GL, Worden RM. Biomembrane disruption by silica-core nanoparticles: effect of surface functional group measured using a tethered bilayer lipid membrane. Biochim Biophys Acta. 2014;1838(1 Pt B):429-37.
Received in July, 2014.
Accepted in January, 2015.
Diana M Gómez-Gallego. Grupo de investigación Infettare. Universidad Cooperativa de Colombia, Sede Medellín. Calle 48 # 28-00, Medellín, Antioquia, Colombia. E-mail: diana.gomezga@campusucc.edu.co