Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Biotecnología Aplicada
versión On-line ISSN 1027-2852
Biotecnol Apl vol.34 no.4 La Habana oct.-dic. 2017
TECHNIQUE
Methods for determining the biological activity of nematicidal products
Métodos para determinar la actividad biológica de productos nematicidas
Idania Wong Padilla1, Danalay Somontes Sánchez1, Francisco Rodriguez Risco1, Rolando Morán Valdivia1, Nemecio González Fernández2, Carlos Pérez Heredia2, Yunier Paneque Díaz2
1 Departamento de Investigaciones, Centro de Ingeniería Genética y Biotecnología de Camagüey. Circunvalación Norte y Ave. Finlay, Camagüey, Camagüey, Cuba.
2 Grupo de Desarrollo Tecnológico, Centro de Ingeniería Genética y Biotecnología de Camagüey, Cuba.
ABSTRACT
Three methods are presented, to evaluate the nematicidal activity of the bioproduct HeberNem® against eggs and larvae of Meloidogyne spp. They are a tool to determine the nematicidal effect of formulations of bioproducts composed of microorganisms that affect the life cycle of the nematodes. The first method or in vitro test allows determining the percentage of inhibition of egg hatching with a coefficient of variation lower than 5 %, and the percentage of reduction larvae survival vs. control with a coefficient of variation lower than 15 %. The second method allows calculating the concentration of microorganisms capable of producing hydrogen sulfide during the product’s development studies. And the third method consists of an essay in 1 L pots containing a 1:1 (v/v) peat-sand mixture indicator plants, to determine the technical effectiveness of the product against eggs and larvae of Meloidogyne spp. Five weeks after sowing, the degree of root infestation is determined and the effectiveness of each treatment is calculated with respect to the untreated control. The HeberNem® formulations tested showed greater than 90 % egg hatching inhibition and 52 % larval survival reduction for the 5 × 106 c.f.u./mL concentration. In the pot trial, the technical effectiveness obtained was greater than 50 % with respect to the control.
Keywords: biological activity, HeberNem, Meloidogyne, hydrogen sulphide, nematodes, nematicides.
RESUMEN
El presente trabajo muestran tres métodos que evalúan la actividad nematicida del bioproducto HeberNem® frente huevos y larvas de Meloidogyne spp. Estos son una herramienta para determinar el efecto nematicida de las formulaciones de bioproductos compuestos por microorganismos que afectan al ciclo de vida de los nematodos. El primer método o ensayo in vitro permite determinar los porcientos de inhibición de la eclosión de los huevos con un coeficiente de variación menor al 5 %. Y el porciento de reducción de la supervivencia de las larvas con respecto al testigo, con un coeficiente de variación menor al 15 %. El segundo método permite calcular la concentración de microorganismos capaces de producir sulfuro de hidrógeno en las formulaciones durante el desarrollo del producto. Y el tercer método consiste en un ensayo en macetas de 1 L que contienen una mezcla de arena:turba en proporción 1:1 (v/v), con plantas indicadoras para determinar in vivo la efectividad técnica del producto frente a la infección con huevos y larvas de Meloidogyne spp. A las cinco semanas posteriores a la siembra se determina el grado de infestación de las raíces y se calcula la efectividad de cada tratamiento con respecto al testigo no tratado. Las formulaciones de HeberNem® ensayadas mostraron una inhibición de la eclosión de los huevos superior al 90 % y una reducción de la supervivencia de las larvas del 52 %, para la concentración de células de 5 × 106 c.f.u./mL. En el ensayo en macetas la efectividad técnica obtenida fue superior al 50 % respecto al testigo.
Palabras clave: actividad biológica, HeberNem, Meloidogyne, sulfuro de hidrógeno, nematodos, nematicidas.
INTRODUCTION
The production of vegetables in Cuba through protected cultivation technology began in the 1990’s with the objective of satisfying the growing demand and reducing the damages and losses caused by the pests of some insects that attack these crops. But soon a new threat emerged affecting plantations: the root-not nematodes of the genus Meloidogyne [1-4]. In studies conducted in eight provinces in Cuba, Meloidogyne spp. was considered the main problem in tomato and melon, followed by pests of insects and several phytopathogens. In the case of pepper, the mite Polyphago-tarsonemus latus constitutes its main problem, followed by insects and Meloidogyne spp. [2, 5].
For several years, the chemical control was the single strategy available against root-knot nematodes, with methyl bromide as the main soil fumigant. Later on, its use was restricted first and further banned due to the harmful effects to the environment and man [1, 6]. Since then, new studies were started and other improved management practices applied worldwide against this pest [7, 8], with biological control among them [9-12]. The most widely used bio-product in Cuba is the bionmaticide HeberNem®, with a total of 2560.65 hectares treated so far. The effectiveness of this product is mediated by the combined action of the metabolites produced by the Tsukamurella paurometabola bacterium which is its active component together with the hydrogen sulfide released in the closest vicinity of the nematode and the ammonium hydroxide formed [13]. Besides, the extracellular excretion of proteases and chitinases weakens the egg wall (composed of 70 % chitin) and damage the cuticle of the larvae [5].
However, to develop and also for the wide application of this type of bioproduct, it is required to implement effective analytical assays, in order to measure the activity of the bioproduct and also to assay its nematicidal potential and effect. Therefore, this work was aimed to present the analytical methods used for both, determining the biological activity of the nematicidal product HeberNem® and to test its nematicidal activity in pots. These procedures could be also applied to test other bioproducts for the same purpose.
MATERIALS AND METHODS
Method 1: In vitro method to determine nematicidal activity against nematode eggs and larvae
Preparation of eggs and larvae
Meloidogyne spp. egg masses were removed with needles under a stereo microscope from the roots of infested plants as previously described [14-16]. Then, the masses were placed in a 0.5 % sodium hypochlorite solution for 3 min and subsequently washed three or four times with sterile deionized water to remove the remaining hypochlorite [5, 15, 18]. Larvae were obtained from eggs by incubating it in sterile deionized water at 28-30 ° C for 5-6 days [15, 16, 18].
HeberNem® sample preparation
All samples were prepared just before testing. Bioproduct samples (1 mL for the liquid or 1 g of the solid pre-sentations of HeberNem®) are resuspended in 10 mL of 0.1 % water peptone solution and suspended in up to 10 mL of the same solution. Then, four 1:10 serial dilutions are prepared.
Assays
Sterile 24-well plates (Nunc, USA) containing 0.9 mL of 0.1 % water peptone solution and 100 eggs or larvae are applied per well. Subsequently, three replicates were prepared and 0.1 mL of the last dilution of each product samples were added to the wells. The controls were prepared by adding the same volume of water peptone solution without product. The plates were incubated at 28-30 °C for 96 h, and hatched larvae were counted on each well, expressed as egg hatching percentage. The percentage of hatching inhibition was calculated for each sample by dividing the percentage of hatched eggs of each treatment over the hatching percentage of the controls [15, 16, 18]. For larvae, they were transferred to sterile deionized water after a 72-h of incubation with the samples, and incubated for other 24 h before the final count of live and dead larvae under the microscope. Larvae survival was calculated at the end of the test and expressed as percentage. Results were statistically analyzed by using the STATGRAPHICS Centurion XVI, version 16. 1. 11 software.
Method 2: Concentration of microorganisms capable of producing hydrogen sulfide
The method is based on the reaction of the hydrogen sulfide produced by the microorganism with lead acetate, leading to the formation of lead sulfide. The reaction occurs on a filter paper Watman 3MM (Whatman, UK) slightly moistened with lead acetate. Samples taken directly from the bioproducts or soil samples previously treated with them were tested. Standard curves were from a HeberNem® stock solution of known concentration in the concentration range of range 104-109 c.f.u./mL.
Samples were analyzed in duplicates. For this, 1 g of each sample was placed into a sterile assay tube containing 9 mL of 0.9 % NaCl (Merck, Germany) solution supplemented with 1 g/L peptone (Oxoid, UK) and 1 mL/L of Tween 80 (Merck, Germany). The tube was gently shaken for 30 min at room temperature. Then, two 1:10 dilutions of each sample were prepared. For the test, 24-well plates were used, by adding 1 ml of each sample dilution in triplicates. The hydrogen sulfide production was induced by adding 0.1 mL of 15 mM Cysteine (Sigma-Aldrich, USA) solution into each well, placing a filter paper on the top of the plate and further adding 20 μL of 0.1 M lead acetate (Sigma, USA) solution on the filter paper at the top of each well. The plate was tightly capped, to avoid gas release as the reaction took place. The assay was run for 18-20 hours at 37 °C for hydrogen sulfide production. The filter paper was subsequently removed from the plate, and the amount of lead acetate formed corresponding to the samples wells was estimated as proportional to the intensity of the dark spots of lead sulfide produced. Spots are then measured by densitometry, using the Scan Prot Software (Bover Fuentes E, CIGB). Bacterium concentration (c.f.u./mL) was estimated by interpolating the values of spot intensity of the samples assayed into a standard curve of spot intensity vs. T. paurometabola samples of known concentration. Data adjustment and the best fit curve equations were generated with the aid of STATGRAPHICS Centurion XVI, version 16. 1. 11 software.
Method 3: Pot trial with indicator plants
In this trial, a 1:1 mix of sterile sand and peat as substrate in 1 kg pots is used. Infestation was performed with 3000 Meloidogyne sp. eggs separated from roots of infested tomato plants [16].
The bioproduct formulations are applied directly to the substrate three days before the infestation. The indicator plant used in this trial was the tomato susceptible variety UC 82B. The degree of root damage was determined as Root Gall Index (RGI), 35 days after planting, according to the scale of 0-10 degrees [19]. Infestation severity was evaluated using the Townsend-Heuberger’s formula [20]:
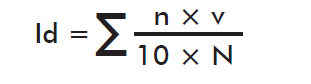
where Id is the index of treatment infestation with the formulation evaluated; n is the degree of infestation according to the scale; v is the number of plants per degree of infestation and N the total number of plants evaluated. Id is also calculated for the control without treatment, in which case it is named Ia.
The technical effectiveness of each Hebernem® product formulation (ET (%)) was calculated using the Abbott’s formula [21]:
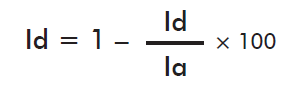
RESULTS AND DISCUSSION
Methods to evaluate the mechanisms of action of nematicidal bioproducts are essential to select the optimal product for nematode control in plants. They also provide us with information on the quality of the different formulations under analysis. Regarding HeberNem®, its active ingredient (T. paurometabola) has been studied and patented as nematicidal agent, including the methods used to determine its biological activity [22, 23]. In those studies, pure cul-tures in LB liquid medium were assayed. However, in this work a new strategy was followed (Method 2), by separating the different components from the bacterium as a pre-treatment before starting the biological activity tests of the product formulation. Therefore, 1 g of HeberNem® was washed twice with a 0.1 % sterile peptone solution and cellS were subsequently resuspended in the same solution before use.
Analyzing the results obtained by this first method, the bioproduct HeberNem® exhibited more potent nematicidal activity against eggs than larvae of M. incognita. This is significant because the egg stage is the one showing the highest resistance during the entire nematode life cycle [24].
The results of two assays for the determination of nematicidal activity of a solid HeberNem® formulation against nematode eggs are shown in Table 1. The product caused 96.14 % of egg hatching inhibition. When evaluating different serial dilutions of the bioproduct (1/10), up to the order of 5.85 × 104 c.f.u./ mL, more than 90 % inhibition of egg hatching was observed (Table 2).
Three product dilutions were also tested to evaluate the effect of HeberNem® on larvae, the lowest containing 5 × 106 c.f.u./well. It was demonstrated at this cellular concentration that the product was able to reduce larval survival by 51.9 % (Table 3).
The results obtained by this method in the evaluation of the nematicidal activity of the different HeberNem® formulations corroborate previous results by Mena et al. [5, 23], by using culture supernatants of T. paurometabola active component of HeberNem®. In that study, the damage caused in eggs and larvae of nematodes was found as caused by proteases, chitinases and collagenases excreted by bacteria into the culture medium.
Many bacteria displaying nematicidal activity are able to produce hydrogen sulfide from amino acids such as cysteine. This is the case of the active component bacterium of the HeberNem® bioproduct [23]. In fact, its evaluation in vitro against different species of nematodes (zoonematodes and phytonematodes) demonstrated that the nematicidal effect was mediated in part by the hydrogen sulfide produced by the bacteria during their growth in liquid LB medium which gets into contact with the eggs and larvae of the nematodes [23].
Results obtained with Method 2 are shown in figure 1. The observed dark areas correspond to the lead sulfide formed by the reaction of the lead acetate and the hydrogen sulfide released by the bacteria contained in the product (Figure 1A). This method supports the quantification of the concentration of microorganisms capable of producing hydrogen sulfide, through the presence of the lead sulfide formed on the surface of the filter paper, further indicating the development of this property on the microorganisms by the pretreatment. The intensity of the lead sulfide spots formed is proportional to the c.f.u./mL present in the formulations assayed. Moreover, the high sensitivity of this method allows detecting small differences in the microbial concentration of the formulations tested. Microorganisms’ concentrations were determined by standard curves (Figure 1B and 1C) and the result of two different formulations of the product is shown in Table 4. The result of the liquid formulation of a known concentration was included as control.
The potting results showed a reduction in nematode infestation rates in the roots of plant treated with the different HeberNem® formulations, in comparison with the degree of infestation in control plants (Table 5). The technical effectiveness of the treatments was calculated from the degrees of final infestation of each treatment with the Bridge and Page’s 0-10 scale as described [19]. It was shown that the new formulations were effective for nematode control (Table 5).
CONCLUSIONS
The methods described here are very useful in the evaluation of the bioproducts for the control of nematodes affecting plants. The specificity of the test against eggs and larvae allowed us to evaluate the effect of the bioproduct formulations over different developmental stages of the nematodes. The HeberNem® formulations tested showed nematicidal activity on eggs and larvae of Meloidogyne spp. The effect on nematode eggs was higher (95.14 % inhibition of hatching) compared to the effect on the larvae (68.9 % reduction in survival).
Through the second method proposed here (Method 2), the concentration of active microorganisms in the bioproducts was estimated by the indirect measure of hydrogen sulfide. Potting assays were used to evaluate the technical effectiveness of the three different treatments and formulations under study, and all the formulations tested exhibited nematode control activity for a technical effectiveness greater than 50 %. Thus, it is possible to give an appropriate evaluation on the quality of the nematicidal bioproduct HeberNem® or other similar bioproducts for nematode control with the use of these three methods.
REFERENCES
1. Cuadra R, Zayas MA, Meléndez O, Ramos N, Alvarez S, Liñeiro LD, et al. Medidas agrotécnicas para el control de Meloidogyne incognita en cultivo protegido del pepino. Fitosanidad. 2013;17(3):161-5.
2. Gómez L. Diagnóstico y potencialidad de la práctica agrotécnica en el manejo de Meloidogyne spp. en la producción protegida de hortalizas [Tesis al grado de Doctor en Ciencias Agrícolas]: CENSA, La Habana; 2007.
3. Wesemael WL, Viviaene N, Moens M. Root–knot nematodes (Meloidogyne spp.) in Europe. Nematology. 2011;13(1):3-16.
4. Seid A, Fininsa Ch, Mekete T, Decraemer W, Wesemael WM. Tomato (Solanum lycopersicum) and root-knot nematodes (Meloidogyne spp.) - a century -old battle. Nematology. 2015;17(9):995-1009.
5. Mena J, Pimentel E, Veloz L, Hernández A, León L, Ramírez Y, et al. Aislamiento y determinación de cepas bacterianas con actividad nematicida. Mecanismo de acción de T. paurometabola C-924 sobre nematodos. Biotecnol Apl. 2003;20(4):248-52.
6. Pérez E. Tecnologías en el proceso de eliminación del bromuro de metilo en tratamientos al suelo en Cuba. La Habana: Editorial Cidisav; 2012.
7. Medina J, Miranda L, Soria C, Domínguez P, Pérez RM, Zea T, et al. Alternativas biológicas al bromuro de metilo en la fresa de Huelva (España). Resultados de dos años de actividades. In: INISAV. II Taller Latinoamericano de Biocontrol de Fitopatógenos. La Habana: INISAV; 2010. p. 53.
8. Rodríguez R. Management of phytopathogenic nematodes: Present situation and challenges, In: Seminario Internacional de Sanidad Agropecuaria; 2011 May 13-16; La Habana, Cuba: CENSA; 2011. p. 63.
9. Mena J, Pimentel E, Borroto C, Smith E, Mesa L, León L, et al. Extensión Nacional del bionematicida HeberNem, para el control de fitonematodos en los cultivos protegidos. La Habana: Instituto de Investigaciones Hortícolas “Liliana Dimitrova”; 2006.
10. Arévalo J, Hidalgo L, Martins I, Sousa JF, Castro JM, Carneiro M, et al. Cultural and morphological characterization of Pochonia chlamydosporia and Lecanicillium psalliotae isolated from Meloidogyne mayaguensis eggs in Brazil. Tropical Plant Pathol. 2009;34(33):158-63.
11. Raddy HM, Fouad AFA, Montasser SA, Abdel-Lateef MF, EL-Samadisy AM. Efficacy of six nematicides and six commercial bioproducts against root knot nematode, Meloidogyne incognita on tomato. J Appl Sci Res. 2013;9(7):4410-7.
12. Raza MS, Imran M, Afzal M, Azeem M, Riaz A, Awais M. Evaluation of bio-nematicides against the development of root knot nematode (Meloidogyne incognita) in tomato. J Entomol Zool Stud. 2014;2(6):333-5.
13. Hernández A, Pimentel E, Mena J. Minibioreactor-gas collector for determining bacteria-produced hydrogen sulfide. Electron J Biotechnol. 2003;6(3):223-32.
14. Zasada IA, Klassen W, Meyer SLF, Collado M, Abdul-Baki AA. Velvetbean (Mucuna pruriens) extracts: Impact on Meloidogyne incognita survival and on Lycopersicon esculentum and Lactuca sativa germination and growth. Pest Manag Sci. 2006;62:1122-7.
15. Tae-Kyun H, Jae-Kook L, Jae-Won H, Soon-II K, Dong-Ro Ch, Young-Joon A. Toxicity of Kaempferia galanga rhizome-derived extract and steam distillate to Meloidogyne incognita juveniles and eggs, and their effects on Lycopersicon esculentum germination and growth. Nematology. 2010;12(5):775-82.
16. Mena J, Pimentel E, Hernández A, León L, Ramirez Y, Wong I, et al. HeberNem: sustituto de tratamientos químicos en Cultivo protegidos. XVI Forum de Ciencia y Técnica. 2007 [cited 2016 Dec 18]. Available from: http://www.forumcyt.cu/UserFiles/forum/Textos/0908001.pdf.
17. Mc Bride RG, Mikkelsen RL, Barker KR. A comparison of three methods for determining root-knot nematode infection of cotton roots. Nematropica. 1999;29(2):147-51.
18. Sharma RN, Saxena KN. Orientation and developmental inhibition in the housefly by certain terpenoids. J Med Entomol. 1974;11(5):617-21.
19. Bridge J, Page SLJ. Estimation of root-knot nematodes infestation levels using a rating chart. Trop Pest Manag. 1980;26:296-8.
20. Townsend GR, Heuberger JV. Methods for estimating losses caused by diseases in fungicide experiments. Plant Dis Rep. 1943;24:340-43.
21. Abbott WS. A method of computing the effectiveness of an insecticide. J Econ Entomol. 1925;18:265-7.
22. Mena J, de la Riva G, Vázquez R, inventors; Center for Genetic Engineering and Biotechnology, assignee. Nematicide agent and method for the bio-control of nematodes. WO 04794 A1, 1996 Feb 22.
23. Mena J, Pimentel E, Hernández A, inventors; Center for Genetic Engineering and Biotechnology, assignee. Pesticidal and antiparasitic compositions. US Patent 0071663 A1, 2004 April.
24. Wharton D A. Nematode survival strategies. In: Lee DL, editors. The biology of nematodes. New York: Taylor & Francis; 2002 p. 389-411.
Received in May, 2017.
Accepted in October, 2017.
Idania Wong Padilla. Departamento de Investigaciones, Centro de Ingeniería Genética y Biotecnología de Camagüey. Circunvalación Norte y Ave. Finlay, Camagüey, Camagüey, Cuba. E-mail: idania.wong@cigb.edu.cu.














