INTRODUCTION
The effects caused by the magnetic field (non-ionizing radiation) on biological systems (from microorganisms to human) can be harmful or beneficial, and influence on the indoor airborne fungi (Anaya et al, 2021a). These magnetobiological effects depend on the intensity (in amperes: A, the voltage (in volts: V) and frequency (in Hertz: Hz) of electrical alternating current (AC), the magnetic field (density is expressed in teslas: T or in gauss: G), the distance and time of exposure to the magnetic field, as well as the intrinsic and extrinsic factors of the biological system (Barnes and Greenebaum, 2007). This radiation can be applied as a static (SMF) or oscillating (OMF) magnetic field treatments which, when the frequency ranges are below 300 Hz, can be classified as extremely low frequency (ELF) (Paunesku and Woloschak, 2007).
For explaining the magnetobiological effects several individual or combined mechanisms have been suggested which overlap to act on the charged particles at membrane level (Buchachenko, 2016). Initially the effect is on calcium ions (Cefalas et al, 2008) and then the enzymatic complexes are activated (Konopacka et al, 2019). In addition, it was suggested that the OMF-ELF acts on the molecules of water and oxygen (Pang and Deng, 2008), which are necessary for cellular metabolism (Funk, 2018).
Regarding to the subject of magnetobiological effects in microorganisms, the filamentous fungi are the least studied. The vast majority of the studies on ionizing radiation effects in fungi were only focused on considering the genetic effects, i.e. mutational or lethal effects (and even at present this trend persists), excluding other non-genetic effects (Casadesus et al, 1985). These effects are expressed in many different forms according to the fungal species exist in nature and these effects can be harmful, beneficial or harmless depending on nutritional state of fungi and whether it is active or not the cellular division at the time of exposure to ionizing radiation. The fungal hyphae show exquisite sensitivity to their environment (Money, 2021).
Several studies with non-ionizing radiation on filamentous fungi show a similar behavior to the above. For example, the immediate effect of OMF-ELF of 50 Hz and 1 mT applied for 4 h on the fungus Aspergillus niger was revealed and an increased production of citric acid and the cellulase enzyme activity were observed with the exposure time and/or density of OMF-ELF increasing (Gao et al, 2011). In another study, it was suggested that the OMF-ELF of 50 Hz and 0.01 mT increased mycelial growth at early stages of development because increased the ergosterol concentration (lipid of membrane) (Berg and Berg, 2006). One other, an important stimulation between 10 and 70% of the conidiogenesis (conidia germination) was observed when OMF-ELF of 50 Hz and 0.1 to 3.5 mT was applied directly on Curvularia inaequalis and Alternaria alternata (Money, 2015).
Furthermore, the fungal pigments, such melanin, are synthesized to protect them of the radiation has been reported (Mattoon et al, 2021). In some fungi, the pigmentation is related to pathogenicity, such as Aspergillus fumigatus which pigments are associated with the virulence of its conidia and Cryptococcus neoformans (pathogenic yeast) which produces melanin both in vitro and in vivo (Urán et al, 2008). In 2004, this yeast species grew on the ruins of the Chernobyl Thermonuclear Central and was also detected in the surrounding land (Zhdanova et al, 2004). In addition, the yeast growth velocity and melanin production increased when the yeast cells to highest ionizing radiation were exposed (Dadachova et al, 2007; Dadachova and Casadevall, 2008). A similar result was obtained with two other microorganisms (Cladosporium sphaerospermum and Wangiella dermatitidis) that naturally contain melanin (Karpenko et al, 2006). The resistance of melanized fungi to cosmic and terrestrial ionizing radiation suggests that melanin also plays a pivotal role in radioprotection, which with Cryomyces antarcticus was also proved (Pacelli et al, 2017).
With this discovery has been rethinking knowledge in this regard the melanin role, suggesting that fungi do not use pigments to protect against radiation, but rather the pigments are involved in the metabolic pathways to capture radiation and use them as an energy source, just as chlorophyll in photosynthetic organisms to use sunlight energy. Although some scientists have been disputing this idea because the exact mechanism behind the protective property of melanin is unclear and poorly explored (Vasileiou and Summerer, 2021), this finding led to the proposal of a new classification of the microorganisms according to energy source: radiotrophic microorganisms (Karpenko et al, 2006; Pacelli et al, 2017; Malo et al, 2019). Accordingly, this topic was studied on innovative radiation shields, for which biotechnology has unique advantages, such as suitability for in situ resource utilization (ISRU), self-regeneration, and adaptability (Shunk et al, 2020).
According to the literature reviewed, there is uncertainty about magnetobiological effects by direct action of OMF-ELF because they are not properly demonstrated. There are a very large contradiction and non-reproducible magnetobiological observations but no explanations and reliable understanding. It is a discouraging circumstance for magnetobiology (Buchachenko, 2016). However, the scientific evidence from these similar studies on fungi suggests that the results, both with ionizing and non-ionizing radiation, could be indicative of a common mechanism of action of radiation on melanin production (Anaya et al, 2020; Anaya et al, 2021b).
The digital imaging processing (DIP) of fungal growth may contribute to information analysis with accuracy and precision about theses magnetobiological effects. For example, Knudsen et al (2006) obtained a fungal growth simulation model and validated it by fungal images analysis. Steinberg (2007) used DIP to analyze the hyphal growth mechanism in Neurospora crassa and Ustilago maydis. In the same year, Kohli (2007) used the DIP to study polarization factors and their location to increase the apical elongation rate of Ashbya gossypii filamentous fungus. The growth and direction of the tips and apical ramifications of the hyphae depend on consumption (depletion) of nutrients and the biomass density, which favors the circular growth of the fungal colony (Knudsen et al, 2006; Steinberg, 2007).
When environmental conditions are good, the fungal spores expand and form a spherical germ bubbles, and at this stage, hyphal elongation rate is about 0.1 µm/min on rich medium. The hyphal growth and lateral branching results in formation of a dense network of intertwined hyphae, a so-called mycelium. Hyphal elongation rates can reach 3.5 µm/min the border of older mycelia. The conidia are formed in the center of the fungal mycelium in a poorly described process (Kohli, 2007). This is the reason why the DIP can be used, since once the mycelium is seen, the circular superficial growth is observed with the naked eye, while the colony color changes during the conidiogenesis process.
For everything discussed above, the aim of this study was to quantify the superficial growth and the pigmentation of the filamentous fungi colonies by effect of OMF-ELF in two culture media.
MATERIALS AND METHODS
Selection of study strains
The fungal strains used in these experiments corresponded to two species that showed the highest appearance relative frequencies in indoor environments of interest premises in Havana, Cuba (Anaya et al, 2016). Aspergillus niger and Cladosporium cladosporioides strains were selected according to the obtained results in another study where the colony size and the mycelium pigmentation were taken into account once they were grown in two different culture media (Anaya et al, 2021a).
These species were identified by conventional methods using taxonomy keys (Barnett and Hunter, 1998; Bensch et al, 2010; Klich and Pitt, 1994) as Aspergillus niger Tiegh. and Cladosporium cladosporioides (Fresen.) G.A. de Vries. The fungal strains were maintained in culture tubes with Malt Extract Agar (MEA) covered with mineral oil at 4 ºC.
Samples preparation
Both strains were re-cultured in slants of MEA and after the sporulation, with 2 ml of saline solution were covered to obtain a spore suspension (105 spores/ml). Then, 0.1 mL of these suspensions was inoculated into the center of 90 mm Petri dishes containing one of the following solid media: Malt Extract Agar (MEA, nutrient-rich medium) or Czapeck Dox Agar (CDA, organic salts medium). The intention of using two cultural media was to evaluate the effect of OMF-ELF on fungal species according to the nutritional content to which they are subject (Casadesus et al, 1985). The strains were incubated at 30ºC for 10 d.
Magnetic treatment
The control samples did not receive magnetic treatment while the other samples after inoculation were immediately magnetic treated with OMF-ELF 2 mT by 60 Hz/200 V during 2 h. These experimental conditions of magnetic treatment were obtained by numerical optimization of a previous experimental design in which was observed the highest convenience (0.954) of the predictive mathematical model (Anaya et al, 2021a). This predictive mathematical model indicated that the Aspergillus niger colony (strain of high-speed growth) treated magnetically on MEA after to 4.5 d should reach 8.4 cm in diameter (area of 56 cm2).
Such magnetic treatment with OMF-ELF was performed using equipment designed and made by Electronic Technologies in Pinar del Río province, which was supported, calibrated and characterized by the National Center of Electromagnetism Applied (NCEA) of Santiago de Cuba province, Cuba. The equipment consists of a signal generator and a coil air nucleus that consists of a type solenoid cardboard coil of 30 cm in diameter and 20 cm in height and a power generator of 60 Hz and 220 V through a copper wire of 2 mm in diameter and 6.1 Ω of electrical resistance (Fig. 1).
Images acquisition
The technique proposal for the DIP was employee in a similar work (Anaya et al., 2015) using a Nikon digital photographic camera that captures images with spatial resolution of 3648 x 2736 pixels 8 bits each. This digital photographic camera on a universal support laboratory was supported to a focal length of 15 cm of the Petri dishes. The photos were taken for each experiment with both treated and untreated fungal strains in triplicate in each culture medium (A. niger on MEA and CDA; C. cladosporioides only on CDA, as negative control, which being a melanized fungus with these two variables can be quantified very well with DIP due to the good definition of the circular shape and the color contrast). The photos of each Petri dish with the colonies in growth were taken every four hours (six photos per day) for ten days and they were processed by DIP.
Procedure for DIP using MatLab®
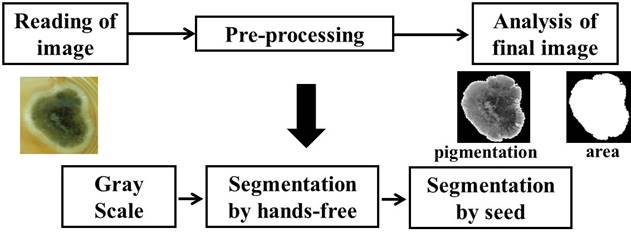
For the DIP the application MatLab® version 2013 was used (Anaya et al, 2015). In general, the procedure was as following: each of the images went through three stages 1- reading of original image, 2- pre-processing and 3- analysis of the final image (Fig. 2). After, the images to grayscale were converted and the histograms were analyzed to determine the thresholds. Regions of interest were segmented by the method of hands-free and the colonies were segmented using a manual segmentation algorithm for sowing seed regions with to remove the edges of Petri dishes.
Finally, the response variables of the colonies to the OMF-ELF were determined: superficial growth (area in cm2) and pigmentation of the mycelium (color intensity). To calculate the area occupied for the colonies on Petri dish was necessary to implement the algorithm for manual segmentation by difference knowing the pixel size. For the pigmentation, was analyzed the average intensities of the segmented gray area in each zone.
Statistical analysis
Three replicates were made of each experiment in triplicate (n = 9), that were processed the results with Statgraphic statistical program. Analysis of variance (ANOVA) was performed to determine the difference of superficial growth (area in cm2) and pigmentation of mycelium (color intensity) by the effect of the magnetic treatment application (controls and treated strains) using Multiple Ranges by the method of least square difference (LSD).
RESULTS
The control colonies (without magnetic treatment) grew typically in each culture medium and the highest fungal growth for both strains was observed on MEA. Figure 3 shows examples of the A. niger growth using a images sequence, which showed a greater variation than the obtained with C. cladosporioides. In addition, it was observed that to both strains the greatest difference in the area began between 3 and 4 days and the pigmentation from 6 days onwards.
Note that the A. niger colony treated on CDA at 10 days (Fig. 3o) reached a similar size that the obtained in MEA at 6 days (Fig. 3d and 3h). Therefore, the maximum stimulation of growth (difference between treated and control strain) was observed in A. niger in CDA similar to C. cladosporioides in this nutrient-poor culture medium (made only with glucose and organic salts), respect to MEA which is a nutrient-rich culture medium.
The strains were inoculated on Petri dishes of 90 mm in diameter after to apply OMF-ELF (60 Hz, 220 V and 2 mT) for 2 h. In MEA: control samples (a-d) and treated samples (e-h); in CDA: control samples (i-l; p-s) and treated samples (m-o; t-λ). From p to s, the size of arrows indicate the average superficial growth between 0.9 and 3.5 cm, and the decreasing size of bars indicate an increase in the pigmented area due to sporulation. The photos are of the days in which the greatest difference with respect to the previous day.
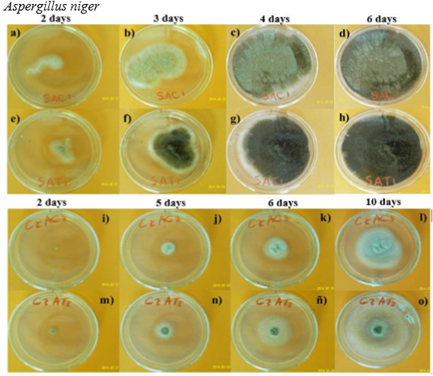
Figure 4 shows some examples that also appear in Figure 3 where images appear in grayscale and segmented images. In the images processed in grayscale with the proposed segmentation method can be distinguished without difficulty the colony pigmentation and the area occupied in the Petri dish. In this respect, it should be noted that in the last two rows of figure 4A the presence of black stains on the colony without OMF-ELF (arrowed) was observed that do not appear on the treated colony and this demonstrate the difference in pigmentation by the magnetic treatment. In the last row of figure 4B it can see the difference between a grayscale segmented image and other non-segmented (before deleting the edges of the Petri dish).

Fig. 4 Grayscale and images manually segmented of colony of Aspergillus niger strain.Note: Strain growth in Petri dishes of 90 mm in diameter, without and with treatment during 2 h with OMF-ELF of 2 mT and 60 Hz/220 V. A) on MEA (the arrows point black stains in the colonies); B) on CDA.
Figure 5 depicts the superficial growth kinetics of the fungal colonies for each experimental condition studied. In the culture medium CDA, the stimulation there was statistically significant for both treated strains against their respective controls (lowercase letters b to e), while this was not observed with A. niger in the culture medium MEA until sixth day of growth (lowercase letter a).

Fig. 5 Kinetics of the superficial growth or area of colonies (cm 2 ) of the fungal strains Aspergillus niger and Cladosporium cladosporioides.
The growth during 10 days of incubation at 30°C, after treatment with oscillating magnetic field (OMF) of 60 Hz / 220 V and 2 mT for 2 h. Colonies grew on Petri dishes of 90 mm in diameter (9 cm; area of 63.5 cm2) with MEA and CDA. The data were fitted to exponential model A = a.t k where: A is colony area (cm2); a: constant model; t: time (day) and k: exponent (between 0.6 and 1.5). Each point represents the average for n = 9 (three replicates of the experiment with the fungal strains cultured in triplicate) and 95% confidence (p ≤ 0.05). The different lowercase letters (a, b, c, d and e) means statistically significant differences.
Statistically significant difference was observed in the pigmentation of all treated colonies regarding to the controls (observe the scales in Fig. 6 and Fig. 7). Therefore, a greater conidiogenesis in the fungi treated was observed. The graphics show the values fluctuations means of the conidiogenesis after each elongation of the hyphae. This behavior was confirmed by interrelating superficial growth kinetics (Fig. 5) with pigmentation (Fig. 6), which indicates similarities between the intensity peaks with increased superficial growth or vice versa.
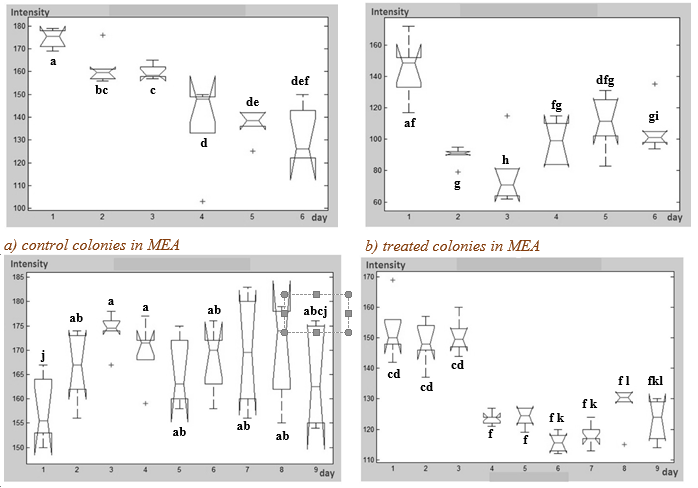
Fig. 6 Variation of color intensity (conidiogenesis) in the Aspergillus niger colonies in MEA and CDA treated with OMF. The central lines of each bar represent the average of all photographs (six photos per day) for n = 9 and 95% confidence (p ≤ 0.05). The different lowercase letters (a to l) means statistically significant differences. Note: The DIP was performed with grayscale-segmented images (Intensity is lower when image is darker and vice versa).
In describing the results shown in Figure 6 highlights that in the culture medium MEA the control of A. niger colony began to sporulate on the second day, in the third day the growth did not show any variation in the intensity of coloration and this behavior continued until the fifth and sixth days. However, the sporulating treated colony in this culture medium continued until the third day and rose sharply until the fifth day with a conidiogenesis to sixth day without significant difference with the achieved the second day. In the case of culture medium CDA, it was observed that the control colony grew until the third day; the sporulation begins on the fourth day and continued to grow almost without variation in the intensity of coloration until the ten days. The treated colony began to sporulate from the first day and the third day showed a stable behavior remained for the rest of the following days of incubation, but with significant differences from control.
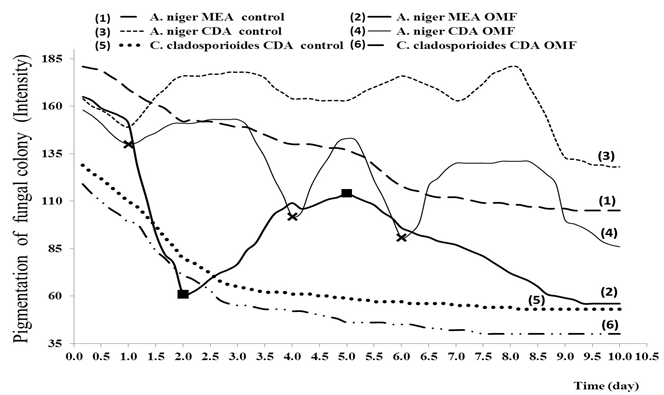
Fig. 7 . Kinetics of the fungal colony pigmentation (grayscale color intensity) as a measure of sporulation. Note: The pigmentation process was monitored for 10 days of incubation at 30°C, after treating the strains with an OMF of 60 Hz / 220 V and 2 mT for 2 h. Colonies grew in 90 mm Petri dishes with MEA and CDA. Each point represents the average of n = 9. The crosses and squares on the lines indicate the most important maximum and minimum values in the behavior observed during this study.
This is shown in details in Figure 7 where it is observed a smooth line of negative slope the graph of A. niger control on MEA medium which differs from the treated colony in same culture medium, while similarity is observed in CDA between the two colonies but with extreme values for the fourth and sixth day.
To verify if the digital processing of imagines was correct, the results of superficial growth (4.5 days) obtained with A. niger was compared to the numerical optimization (8.4 cm on MEA and in 4.2 cm on CDA) obtained by the implemented algorithm (8.3 cm and 3.8 cm, respectively). This represented an error between 4.22 and 8.10%.
DISCUSSION
The fungus can remember the first stressful experience for up to 12 h (Money, 2021). The cellular metabolism was stimulated by magnetic treatment applied which coincides with similar studies (Berg and Berg, 2006; Gao et al, 2011; Anaya et al, 2020; Anaya et al, 2021a), and this was evidenced by the increased superficial growth and pigmentation of the strains. This growth was very closed to the value of the maximum time (4.5 days) indicated by the predictive mathematical model (Anaya et al, 2021a) that was mentioned in the section 2.3.
This demonstrated that OMF-ELF applied could influence the fungal metabolism based on their nutritional state, agreed with another authors (Casadesus et al, 1985). This result could be explained mainly for three reasons: increasing the electrical conductivity of water (Pang and Deng, 2008), the action on charged particles (Buchachenko, 2016) and calcium ions (Ca2+) in both culture media (Cefalas et al, 2008). The metabolism of microorganisms is influenced by these factors, and for fungi case, the amount of calcium ions is decisive for the growth of the polarized apical hyphae (Knudsen et al, 2006; Steinberg, 2007).
A. niger is a hyaline fungi specie (seen under the optic microscope) and their pigmentation evidences the conidiogenesis, which develops depending on their nutritional status (Casadesus et al, 1985). In this study, for example, on the MEA culture medium the conidia were quickly spread throughout the Petri dish while on CDA only they gradually unfold around the point where the initial inoculation occurred. Therefore, to quantify every day the fungal pigmentation was calculated the average of color intensities in the images of A. niger in MEA throughout the segmented area and in CDA was determined only in the central zone of the Petri dish.
Hence, the observed behavior led to the conclusion that the effect is greater the lower the availability of nutrients, which could be explained as an adaptive response to stress (low nutrient and oscillations at the frequency of OMF-ELF applied) that activates the metabolic mechanisms for maximum velocity of reproduction and therefore a maximum fungal survival. That is according with similar studies cited by Money (2021) with radioactive isotopes that have demonstrated that materials are transported between locations via a mycelium, such that a part of the colony that accesses food can feed distant hyphae that they have depleted the available nutrients.
This result of stimulation of fungal metabolism by non-ionizing radiation (OMF-ELF) with a general exponential equation of the form Y = m.x k , agrees with what was observed for ionizing radiation (space radiation) in whose study was obtained a linear model of the form Y = m.x + c (Shunk, 2020). For both cases “x” is the time, but “Y” in the first one is superficial growth of the fungus in the Petri dishes and in the second one is the accumulated radiation counts by the fungus during its growth. That means be indicative of a common response by the same biological mechanism to the radiations.
Regarding to conidiogenesis, an increase in this process was observed in the treated fungi, which is consistent with similar studies (Nagy, 2013). This shows that the fungal strains have a greater sporulation capacity, something was highlighted the 2nd day in the MEA culture medium and the 4th day in CDA.
The behavior described above, it could infer that the effect of the applied OMF-ELF stimulated the growth of A. niger strain, accelerating the elongation of hyphae without changing in the typical way of doing. This indicates that there occurred no genetic changes in accordance with that reported by other authors in relation to ionizing radiation (Casadesus et al, 1985). However, the sporulation capacity was significantly improved.
Therefore, it can be inferred that OMF-ELF causes changes in conidiogenesis when there is abundant availability of nutrients (when the composition of MEA and CDA culture media are compared). But these changes are more significant when the amount of nutrients is decreased causing an increase in the sporulation intensity up achieved highest values when there are sufficient nutrients (marks with crosses in Fig. 7). A demonstration of this explication is the behavior of C. cladosporioides in the CDA, which grew radially and its contrast was well defined because it is a pigmented fungi. Pigmentation of colony treated was stimulated and no fluctuations were observed in the curve of color intensity (completely downward in Fig.7) corresponding to the growth superficial (fully upward in Fig. 5).
The increased accuracy of the measurement in this experiment was due to increase of the pigmentation intensity in the treated colony. However, as A. niger is a hyaline fungus, a good contrast between the mycelium and the culture medium (translucent) cannot be achieved and this makes it difficult to manually define the growth limit surface, while with C. cladosporioides this did not happen because it is a pigmented fungus. Therefore, these results demonstrate that the technique proposal DIP is a useful technique because the procedure used in the algorithm that performs automated quantification had an error below 10%.
This demonstrates the usefulness of the DIP to relate the observation of the macroscopic phenomena (colony) of the magnetobiological effects happened at microscopic level (cell), fundamentally, when applying the gray scale, with the intention of quantifying the superficial growth and pigmentation of filamentous fungi by the effect of OMF-ELF under different nutritional conditions.
CONCLUSIONS
With the application of digital image processing and the segmentation techniques can be monitored the superficial growth of filamentous fungi with accuracy and precision. In this case, the techniques used allowed to quantify the differences obtained not only on growth but also in colony pigmentation due to stimulation with an OMF-ELF on two strains of environmental fungi: A. niger (hyaline mycelium seen under the optic microscope) and C. cladosporioides (pigmented mycelium). Under a low nutrient availability, the OMF-ELF significantly stimulates the superficial growth and the conidiogenesis of fungi, perhaps as a potential adaptive response to stress.