Mi SciELO
Servicios Personalizados
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista Cubana de Química
versión On-line ISSN 2224-5421
Rev Cub Quim vol.29 no.1 Santiago de Cuba ene.-abr. 2017
ARTICULOS
Comportamiento electroquímico de un novedoso electrodo grafito-epóxido modificado para inmovilización covalente de ADN
Electrochemical Performance of a New Modified Graphite-Epoxy Electrode for Covalent Immobilization of DNA
Dr. Abel I. Balbin-TamayoI,II, Dr. Laura S. RisoII, Dr. Ana Margarita Esteva-GuasII, Dr. Pércio Augusto Mardini-FariasI, Dr. Aurora Pérez-GramatgesI*
IDepartamento de Química, Pontifícia Universidade Católica do Rio de Janeiro, Brasil, aurora@puc-rio.br
IIDepartamento de Química Analítica, Facultad de Química, Universidad de La Habana, Cuba
RESUMEN
Un nuevo material compuesto conductor preparado a partir de resina epóxica, grafito y ácido benzoico fue desarrollado para la fabricación de electrodos, los cuales fueron caracterizados por voltametría cíclica, espectroscopia Raman y microscopia electrónica de barrido por emisión de campo (FESEM). La dependencia del potencial pico-a-pico, la corriente anódica máxima, y la relación corriente-anódica-máxima/corriente-catódica-máxima con la velocidad de barrido fueron evaluadas por voltametría cíclica del sistema redox estándar Fe(CN)63-/4-. Se obtuvieron las propiedades voltamétricas del material compuesto conteniendo 1 % en peso de ácido benzoico. El comportamiento electroquímico de la especie electroactiva modelo fue cuasi-reversible, bajo control de difusión lineal. Las constantes de transferencia electrónica heterogéneas fueron determinadas y los valores obtenidos mostraron que la presencia de grupos carboxílicos producto de las modificaciones en el electrodo con 1 % de ácido benzoico no interfiere en la respuesta electroquímica de bases libres y de ADN, siendo esta una propiedad esencial para lograr modificaciones químicas mediante la inmovilización covalente de ADN.
Palabras clave: grafito-epóxido, compositos, electrodo modificado, DNA.
ABSTRACT
A new epoxy conducting composite material prepared from epoxy resin, graphite and benzoic acid was developed and used for the manufacture of electrodes, which were characterized by cyclic voltammetry, Raman spectroscopy and field-emission scanning electron microscopy (FESEM). The dependence of peak-to-peak potential, peak anodic current, and the anodic peak/cathodic peak current ratio with scan rate were evaluated by cyclic voltammetry taking into account the Fe(CN)63-/4- standard redox system. V oltammetric characteristics were obtained using the graphite-epoxy composite containing 1wt % of benzoic acid. The electrochemical behavior of the electroactive model species demonstrates to be quasi-reversible under linear diffusion control. The heterogeneous electron transfer constants were also determined, and the values obtained show that the presence of carboxylic groups from modifications in the graphite-epoxy electrode using 1 % benzoic acid do not interfere with the electrochemical response of free and DNA bases, which is the essential property to pursue further chemical modifications through covalent immobilization of DNA.
Keywords: graphite-epoxy, composite, modified electrode, voltammetry, DNA.
INTRODUCTION
Many research groups are focused on DNA biosensors based on carbon electrodes. Carbon electrodes are particularly suitable for sensing applications because of their low cost, wide working potential window, good electrical conductivity and relatively low background currents [1]. The development of electrodes based on carbon composites of polymeric matrices has particularly led to significant progress in analytical electrochemistry and sensing since different materials can be immobilized within the composites to improve robustness and sensitivity [2, 3]. In addition, the sensing surface can be renewed by a simple polishing procedure, while both, the proximity of the redox centers in the material and the conducting sites on the sensing surface favors the transfer of electrons between the electroactive species [4-6]. In general, these materials demonstrate to have stable electrochemical responses, lower ohmic resistance [7] and longer functional lifetime than typical voltamperometric sensors [8]. Composites are hybrid materials that have well - defined boundaries at microscopic scale despite their homogeneous aspect at macroscopic scale. In Electrochemistry, composite electrodes consist of, at least, one conductor phase (e.g., silver, gold or carbon derivatives such as graphite), and one insulator phase (e.g., organic solvents, silicone, polyurethanes methacrylate or epoxy resin) [2-4, 9-11]. The proportion between these two phases will determine not only the conductivity of the sensor but also the mechanical properties (such as hardness) of the electrode [2, 7]. This kind of electrodes can be manufactured in different shapes and sizes, which allows applications in various media.
In the last years, a great deal of attention has been given to the use of graphite in composite materials, taking advantage of their exceptional mechanical and electrical properties for developing electrochemical platforms [2, 12]. In particular, graphite composites have highly intrinsic resistance and very low relaxation time, within the time scale of the electrochemical processes [9, 13, 14]; thus, parameters and details about the percolation phenomena through the solid can be neglected [15] . The voltammetric responses for these composite materials are reminiscent of those found for microelectrode arrays with "edge effects", high local current densities, as well as partially blocked electrode regions [16]. The graphite composite electrodes can also be easily modified, allowing the incorporation of different components such as ligands, enzymes, cofactors, mediators, catalysts. Therefore, there is a special interest in using these electrodes in the development of feasible sensors for the detection of biologically-relevant molecules, such as DNA of viruses. Detecting viral diseases is usually performed by specific techniques such as immunoassay (ELISA), Polymerase Chain Reaction (PCR), and others [17] . However, these techniques have several disadvantages, such as expensive equipment and reagents involving laborious procedures and requiring experienced personnel.
On the other hand, in the development of an electrochemical DNA biosensor, the immobilization of the probe plays the major role in determining the overall sensing performance. Achieving high sensitivity requires hybridization increase while specific adsorption improves selectivity [2, 12, 18]. In order to develop composite electrodes for such a purpose, the electrode surface modification must improve the mass/electron transport via generation or immobilization of electroactive species close to the electrode-solution interface [19]. In addition, control of the chemical surface and coverage is essential to assure high reactivity, orientation, accessibility and stability of the surface-confined probe, as well as to minimize non-specific adsorption events.
Different immobilization strategies, including entrapment, adsorption and chemical binding, are used to couple nucleic acids onto surfaces [20]. One of the strategies for covalent immobilization is achieved by forming amide bonds between the carboxylic functions at the electrode surface and the amino-terminal group of the oligonucleotides [1, 21, 22]. For this approach, the composite acting as transducer should have carboxylic groups available at the electrode surface to allow a high hybridization efficiency of the immobilized DNA probes; then, regeneration of the probe-modified surface can be achieved by simply rinsing in hot water.
This article aims to conduct the electrochemical characterization and the creation of novel epoxy-based modified graphite electrodes as starting materials for the development of biosensors for DNA viral diseases. The composite electrode preparation included the use of an epoxy resin modified by benzoic acid to introduce the carboxylic groups required for potential covalent binding of biologically relevant species. The composite preparation was optimized using different modifier concentrations, and a (Fe(CN)63-/4-) redox system model to test the influence of benzoic acid on the electrochemical response. The final electrodes were tested using a DNA nitrogen bases-free solution.
EXPERIMENT
Chemicals and reagents
Potassium ferricyanide K3[Fe(CN)6] was purchased from Sigma-Aldrich (USA). Potassium chloride was purchased from Fluka (Switzerland). Graphite powder, benzoic acid and benzyl alcohol were from Merck (Germany) and Araldite® from Beschleuniger DY 964 (Serva Feinbiochemica GmbH, German). Free DNA bases (adenine and guanine), single-stranded DNA (ssDNA) from calf thymus and stock solutions of TRIS-Cl pH buffers (1 mol L-1 pH 8.0) were purchased from Sigma-Aldrich (Germany). Solutions were prepared with deionized distilled water.
Instrumentation
Voltammetry measurements were performed using a BAS CV50W potentiostat with a three terminal electrochemical standard cell containing a Ag/AgCl reference electrode (BAS), a platinum wire counter electrode, and a working electrode (1 mm-diameter graphite-epoxy composite electrodes developed in this work).
The prepared composite electrodes were characterized using Field Emission Scanning Electron Microscopy (FESEM, JSM- 6701F) for surface morphology, and Raman confocal NT-MDT NTEGRA spectrometry (473 nm laser) for chemical composition characterization
Procedures
Composite electrodes preparation: Composite mixtures for the graphite-epoxy electrodes (GEC) were prepared by mixing 0,26 g of graphite powder (50-µm particle size) with 280 µ L of the epoxy resin containing the curing agent, at 5:2 volume ratio. Electrodes which were prepared using this composition showed an electrochemical response similar to the classical response for K3[Fe(CN)6] that is reported for epoxy-graphite electrodes [15]. Benzyl alcohol was used to reduce the viscosity of curing agent. In the case of chemically modified electrodes, the same procedure was used, except that graphite powder was previously mixed with benzoic acid at 1wt % and 5 wt %, and denoted as GEC-1 % and GEC-5 %, respectively.
The electrode body consisted of a copper wire inserted into a glass capillary tube of 1 mm-diameter, which end was previously packed with the composite mixture. The electrode was heated in an oven at 40 °C during 48 h. Before each measurement, the electrode surface was moistened with water and then thoroughly smoothed, first using abrasive sandpaper and then, alumina paper in order to obtain a reproducible electrochemical surface.
Preparation of DNA Bases and ssDNA Solutions: Stock solutions of adenine (free DNA base, 0,2 µmol L-1) and Calf Thymus (ssDNA, 5 µg mL-1) were prepared in TRIS-Cl (1,0 mol L-1, pH 8,0). As guanine is not completely soluble in this buffer, a saturated guanine solution in TRIS-Cl (pH 8,0) was used.
Analytical Procedures: Voltammetric experiments were performed by measuring the potential and peak current of Fe(CN)63-/4- redox system, which was used as a mediator to evaluate the electrochemical behavior of composite electrodes. Cyclic voltammetric measurements were made using a potential scanning from 0 to 0,350 V, then reversing the direction back to 0 V. Square wave voltammetry of DNA bases was also performed with anodic scanning from 0,100 to +1,300 V, amplitude of 0,025 V. All experiments were performed in triplicate, at room temperature (about 25 °C).
RESULTS AND DISCUSSION
Surface Characterization of Graphite-Epoxy Composites
Field Emission Scanning Electron Microscopy (FESEM, JSM- 6701F ) was used to characterize the surfaces of the graphite-epoxy electrodes prepared in this work (figure 1). In both surfaces, it is possible to observe clusters of material gathered at random areas over the surface. The light points are associated with the epoxy resin (insulator) and the dark areas correspond to the graphite particles (conductor) in the blend. The graphite-epoxy electrode surface appears to be relatively rough, not showing significant differences between the unmodified and the modified composites. These images are similar to those reported for a graphite-epoxy electrode used as conducting carbon composite in bio-affinity platforms [23]. Therefore, we can consider that the introduction of benzoic acid in the composite mixture does not modify significantly the morphology of the surface layer of the electrode, which likely retains its conductive properties.
This type of composite electrode, formed by graphite particles that are randomly distributed and oriented in the epoxy resin, can be seen as a macroelectrode formed by an array of a large number of carbon microdisc or microelectrodes fiber [28]. As a result, the sum of the individual currents generated by each microelectrode would generate a net stronger signal, as that of a macroelectrode, but with high signal-to-noise ratio, which is characteristic of microelectrodes. This behavior has been associated with the advantages of using graphite epoxy electrodes in the development of different biosensors, such as performing higher-speed voltammetry and enhanced rate of mass transport of electrochemical species [9, 23, 24].
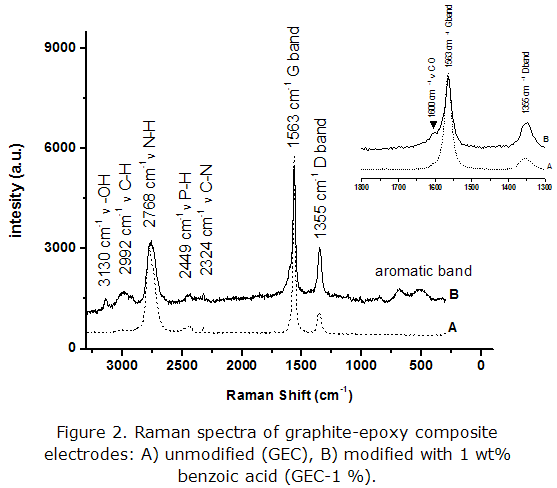
The presence of functional groups that could confirm modifications on the surface of the graphite-epoxy composites was assessed by Raman spectroscopy, an important technique to characterize carbon-based materials (figure 2).
In both spectra, peaks corresponding to C-N and P-H bonds (2 324 cm-1 and 2 449 cm-1, respectively) can be observed, a characteristic element of the epoxy resin groups, as well as those related to the G and D bands of graphite (1 563 cm-1 and 1 355 cm-1, respectively) [25-28]. The Raman spectrum of the electrode modified with benzoic acid (GEC-1 %) also showed signals corresponding to vibrations around 1 600 cm-1 (C-O bond), as a small shoulder in the 1 563 cm-1 peak of graphite. This kind of signal was not observed in the spectrum of the unmodified electrode. Moreover, some typical signals related to aromatic moieties (around 500-650 cm-1), can also be observed, with low intensity, in the GEC-1% spectrum (figure 2, inset). All these results are consistent with the presence of carboxylic groups on the electrode surface GEC-1 %.
Characterization of Graphite-Epoxy Composite Electrodes (GEC)
Electrochemical Behavior of Fe(CN)63-/4- Redox System Using Graphite-Epoxy Composite Electrodes
Cyclic voltammetry study of electroactive species enables to obtain information about both, the electrochemical reactivity and the transport properties of a system [29]. In this work, the difference between the maximum cathodic and anodic potential peaks (ΔEp) in cyclic voltammograms obtained using the graphite-epoxy electrodes and Fe(CN)63-/4- as redox system model were compared. This practice was carried out to assess possible differences regarding mass transport or electron transport at the electrode surface of the unmodified and modified composites (figure 3).
The results indicated differences in the electrochemical response achieved using the unmodified and the modified electrodes. In addition, the different contents of benzoic acid influenced the electrochemical response. The GEC showed a ΔEp value of 66 ± 9 mV, which falls within the range for reversible processes (ΔEp = 59 ± 20 mV) [30]. In comparison this value, the voltammogram obtained from the composite modified with benzoic acid at 1 wt% (GEC-1 %) showed a higher value of ΔEp (123 ± 10 mV), typical of a quasi-reversible behavior. This probably results from the combination of an increase in the resistance of the composite (higher energy required for the redox process), and the negatively-charged nature of the surface after modification with benzoic acid, which reduces electrode kinetics [31].
Another interesting feature is the reduction of the residual current in the GEC-1 % profile, compared to that of the unmodified electrode; the latter shows a wider voltammogram shape. On the other hand, the electrode obtained using the composite with benzoic acid at 5 wt% (GEC-5 %) showed an almost linear profile in a wide potential window, with a well-defined slope, which is characteristic of a material with large resistance. Based on these results, further work was made using the GEC-1 %.
The reversibility of the electrochemical processes occurring at the electrode surface was also studied by the influence of the scan rate on the potential difference (figure 4).
As the scan rate increases, it can be seen that the GEC electrode shows an almost constant profile around 67 mV, within the 59 ± 20 mV range level. In contrast, for the GEC-1 % electrode, the observed values were well off the ones characteristic of a reversible system, confirming the quasi -reversible behavior. In addition, the ΔEp value increased with scan rate, which is probably due to the electrostatic repulsion effect of benzoic acid carboxylic groups on the surface of the electrode. That quasi -reversible electrochemical behavior suggests that although some structural reorganization is accompanying the redox step, the main molecular framework does not undergo fragmentation [30, 32]. This reorganization could be related to the electrostatic repulsion between the carboxylic groups of the modifier molecules on the surface of the electrode, which can affect the entry/exit of reagents and products.
Study of Electron Transfer Processes in Graphite-Epoxy Composite Electrodes Using Fe(CN)63-/4- Redox System
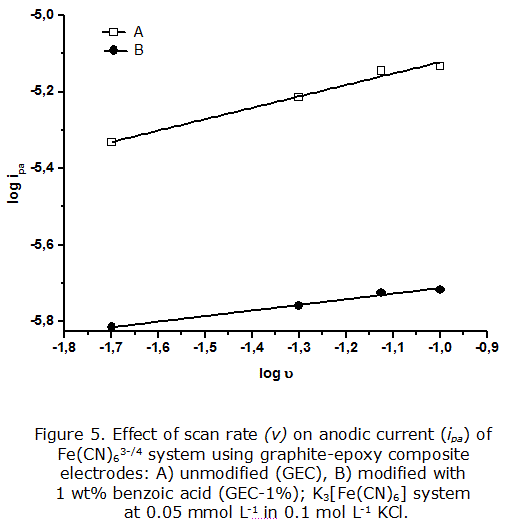
The result obtained with the GEC-1% indicates possible kinetic effects in the electrochemical response; thus, a further study was made to evaluate the variation of current ratio profiles with the scan rate (figure 5). The kinetic phenomena associated to electrode processes can be estimated by analyzing the ratio between the anodic (ipa) and cathodic (ipc) peak currents, which should be roughly equal for a reversible process without coupled reactions, while differences would be indicative of the kinetics affecting the electrode processes [32]. In addition, the dependence of ipa/ipc ratio with the scan rate allows determining the type of processes, and understanding the possible mechanisms of chemical reactions occurring on the electrode surface [30].
The slopes of the anodic process were calculated using the Randles-Sevcik equation [27] and the results for the GEC and the modified GEC-1 % electrodes were 0,50 ± 0,08 (r2 = 0,974 9) and 0,48 ± 0,05 (r2 = 0,983 1), respectively. These values suggest that the processes occurring at both electrodes are diffusion-controlled and electrochemically reversible (slope = 0,5), following a 'Nernstian' behavior characteristic of an ideal process under lineal diffusion control, within the experimental error and the timescale of the experiment [26].
It is interesting to note that although the slopes in both profiles are very similar, there is a difference regarding the absolute value of anodic peak currents. The higher currents obtained with the GEC electrode, compared to those using the GEC-1 %, indicate a fast kinetics of the electrochemical processes occurring at the unmodified electrode surface. On the other hand, the modified electrode surface, affected by the presence of benzoic acid, limits somehow the kinetics of the reactions. This result confirmed the existence of modifications on the electrode surface, but also pointed out to the necessity of further investigations of the possible mechanisms of the electrochemical processes.
With this purpose, the ipa/ipc ratios were also calculated using both electrodes at scan rates between 20 and 100 mV s-1. The ratios obtained were close to unity in both cases, being 0,98 ± 0,02 for GEC and 1,0 ± 0,1 for GEC-1 %. This behavior is representative of quasi-reversible systems, without associated kinetic phenomena, such as adsorption or chemical reactions.
Heterogeneous Electronic Rate Constant (ks)
In general, any electroanalytical technique requiring a mass transport limited current must be conducted under conditions of fast electron transfer. The use of voltammetry to probe reaction mechanisms depends particularly on suitable electrode kinetics to ensure fast electron transfer, which determines the shape and magnitude of voltammograms. Narrow and intense peaks yield better voltammograms, increasing the sensitivity of the electrochemical response. The key parameter in the kinetic process is the apparent rate constant (ks), which can also limit the range of useful scan rates to values where reactions can occur at the surface of the electrodes at a suitable rate [33].
The apparent rate constants (heterogeneous electron transfer rate constants, ks) of the redox processes occurring at the surface of both electrodes developed in this work were determined using the Nicholson method [34] and the ΔEp values from CV profiles (obtained under linear diffusion conditions). The calculations yielded ks values of (6 ± 2) X 10-4 m s-1 and (4 ± 1) X 10-4 m s-1, for the GEC and GEC-1% electrodes, respectively, at scan rates of 20-100 mV s-1 (figure 4). These values are in the same magnitude order to those previously reported for K3[Fe(CN)6] systems using glassy carbon ultra-polished (5 X 10-4 m s-1), carbon paste (4,3 X 10-4 m s-1), and highly oriented pyrolytic graphite (HOPG) edge plane (6 X 10-4 m s-1) electrodes [35].
As it can be seen, the electrochemical reactions occurring at both electrode surfaces fall well into the range for quasi -reversible processes, considering the limiting scan rates (3x10-3 v1/2 ≥ ks ≥ 2x10-7 v-1/2, [30]). The slightly higher ks value obtained in the case of GEC electrode suggests a more reversible behavior, compared to the value for GEC-1% This behavior is in agreement with the initial findings, based on the dependence of ΔEp with the scan rate, which is constant and near 59 mV for the GEC electrode while for the modified electrode, ΔEp shows higher values with increasing scan rates due to electrostatic repulsion effects of carboxylic groups on the surface of the electrode surface. The introduction of these benzoic acid groups in the graphite-epoxy composite can influence the electron transfer rate of processes occurring on the electrode surface due to a greater interaction between the negatively-charged carboxylic groups and the solvent layer (outer-sphere' mechanism, [32]). It has also been reported that e lectron transfer in redox processes can be restricted by the presence of oxygen functional groups at the surface of the electrodes, and the kinetics is reduced if the C/O ratio at the surface is decreased [36] .
Oxidation of Free DNA Bases and ssDNA (Calf Thymus)
The modifications introduced in the composite electrode developed in this work were carried out in order to have carboxylic groups available on the electrode surface that will allow further chemical reactions leading to peptide bonds (covalent immobilization of DNA probes) [1, 21, 22]. However, this type of transductors should also allow a high electron transfer at the electrode surface to ensure high signal-to-noise ratios of the analytical response [22]. Based on the results obtained in the electrochemical characterization, the GEC-1% electrode not only maintains a performance similar to the unmodified electrode, but also showed a high heterogeneous electron transfer rate constant (ks = (4 ± 1) X 10-4 m s-1) in the working conditions. Therefore, the next step was to test the electrochemical response of the modified electrode in the presence of purine bases and ssDNA, in order to assess their potential for development of DNA hybridization biosensors.
The purine bases, adenine and guanine, are involved in cellular energy transduction and signaling mediated by enzymatic oxidation reactions. These nitrogenous bases are reported to be the DNA electroactive indicators [1], and thus, electrochemical processes involved in purine base oxidation can be of crucial importance to assess label-free DNA hybridization in the development of biosensors.
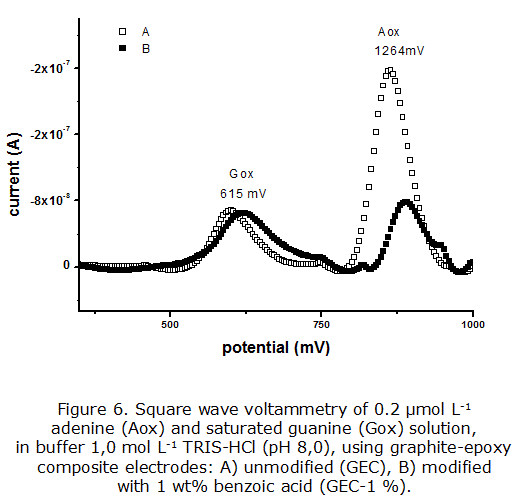
The oxidation signals of free DNA bases (adenine and guanine) were measured by square-wave voltammetry, using the two graphite-epoxy composite electrodes (figure 6).
As can be observed, the free DNA bases generated defined oxidation peaks in both graphite-epoxy electrodes, indicating that the electrochemical response was not affected by the presence of the modifier. Adenine was oxidized at 1 264 mV, while guanine oxidation occurs at 615 mV. These results correspond to the characteristic oxidation potentials on carbon electrode surfaces [1, 37], which can be used for electrochemical DNA detection. Furthermore, the GEC-1 % electrode response showed a lower residual current, in agreement with the results discussed for the Fe(CN)63-/4- redox system (figure 3), which can lead to achieving lower detection limits in the determination of biological species of interest.
The oxidation signal of ssDNA Calf Thymus was also determined using the composite electrodes developed in this work, at a concentration of 5 µg mL-1 in 1,0 mol L-1 TRIS-HCl buffer (pH 8,0). Similarly to the results obtained for the free bases, the guanine oxidation signal appears at 630 mV in both GCE surfaces (figure 7). Therefore, it can be inferred that not only the oxidation of the guanine base occurs in the same manner both as free base and as part of the DNA molecule, but also that the redox process occurs in a very similar way at the surface of both graphite-epoxy electrodes. The latter indicates that the modification of the composite with the introduction of carboxylic acids does not imply changes in the electrochemical response of the electrode for the oxidation of guanine.
These results show that the presence of carboxylic groups from modifications in the graphite-epoxy electrode using 1 % benzoic acid are not interfering in the electrochemical response of free and DNA bases, which is the desirable property for pursuing further chemical modifications through covalent immobilization of DNA probes. This immobilization step is an important requirement towards achieving highly sensitive and reliable detection of DNA virus using hybridization, which also avoids non-specific adsorption and improves selectivity of the method.
CONCLUSIONS
Benzoic acid was used as a new modifier in epoxy-graphite composites to introduce carboxylic groups at the electrode surface for immobilization of DNA probes. The electrochemical characterization of the modified electrode using 1wt% of benzoic acid showed a typical quasi-reversible behavior, without significant differences compared to the response using the non-modified composite. It can therefore be suggested as a suitable candidate for the development of the biosensors. In contrast, the composite prepared using higher content of modifier (GEC-5 %) showed an almost linear profile, characteristic of a material with large resistance.
Raman spectroscopy confirmed the presence of the carboxylic groups at the surface of the modified composite, while there were not significant alterations in the morphology of the surfaces of the electrodes, as evidenced by electronic microscopy. The ? Ep values obtained by cyclic voltammetry study using the GEC-1% were higher compared to the pristine GEC electrode, as a consequence of the negatively charged nature of the surface modified with carboxylic groups. In addition, the profiles of current with scan rate suggested that the processes occurring at both electrodes are diffusion-controlled, while the ipa / ipc ratios indicated that there is also a possibility of associated kinetic phenomena, such as adsorption or chemical reactions, affecting the electron transfer processes. The heterogeneous electron transfer rate constants were also determined, with values that fall into the range for quasi- reversible processes.
ACKNOWLEDGEMENTS
The authors are grateful to Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ), to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, project CAPES/MES 214/13) for financial support, and to the Van de Graff Laboratory of PUC-Rio for the microscopy analyses. Special thanks to Prof. Ricardo Q. Aucélio for critical reading of the manuscript and many helpful comments.
REFERENCES
1. Palecek, E.; F. Jelen. Electrochemistry of Nucleic Acids. In F.S. Emil Palecek and J. Wang eds. Perspectives in Bioanalysis. Elsevier, 2005, 1, p. 73-173.
2. Bojorge, N.; E. Alhadeff Graphite-Composites Alternatives for Electrochemical Biosensor. Edtion ed., 2011.
3. Cavalheiro, É., et al. "Bioelectroanalysis of pharmaceutical compounds", Bioanalytical Reviews, 2012, 4(1), 31-53.
4. Lian, Y., et al. "Lysozyme aptamer biosensor based on electron transfer from SWCNTs to SPQC-IDE", Sensors and Actuators B: Chemical, 2014, 199(0), 377-383.
5. Banks, C.E.; R.G. "Compton Edge plane pyrolytic graphite electrodes in electroanalysis: an overview". Anal Sci, Nov 2005, 21(11), 1263-8.
6. Banks, C.E., et al. "Electrocatalysis at graphite and carbon nanotube modified electrodes: edge-plane sites and tube ends are the reactive sites". Chemical Communications, 2005, (7), 829-841.
7. Wissler, M. "Graphite and carbon powders for electrochemical applications", Journal of Power Sources, 2006, 156(2), 142-150.
8. Kiryushov, V.N.; L.I. Skvortsova; T.P. Aleksandrova. "Electrochemical behavior of the system ferricyanide-ferrocyanide at a graphite-epoxy composite electrode", Journal of Analytical Chemistry , 2011, 66(5), 510-514.
9. Céspedes, F.; S. Alegret. "New materials for electrochemical sensing II. Rigid carbon–polymer biocomposites", TrAC Trends in Analytical Chemistry, 2000, 19(4), 276-285.
10. Apetrei, C., et al . "Advantages of the Biomimetic Nanostructured Films as an Immobilization Method vs. the Carbon Paste Classical Method", Catalysts, 2012, 2(4), 517-531.
11. Apetrei, C., et al . "Carbon Paste Electrodes Made from Different Carbonaceous Materials: Application in the Study of Antioxidants", Sensors, 2011, 11(2), 1328-1344.
12. Lucarelli, F., et al . "Carbon and gold electrodes as electrochemical transducers for DNA hybridisation sensors", Biosensors and Bioelectronics, 2004, 19(6), 515-530.
13. Banks, C.E.; R.G. Compton. "New electrodes for old: from carbon nanotubes to edge plane pyrolytic graphite", Analyst, Jan 2006, 131(1), 15-21.
14. Bagotsky, V.S. "Structure and Properties of Surface Layers". In Fundamentals of Electrochemistry. John Wiley & Sons, Inc., 2005, p. 147-180.
15. Hsu, W.; W. Holtje; J. Barkley. "Percolation phenomena in polymer/carbon composites", Journal of Materials Science Letters, 1988, 7(5), 459-462.
16. Kneten, K.R.; R.L. McCreery. "Effects of redox system structure on electron-transfer kinetics at ordered graphite and glassy carbon electrodes", Analytical Chemistry, 1992, 64(21), 2518-2524.
17. Longjam, N., et al. "A Brief Review on Diagnosis of Foot-and-Mouth Disease of Livestock: Conventional to Molecular Tools", Veterinary Medicine International, 2011, 905768.
18. Palecek, E. "NUCLEIC ACIDS | Electrochemical Methods". In P.W.T. Poole ed. Encyclopedia of Analytical Science (Second Edition). Oxford: Elsevier, 2005, p. 399-408.
19. vancara, I., et al. "Carbon Paste Electrodes in Facts, Numbers, and Notes: A Review on the Occasion of the 50-Years Jubilee of Carbon Paste in Electrochemistry and Electroanalysis", Electroanalysis, 2009, 21(1), 7-28.
20. Walsh, M.K., X. Wang; B.C. Weimer. "Optimizing the immobilization of single-stranded DNA onto glass beads", J Biochem Biophys Methods, 2001, 47(3), 221-31.
21. Millan, K.M.; S.R. Mikkelsen. "Sequence-selective biosensor for DNA based on electroactive hybridization indicators", Anal Chem, 1993, 65(17), 2317-23.
22. Schulein, J., et al. "Solid composite electrodes for DNA enrichment and detection", Talanta, 2002, 56(5), 875-85.
23. Pividori, M.I., et al. "Bioaffinity platforms based on carbon-polymer biocomposites for electrochemical biosensing", Thin Solid Films, 2007, 516(2–4), 284-292.
24. Ramirez-Garcia, S., et al. "Carbon composite electrodes: surface and electrochemical properties", Analyst, 2002, 127(11), 1512-1519.
25. Colthup, N.B., L.H. Daly; S.E. Wiberley. CHAPTER 8-AROMATIC AND HETEROAROMATIC RINGS. In N.B. Colthup, L.H. Daly, and S.E. Wiberley eds. Introduction to Infrared and Raman Spectroscopy (Third Edition). San Diego: Academic Press, 1990, p. 261-288.
26. Colthup, N.B., L.H. Daly,; S.E. Wiberley. CHAPTER 9-CARBONYL COMPOUNDS. In N.B. Colthup, L.H. Daly, and S.E. Wiberley eds. Introduction to Infrared and Raman Spectroscopy (Third Edition). San Diego: Academic Press, 1990, p. 289-325.
27. Colthup, N.B., L.H. Daly; S.E. Wiberley. CHAPTER 11-AMINES, C=N, AND N=O COMPOUNDS. In N.B. Colthup, L.H. Daly, and S.E. Wiberley eds. Introduction to Infrared and Raman Spectroscopy (Third Edition). San Diego: Academic Press, 1990, p. 339-354.
28. Colthup, N.B., L.H. Daly; S.E. Wiberley. CHAPTER 12-COMPOUNDS CONTAINING BORON, SILICON, PHOSPHORUS, SULFUR, OR HALOGEN. In N.B. Colthup, L.H. Daly, and S.E. Wiberley eds. Introduction to Infrared and Raman Spectroscopy (Third Edition). San Diego: Academic Press, 1990, p. 355-385.
29. Hogan, C.F., et al. "Facile Analysis of EC Cyclic Voltammograms", Analytical Chemistry, 2004, 76(8), 2256-2260.
30. Holze, R. Piero Zanello. "Inorganic electrochemistry: theory, practice and applications", Journal of Solid State Electrochemistry, 2006, 10(7), 512-513.
31. Barsan, M.M., et al. "Development and characterization of a new conducting carbon composite electrode", Analytica Chimica Acta, 2009, 635(1), 71-78.
32. Bard, A.J. Electrochemical Methods; Fundamentals and Applications . 2nd ed. 2001: United States of Americ.
33. Bond, A.M. Laboratory techniques in electroanalytical chemistry: P.T. Kissinger and W.R. Heineman (Editors); Marcel Dekker Inc., New York, Basel; Hong Kong, 1996, xx+986 pp., ISBN 07-82477-94457-1.
34. Nicholson, R.S. "Theory and Application of Cyclic Voltammetry for Measurement of Electrode Reaction Kinetics", Analytical Chemistry, 1965, 37(11), 1351-1355.
35. McCreery, R.L. "Advanced carbon electrode materials for molecular electrochemistry", Chem Rev, 2008, 108(7), 2646-87.
36. Chen, P.; R.L. McCreery. "Control of Electron Transfer Kinetics at Glassy Carbon Electrodes by Specific Surface Modification", Analytical Chemistry, 1996, 68(22), 3958-3965.
Recibido: 20/03/2016
Aceptado: 2/09/2016
Aurora Pérez-Gramatges, Departamento de Química, Pontifícia Universidade Católica do Rio de Janeiro, Brasil, aurora@puc-rio.br