Introduction
Theories concerning the origin and evolution of life must necessarily provide a chemical-physical reason for complexation at all biological levels. The traditional “survival of the fittest” paradigm at the macroscopic level or the “chemical stability” paradigm at the microscopic molecular level are both deficient when it comes to explaining complexation. A more prudent approach would be to consider only what is well established concerning complexation in chemical-physical systems; that the ordering of material out-of-equilibrium occurs exclusively as a response to the dissipation of an externally imposed generalized thermodynamic potential, or in other words, complexation is concomitant with an increase in global entropy production of the system interacting with its environment. Such ordering of material was given the name ``dissipative structuring'' by Ilya Prigogine.1
The thermodynamic dissipation theory for the origin and evolution of life 2-5 suggests that every incremental complexation in the history of biological evolution; from the formation of complex prebiotic molecules from simpler precursor molecules, their associations into cellular and multi-cellular organisms, to the hierarchical coupling of biotic with abiotic processes of today's biosphere, must necessarily have coincided with an incremental increase in the dissipation of some externally imposed generalized thermodynamic potential.1,6,7 We have identified 2 the important thermodynamic potential driving complexation at life's origin as the long-wavelength UVC and UVB solar photon potential arriving at Earth’s surface during the Archean (see fig. 1) since these wavelengths have sufficient free energy to break and reform covalent bonds of carbon-based molecules, but not enough energy to disassociate these molecules.2,4 Such wavelengths reached Earth's surface from before the origin of life and lasting for at least 1,000 million years until the end of the Archean 8,9 (fig. 1), at which time oxygenic photosynthesis overwhelmed natural oxygen sinks, allowing the formation of an atmospheric ozone layer.

Fig. 1 The wavelengths of maximum absorption of many of the fundamental molecules of life coincide with an atmospheric window predicted in the UVC and UVB at the time of the origin of life at 3.85 Ga (black) and until at least 2.9 Ga (red). At 2.2 Ga (yellow), UVC light at Earth's surface had been extinguished by the buildup of oxygen and ozone. The green curve corresponds to the present surface spectrum. Energy flux for the sun at the zenith. Adapted with permission from Michaelian and Simeonov 9
Elsewhere 5, we have analyzed the autocatalytic photochemical production and proliferation of adenine and single strand DNA at the ocean surface under the UVC+UVB Archean photon potential. In this paper, after describing the general chemical and physical characteristics of fatty acids, we analyze the autocatalytic photochemical production and proliferation of fatty acids and their vesicles at the ocean surface, under this same photon potential and give non-equilibrium thermodynamic justification for the association through encapsulation of nucleic acids and other fundamental molecules within these first vesicles.
Theoretical foundations
Characteristics of Fatty Acids
Natural fatty acids contain a carbon plus hydroxy plus oxygen (carboxyl) head group and a hydrocarbon tail of from 4 to 40 carbon atoms 10 which may be saturated with hydrogen or partially unsaturated (fig. 2). Fatty acids, fatty alcohols and fatty acid glycerol esters are generally considered to be more relevant to vesicles of early life than the phospholipids composing the cellular wall of most present day organisms because phospholipid biosynthesis is very different between bacteria and archaea, suggesting that their common ancestor was devoid of phospholipid membranes (although this view has been challenged).11,12 Also, fatty acids are simpler single chain molecules that are more easily produced through abiotic heat activated Fischer-Tropsch or photochemical polymerization of smaller chain hydrocarbons such as ethylene (C2H4). Ethylene itself, and other small chain hydrocarbons, can be derived from the reduction of CO2 or CO employing water as the electron donor and either chemically or photochemically catalyzed. Ethylene can also be produced by the UV photolysis of methane which appears to be an important mode of hydrocarbon production in the atmosphere of Titan.13
Fatty acids are conserved as components of the cell walls in organisms from all three domains of life. On today's ocean surface, and in the Archean fossil record, there is an abundance of even number carbon fatty acids over odd number and this could be explained if indeed such fatty acids are formed by polymerization (telomerization) of ethylene. In particular, there is a predominance of 16 and 18 carbon atom fatty acids in the whole available Precambrian fossil record 14,15 and, in fact, in today's organisms, and in the aerosols obtained above today's ocean surface.16,17
In general, the longer the hydrocarbon tail and the greater the degree of saturation, the greater the melting temperature; the degree of saturation usually having the greater influence because double bonds introduce a curvature into the hydrocarbon tail preventing them from bonding strongly with neighbors. For example, lipid desaturation by the enzyme desaturase is a key factor in the protection of the photosynthetic apparatus from low-temperature and high-light stresses, as well as being an effective tool for manipulating membrane microviscosity.18 For example, saturated myristic acid (C14:0) has a melting temperature of 55 °C 19 while linoleic acid with two unsaturated bonds (18:2) has a melting temperature of only -5 °C. However, if the unsaturation occurs as conjugated double bonds in the trans configuration near the tail end of the hydrocarbon, such as in parinaric acid (C18:4n-3, see figure 2), then unsaturation is not greatly destabilizing. Parinaric acid has a high melting temperature of 85 °C.
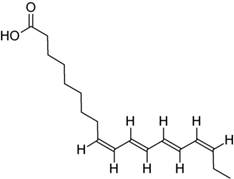
Fig. 2 Parinaric acid is a conjugated fatty acid of 18 carbon atoms having a melting temperature of (85-86 °C), roughly the temperature of the Archean ocean surface at the origin of life
The 4 conjugated carbon double bonds lead to delocalized electrons, giving it strong absorption over UVB-UVA region of 310 to 340 nm (fig. 1). Conjugated fatty acids have conical intersections allowing for rapid dissipation to heat of the photon-induced electronic excited state energy, making them excellent candidates for Archean dissipative structures. UVC-induced cross linking with other fatty acids at the site of a double bond would convert this conjugated parinaric acid into conjugated linolenic acid (C18:3n-3) with absorption in the UVC at 269 nm (see fig. 1).
Direct photoionization (peroxidation) under UVC light (254 nm) of fatty acids can lead to conjugation of the carbon bonds of the acyl tail.20 The steps involved in the conjugation process are i) photon-induced deprotonation, ii) bond migration to give a conjugated diene or triene. The greater the degree of unsaturation, the greater the probability of it undergoing peroxidation.20 The same UVC light, however, can also cause cross linking between adjacent acyl tails at the site of a double bond which would reduce the average conjugation number. 21 Under the constant UVC flux existing at the origin of life, a stationary state of particular conjugation numbers would therefore have arisen. Under the UV light of the Archean, and according to the postulates of thermodynamic dissipative structuring 5, the conjugation numbers of fatty acids in this stationary state would be either 2, 3, or 4 since these give rise to absorption at 234, 269, and 310-340 nm respectively, which all lie within the Archean surface solar spectrum (fig. 1).
In the following, we will concentrate on vesicles formed from mainly conjugated fatty acids of 18 carbons for the following reasons; 1) fatty acids of 18 (and 16) carbon atoms are predominant in sediments as early as 3.4 Ga and throughout the Archean 14,15 and indeed are the most prevalent sizes in the three domains of today's organisms 15, 2) vesicles containing fatty acids with three conjugated double bonds required for light absorption around the peak in the Archean UVC spectrum at 260 nm (see below) must contain fatty acids of at least 18 carbon atoms in order to be stable at high surface temperatures of the early Archean of around 85 °C, and 3) apart from the telomerization of ethylene, there is a rather simple photochemical route to the production of 18 carbon atom fatty acids from the UVC induced polymerization of highly surface active 9 carbon nonanoic acid C9H18O2 which can be produced through UVC photochemical dissipative structuring of CO, CO2 saturated water.22 Our postulate for the first fatty acids, with chain lengths of 18 carbon atoms and unsaturated and conjugated, is distinct to that previously asserted as being the most plausible prebiotic fatty acids for the first vesicles -- of saturated short chain (( 10 carbon atoms) lengths (such as myristoleic acid and its alcohol) because these are the most likely to arise from a Fischer-Tropsch type chemistry.23 This conclusion, however, ignores UVC induced surface chemistry for synthesis, polymerization, and cross linking (see below) of fatty acids, and the fact that a greater stability of the vesicles is required for the high surface temperatures of the early Archean. Inclusion of even longer chain lengths (≥ 20 carbon atoms), for which there is some evidence in the early fossil record (see 14,24 and references therein) increases still the stability with respect to pH range 25 and temperature.
Photochemical Synthesis of Fatty Acids
A plausible alternative to ocean floor Fischer-Tropschsynthesis of the hydrocarbon chains is ocean surface photochemical synthesis. The fatty acid hydrocarbon tails can be built up from the sequential photon-induced polymerization of an initiator molecule such as ethylene. Photopolymerization occurs through photon-induced sensitization in the UV region of the spectrum. It generally occurs through direct photon-induced cleavage of the initiator molecule producing a free-radical which subsequently attacks the carbon-carbon double bonds of an existing polymer, thus initiating further polymerization.26 Polymerization rates are more than two orders of magnitude larger at wavelengths of 254 nm (UVC) than at 365 nm (UVA).26 Oxygen acts as a strong inhibitor to polymerization by rapidly reacting with the radical to form a peroxy-based radical which does not promote polymerization.26 Such an oxidation reaction following hydrolysis is the origin of the carboxyl head group of the fatty acids. The presence of oxygen and the lack of surface UVC light today (Fig. 1) means that hydrocarbon chain polymerization at today's ocean surface 16,17 is only a mere ghost of what it probably was at the origin of life.
Indications that ultraviolet light may have played an important role in the formation of hydrocarbons have come from different experiments. Since the early 1960’s it was known that irradiation with UVC light of CO2 saturated water containing ferrous salts results in the production of formic acid and formaldehyde.27 Later, C1 hydrocarbons such as methane, methanol, ethanol and formaldehyde, and formic acid were produced from CO2 and H2O in a photoelectrochemical reactor consisting of a TiO2-coated electrode suspended in CO2 saturated aqueous solution and subjected to UVC light.28,29 Klein and Pilpel (1973) 30 demonstrated that short chain amphiphiles can be synthesized by a light-dependent reaction from common simple hydrocarbons and an aqueous film of poly aromatic hydrocarbons (PAHs), ubiquous throughout the universe 9 acting as photosensitizers. It has also been shown that imidazole or porphoryins (pyridine) under UVC light can act as a catalyst for the reduction of CO2 to n=2 and longer chain hydrocarbons.31
Later, Varghese et al.32 and also Roy et al.33 identified longer chain hydrocarbons in similar mixtures of CO2 and water irradiated with UV light. In general, and in contrast to the Fischer-Tropsch process, the higher the temperature, the higher the pressure, or the greater the ratio of CO2 to H2O, the larger the quantity of longer chain hydrocarbons obtained.
Another plausible route to the formation of fatty acids under a UV environment has been observed Botta et al.33 By impinging UV and visible light (185 to 2000 nm) from an xenon lamp on formaldehyde (a product of UVC light on HCN and water) using ZnO and TiO2 as photocatalysts, Botta et al. found that this resulted in fatty acids of chains of from 2 to 5 carbon atoms as well a host of other fundamental molecules of life such as nucleic acids, and amino acids, as well as glycolaldehyde, a precursor to sugars through the formose-reaction. The mechanism of formation of these fatty acids suggested by Botta et al. follows that proposed earlier by Eschenmoser 34 entailing, first the generation of HCN from formaldehyde followed by its oligomerization to diaminomaleonitrile DAMN (detected in their reaction mixture), hydrolysis and successive electron transfer processes. The formation of DAMN from HCN requires the absorption of photons in the long wavelength UVC region and is exactly the same process which we have described as the photochemical dissipative structuring of the purines from UVC light on HCN and water 5 first observed by Ferris and Orgel.35
The ZnO and TiO2 photocatalysts are suspected of providing electron transfer reactions after absorbing a UV photon. Although these catalysts are rather common they are not very soluble in water and would have tended to aggregate and fall to the ocean floor. There are, however, other electron donors which would have been more available on the Archean ocean surface for the reduction of CO₂. For example, it is known that ferrous ions can reduce water to hydrogen upon excitation under ultraviolet light with high yield. This released hydrogen and more reactive intermediates could have induced the reduction of CO2. There is ample evidence that the Banded Iron Formations were derived from the photooxidation and not the oxidation of ferrous ions in Archean oceans and thus this electron donor would have been widely available on the early ocean surface.
Another electron donor which could have equally catalyzed the reduction is the amino acid histidine or its intermediate known as amino-imidazole-carbon-nitrile (AICN) and those amino acids with a negatively charged R-group; Glu monosodium salt (Glu(Na), and Asp potassium salt (Asp(K). These, we have argued 36, were among the first amino acids to have formed complexes with RNA and DNA in order to increase global UVC dissipation as evidenced by their charge transfer absorption spectrum peaking at 270 nm 37 falling in the middle of the Archean UVC atmospheric window, and the chemical affinity of these amino acids to their codons and/or anticodons.38
Here we propose that the long chain, n ≥18, hydrocarbons which became incorporated into the fatty acid vesicles of early life were produced by such surface-sensitized UVC-induced polymerization processes acting on shorter chain hydrocarbons such as ethylene produced through UVC light on CO2 saturated surface water or formaldehyde and HCN saturated water at high temperature (> 85 °C) and possibly a higher than present atmospheric pressure (up to 2 bar).39 In contradistinction to the Fischer-Tropsch polymerizations operating at very high temperatures and pressures (perhaps at deep sea hydrothermal vents) such photochemical polymerization could only have occurred on the ocean surface where concentrations of fatty acids would have been sufficiently high enough to allow access to the reactive triplet state, and where UVC light was most intense. The experiments of Rossignol et al.22 suggest that catalysts are not needed for such surface polymerizations, however, the existence of catalyst transition metals for the reduction of CO2 such as Fe, Mn, Co, Ni, Cu, or Zn (particularly Fe) would have been available at the Archean ocean surface 40 and these would have undoubtedly increased the rates of hydrocarbon chain growth. There is again also the possibility that an imidazole, such as the amino acid histidine or its intermediate AICN formed in the process of UVC microscopic dissipative structuring of the purines from HCN 5, could have been the reduction agent. It is well known that imidazole is a strong catalyst that can act as either an acid or a base and furthermore, direct evidence has been obtained for its catalytic function in the formation of phosphatic acids from simpler compounds.41
Dissipative Structuring
Saturated fatty acids do not absorb in the UV except for disassociation at < 180 nm and the carboxyl head group which absorbs with a small peak at 207 nm.42 Under the Archean UVC flux, photon-induced deprotonation could lead to a double carbon bond forming at any point on the hydrocarbon tail. A single double carbon bond in the tail will absorb at 210 nm. Migration of the double bonds along the tail is known to occur 20 leading to conjugated bonds. Two double bonds in a conjugated configuration (diene) will lead to strong absorption at 230 nm, those having three in a conjugated configuration (triene) will lead to absorption at 269 nm, while those with 4 (tetraene, see fig. 2) will lead to absorption at 310-340 nm. All of these latter three absorptions lie within the important UVC spectrum arriving at Earth’s surface during the Archean (see fig. 1).
Hydrocarbons having conjugated dienes, trienes, or tetraenes almost always have conical intersections 43 allowing rapid dissipation of the electronic excited state energy. Reaching the conical intersection when in the electronic excited state involves a twisting about two C=C bonds and decreasing one of the C-C-C angles producing a kink in the carbon backbone.43
Therefore, the same photons that structured the material are the ones that will be dissipated efficiently by the final photochemical product. These photochemically synthesized and conjugated fatty acid structures can thus be identified as microscopic dissipative structures. 5 The steps involved in the dissipative structuring are thus the following i) UVC-induced reduction of CO2 and CO in water saturated with these to form ethylene, ii) UVC-induced telomerization of ethylene to form long hydrocarbon tails of an even number of carbon atoms, iii) oxidation and hydrolysis events to stop the growing of the chain and form the carboxyl group, iv) UVC-induced deprotonation of the tails to form a double bond, v) double bond migration to give a conjugated diene, triene, or tetraene with a conical intersection (Fig. 3). These dissipatively structured fatty acids are, of course, robust to further photochemical reactions because of the sub picosecond decay times of their electronic excited states, due to their conical intersections, which is too fast to allow appreciable further chemical transformation.
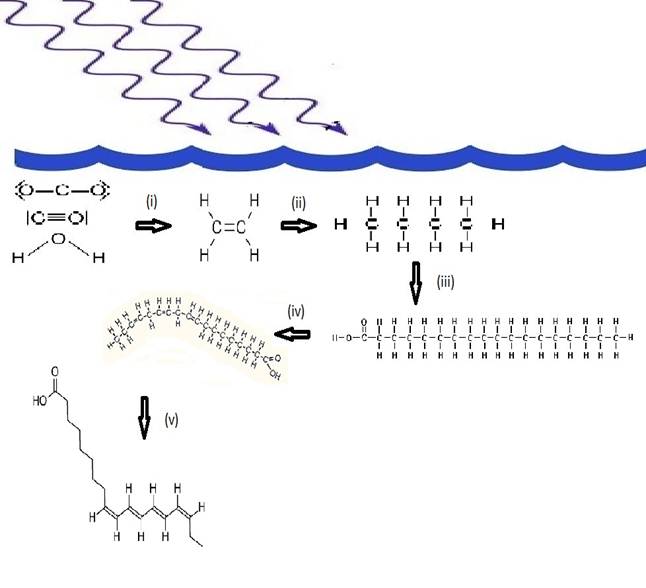
Fig. 3 The photochemical dissipative structuring of (-parinaric acid under UVC+UVB light. i) UVC-induced reduction of CO2 and CO in water to form ethylene, ii) UVC-induced telomerization of ethylene to form long hydrocarbon tails, iii) oxidation and hydrolysis to stop the growing of the chain and form the carboxyl group, iv) UVC-induced deprotonation of the tails to form double bonds, v) double bond migration to a conjugated tetraene (-parinaric acid (C18:4n-3) with a conical intersection
We have identified a similar dissipative structuring route for the synthesis of the nucleobase adenine from HCN under the same UVC photon potential.5 In this case, the steps involve i) a thermal exothermic process leading from 4HCN molecules to the stable HCN tetramer cis-DAMN, ii) a UVC-induced cis to trans transformation through rotation around a carbon-carbon double bond, iii) a UV-B induced tautomerization, and iv) a UVC-induced ring closure. We believe that similar dissipative structuring routes involving different UVC- or UVB-induced processes exist for all of the fundamental molecules of life and this explains their strong absorption in the UVC (fig. 1).
Vesicle Formation
The most notable problems associated with the proposal of fatty acids conforming the first protocell walls are that; 1) vesicles of these form only in a narrow alkaline pH range, 2) they have a tendency to aggregate and crystallize in high salt conditions (salt flocculation), leading some to conclude that life must have started in a fresh water environment 44, and 3) a critical vesiculation concentration (CVC) of the fatty acids is required for spontaneous formation of vesicles and this concentration is considerably higher than that required for phospholipid vesicle formation.
Recent experiments have shown, however, that the pH range of stability and resistance to salt flocculation of fatty acid vesicles can be greatly increased through covalent cross linking among neighboring chains, which can be induced by either UVC light 21, or by moderate temperatures (~50 - 70 °C), or by simple aging.21,45 The same heat treatment applied to non-conjugated unsaturated fatty acids does not appear to have an as large effect on the increase in range of pH stability or stability against salt flocculation. Under all conditions, vesicles of heat treated conjugated linoleic acid showed better stability than non-conjugated linoleic acid.45 At the high surface temperatures and large UVC flux of the early Archean, considerable cross-linking between fatty acids could be expected, and conjugated fatty acids would thus form robust vesicles under the high temperatures, low pH, and high salt conditions of the Archean ocean surface. Furthermore, mixtures of fatty acids with fatty alcohols and fatty acid glycerol esters of differing lengths provide surprising resistance to salt flocculation.23 Esterification of fatty acids which helps with resistance to salt flocculation (see section “Characteristics of Fatty Acids”) can be induced by UVC activated transition metal catalysts to provide electrons for the reduction of the fatty acid.46 UV radiation also leads to the formation of aldehydes from fatty acids.22
There is also another more likely possibility giving rise to stability over a greater range of pH. Rather than the end to end model of the hydrophobic tails, the observed structure appears to be more like side-by-side 47 with overlap of respective double bonds (see figure 4). This would allow for a natural aging, or temperature- or UVC induced-, cross linking among fatty acids, which makes these vesicles stable over a very wide range of pH values, from 2 to 14.47
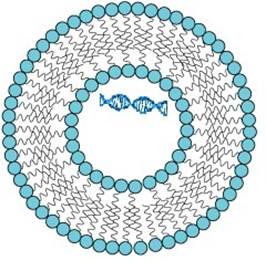
Fig. 4 A vesicle made from variously conjugated (n≥18) fatty acids, e.g., (-parinaric (C18:4n-3) and linolenic (C18:3n-3), with an encapsulated DNA oligo. The inner and outer layers of the hydrocarbon tails have a side-by-side (rather than end-to-end) orientation which facilitates UVC-induced cross-linking, giving greater stability at the mild acid pH values of the Archean ocean. The result is an efficient photon dissipating system in the UVC+UVB+UVA regions of the Archean spectrum (fig. 1)
Results and discussion
We have described plausible photochemical routes to the dissipative structuring of the earliest Archean long chain fatty acids and their vesicles from CO2 and CO saturated water under the UVC light prevailing at Earth's surface at the origin of life and throughout the Archean. Ethylene would be a common product synthesized by this UVC light incident on the surface water and these would polymerize (telomerize) under the same light into longer chain hydrocarbons of mainly an even number of carbon atoms. These would stop growing after an oxidation or hydrolysis event. They would then become conjugated through the dissipative structuring under the UVC light giving rise to conjugated parinaric acid (C18:4n-3) and linolenic acid (C18:3n-3). Because of the high surface temperatures (~85 °C), only long chain (≥ 18 carbon atoms) with conjugation near the end of the tail (n-3) would bind together to form vesicles. Cross-linking would occur at the overlap of the inner and outer wall hydrocarbon tails at the site of a double bond, making the vesicles stable over somewhat acidic pH ranges (6.0-6.5) and high salt conditions of the Archean sea surface.
Many of the fatty acids would have their own conical intersection allowing the rapid dissipation of the photon-induced electronic excitation energy. These vesicles would then be performing the non-equilibrium thermodynamic function of dissipating into heat the prevailing UVC+UVB photon flux reaching Earth’s surface. This was the initial driving force for complexation of material, from synthesis, to proliferation and evolution, all through dissipative structuring. For example, chemical affinity of the fatty acids, mediated through divalent cations, to RNA and DNA, or with the carotenoids, would provide for greater dissipation, especially for those fatty acids which did not have their own conical intersections. Resonant energy transfer would allow the electronic excitation energy of the fatty acids to be dissipated through the conical intersection of the nucleic acid or the carotenoids. This scenario provides a physical-chemical basis for the origin, proliferation, and evolution of the fatty acids in conjunction with other fundamental molecules of life.
Conclusions
We have presented evidence to suggest that the fatty acids incorporated into the first protocells of life may have been pigments dissipatively structured from CO or CO2 saturated water under the long wavelength UVC region of the solar spectrum which penetrated to the surface of the Earth during the Archean. Similar microscopic dissipative structuring has also been suggested for the prebiotic formation and proliferation of the nucleobases and these results are consistent with the thermodynamic dissipation theory for the origin and evolution of life














