Entre las acciones a desarrollar se encuentra la revisión de los derechos de titulares en los territorios de interés. Es necesario conocer si existen titulares que poseen derechos y en qué países; de aquí la importancia de que se realice un análisis exhaustivo de estado legal de las patentes relacionadas con los medicamentos de interés. Para esto es fundamental la revisión de bases de datos de patentes, los cuales se muestran en el siguiente cuadro:
Cuadro. Bases de datos de patentes
Bases de datos de patentes
Sitio web
]]> Bases de datos de patentes de oficinas nacionales
y regionales de Propiedad Industrial.
Guía de Oficinas de Propiedad Intelectual
http://www.wipo.int/directory/es/urls.jsp PatentScope (OMPI)
Esp@cenet (Oficina Europea de Patentes)
http//patentscope.wipo.int/search/en/search.jsf Base de datos de la Oficina de Patentes y Marcas de USPTO http://www.uspto.gov/patents/process/search/ DepatisNet https://depatisnet.dpma.de Medicine Patent Pool
Base de datos de patentes de medicamentos antirretrovirales
http://www.medicinespatentpool.org/datos-sobre-patentes/estado-de-patentes-de-antirretrovirales/?lang=es WIPO Re: Search ]]> Base de datos de patentes de enfermedades tropicales desatendidas, paludismo y tuberculosis
http://www.wipo.int/research/en/search/
- ]]>
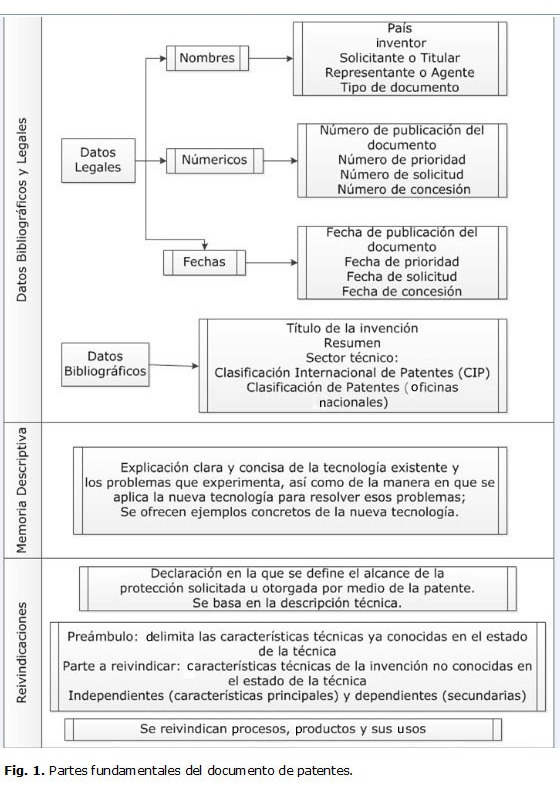
El documento de patente.
]]>
]]>
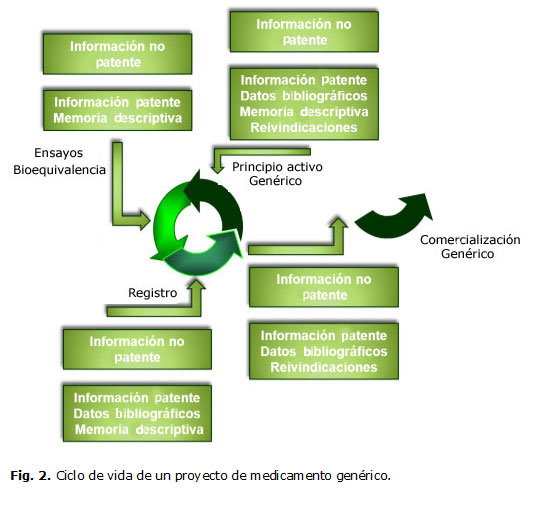
En el caso de las patentes farmacéuticas, la información técnica, es de gran utilidad en los diferentes pasos del ciclo de vida de un proyecto de I+D+i. Esto se relaciona con la información que contienen y que puede incluir, entre otros, un procedimiento para la obtención de un principio activo, de un compuesto farmacéutico, así como las diferentes formas farmacéuticas (tabla 1). Esta información es de suma importancia para los investigadores y productores de medicamentos genéricos.
- Guías o metodologías para la búsqueda de la información contenida en los documentos de patentes.
]]> -Guía de la OMPI para la utilización de la información de patentes.22-PATENTSCOPE Búsqueda y CLIR.23-Propiedad Intelectual: Guía de buenas prácticas.24-Las patentes: fuente de información tecnológica.25-Guía de buenas prácticas para la búsqueda de información en patentes.26 -Manual didáctico sobre patentes.27
Estos documentos de gran utilidad permiten realizar la búsqueda en cualquier campo temático y han sido elaborados, entre otros, por organismos internacionales, universidades y firmas relacionadas con estos temas.
- Metodologías para la recuperación de la información de patentes de medicamentos.
Por la importancia que tiene el tema de las patentes de los medicamentos y, en especial, aquellas que reivindican productos o procesos de obtención de fármacos esenciales y las correspondientes a las denominadas enfermedades olvidadas, algunos organismos internacionales, como es el caso de la OMS, el PNUD y la OMPI, han dedicado serios esfuerzos a elaborar metodologías y bases de datos para realizar la búsqueda de la información de patentes relacionadas con estos documentos.28
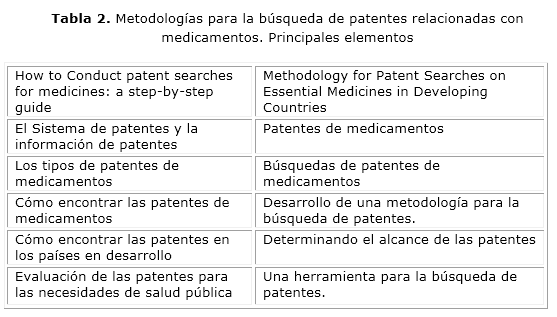
Entre las guías y/o metodologías relacionadas con la búsqueda de patentes farmacéuticas, se destacan:
-How to Conduct Patent Searches for Medicines: a Step-By-Step Guide, elaborada bajo el auspicio de la OMS, por Tahir Amin.29-Methodology for Patent Searches on Essential Medicines in Developing Countries,30 editada bajo el auspicio del PNUD y escrita por Barbara Milani y Cecilia Ohh.
Al analizar y comparar los referidos documentos, se puede observar que en ambos existe una gran similitud entre los temas tratados, así como complementariedad en otros (tabla 2) y, por consiguiente, enriquecen la información al respecto.
]]>
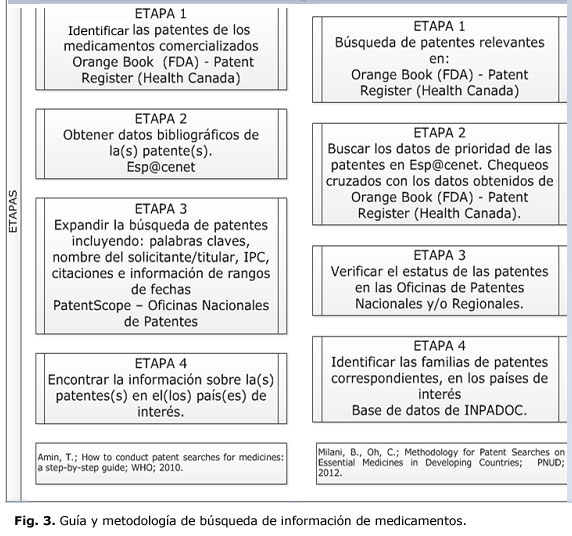
Analizando las diferentes etapas reportadas en ambos documentos (Fig. 3) se puede observar su semejanza. Es importante señalar que la génesis metodológica de ambas metodología sustenta fundamentalmente los problemas que presenta la búsqueda de patentes de medicamentos en forma general, específicamente en los países en desarrollo. Se observa que en ambas metodologías se dedica especial atención a la identificación de las patentes de medicamentos, mediante la búsqueda por compuestos químicos que pueden ser realizadas utilizando diversos métodos.
Ambos documentos plantean las limitaciones de las metodologías que describen y señalan que no contemplan el análisis de las patentes, especialmente de sus reivindicaciones, por lo que no se puede considerar que con su aplicación se da respuesta a todas las necesidades de los usuarios (investigadores, productores u otros).
Se plantea por Amin28 y por Milani29 que dos características de los sistemas de patentes actuales inciden negativamente en la recuperación de patentes de medicamentos: la complejidad técnica relacionada con el patentamiento de los productos farmacéuticos y la falta de capacidad institucional para la gerencia de los sistemas de patentes en los países en desarrollo, y señalan que inciden negativamente en los resultados de la búsqueda lo siguiente:
-La falta de referencia a la denominación común internacional (DCI)i.-La información no se encuentra actualizada o es inexacta.
-Un medicamento puede protegerse por más de una patentej.
-La información no es fácilmente asequible en la oficina de patente nacional.
]]> -No todas las oficinas nacionales de propiedad industrial de los países en desarrollo publican a los 18 meses de la fecha de prioridad (fecha de la solicitud inicial de una patente) las invenciones.-Existe un rango de 18 meses a partir de la fecha de prioridad en que no se publica el documento, por lo que no se cuenta con esa información.-Las solicitudes de patentes no entran en la fase nacional en otras oficinas nacionales, por lo menos hasta el año de la fecha de prioridad. En el caso de que se presenten mediante el Tratado de Cooperación y Patentes (PCT) de la OMPI, no se conocerán los países (pertenecientes al PCT) en que va a entrar en fase nacional hasta los 30 meses de la fecha de prioridad.
Lo anterior implica que va a existir en algunos casos información a la que no se tendrá acceso en el momento de la búsqueda inicial, por lo cual se le debe dar seguimiento a la búsqueda de información de patentes de forma periódica y durante todo el ciclo de vida de los proyectos.
- Metodologías o normas relacionadas con la generación de conocimiento para estudios de vigilancia o inteligencia (tecnológica y/o comercial).
Las metodologías de búsqueda de patentes son muy importantes; pero para dar solución a las problemáticas o necesidades de los usuarios (por ejemplo, investigadores, productores y comercializadores) deben ser un elemento a integrar en una metodología general que comience con la identificación de las necesidades de los usuarios, establezca el objeto y el objetivo del estudio, así como una estrategia de búsqueda integral. A partir de la estrategia establecida es que se debe incorporar una metodología para la búsqueda de la información de patentes y después del procesamiento de la información recuperada y su análisis (incluyendo en los casos que sea necesario el análisis del contenido de los requerimientos), generar un nuevo conocimiento que permita al usuario dar respuestas a las necesidad o problemática identificadas al inicio.
]]> La literatura reporta diferentes metodologías31-38 o normas, relacionadas con la vigilancia tecnológica, la inteligencia (tecnológica o competitiva)39,40 y la generación de conocimientos,41 que serán de utilidad al combinarse con elementos de las metodologías de búsqueda de documentos de patentes de medicamentos descritas en este estudio.
CONSIDERACIONES FINALES Y PROPUESTA DE METODOLOGÍA INTEGRADA
A partir de la importancia que tienen los medicamentos genéricos para el acceso a estos por la población, es indispensable que se cuente con una cultura en el uso con valor agregado de la información contenida en los documentos de patentes que permita generar los conocimientos necesarios para el desarrollo, producción y comercialización de medicamentos genéricos.
Lo anterior cobra especial importancia en los proyectos a ciclo completo que tienen como objetivo el desarrollo de medicamentos genéricos. En este caso específico la información contenida en los documentos de patentes es de un valor incalculable, especialmente la información contenida en la memoria descriptiva y en las reivindicaciones.
Se debe dominar la información contenida y el alcance de las diferentes bases de datos de patentes libres y comerciales, así como conocer las metodologías o guías existentes relacionadas con los procesos de búsqueda de esta información, y su procesamiento y análisis.
Las metodologías How to Conduct Patent Searches for Medicines: a Step-By-Step Guide28 y Methodology for Patent Searches on Essential Medicines in Developing Countries,29 son de gran utilidad para la búsqueda de patentes; de aquí la importancia de su uso por profesionales vinculados al campo de los medicamentos genéricos. No obstante, no se puede considerar que con su sola aplicación se brinda una respuesta integral a las necesidades de los usuarios.
De lo expuesto en este estudio se puede concluir la necesidad del desarrollo de una metodología integrada (Fig. 4) diseñada a partir de la metodología de generación y gestión de conocimientos (MGGC),41 a la cual se integre en la etapa de búsqueda de información de patentes, aspectos indispensables que aparecen en las metodologías referidas en el párrafo anterior.
]]>
REFERENCIAS BIBLIOGRÁFICAS
1. Organización Mundial del Comercio. Acuerdo sobre los aspectos de los derechos de propiedad intelectual relacionados con el comercio. 1994 [citado 16 de septiembre 2013]. Disponible en: http://www.wto.org/spanish/docs_s/legal_s/27-trips.pdf
http://www.wto.org/spanish/thewto_s/minist_s/min01_s/mindecl_trips_s.pdf
3. Correa C. Repercusiones de la Declaración de Doha relativa al acuerdo sobre los ADPIC y la salud pública, economía de la salud y medicamentos. Serie EDM N° 12. 2002 [citado: 3 de septiembre 2013]. Disponible en: http://apps.who.int/medicinedocs/pdf/s4904s/s4904s.pdf
4. Velásquez G. Medicamentos genéricos y el Acuerdo de los ADPIC (TRIPS); medicamentos esenciales y política farmacéutica. OMS; 2002 [citado 10 de septiembre de 2013]. Disponible en:
http://www.femeba.org.ar/fundacion/quienessomos/Novedades/congreso_genericos/velasquezauditorio10hs.pdf
5. Mirza Z. Access and information needs from a public health perspective. Access to medicines, patent information and freedom to operate. Geneva: Joint WHO, WIPO, WTO Technical Symposium; 2011 [citado 10 de septiembre de 2013]. Disponible en: http://www.wto.org/english/tratop_e/trips_e/techsymp_feb11_e/mirza_18.2.11_e.pdf
6. Organización Panamericana de la Salud/Organización Mundial de la Salud. Resolución CD45.R7 Acceso a los Medicamentos; 45to. Consejo Directivo; 56ta. Sesión del Comité Regional; 2004 [citado 10 de septiembre de 2013]. Disponible en: http://iris.paho.org/xmlui/bitstream/handle/123456789/256/CD45.r7-s.pdf?sequence=2
7. Organización Panamericana de la Salud. Guía para la implementación de estrategias de medicamentos genéricos en los países de América Latina y el Caribe como mecanismo para mejorar el acceso a los medicamentos (Documento No. 3: Serie técnica de medicamentos esenciales, políticas farmacéuticas). Washington, DC: OPS, 2011 [citado 10 de sept 2013]. Disponible en: http://apps.who.int/medicinedocs/documents/s19196es/s19196es.pdf
8. Mirza Z. WHO perspective on medicine patents and FTAs; WHO; 2005 [citado 12 de septiembre 2013]. Disponible en:
http://twnside.org.sg/title2/FTAs/Intellectual_Property/IP_and_Access_to_Medicines/WHOPerspectiveonMedicinePatents&FTAs-Zafar%20Mirza.ppt
9. Organización Mundial de la Salud/Organización Mundial del Comercio. Los acuerdos de la OMC y la Salud Pública: un estudio conjunto de la OMS y la Secretaría Conjunta de la OMC. OMS-OMC; 2002 [citado 12 de septiembre de 2013]. Disponible en: http://www.wto.org/spanish/res_s/booksp_s/who_wto_s.pdf
10. Organización Panamericana de la Salud. El acceso a los medicamentos de alto costo en las Américas. Contextos, desafíos y perspectivas (Serie técnica No. 1: Medicamentos esenciales, acceso e innovación). Washington, DC: OPS; 2009 [citado 12 de septiembre de 2013]. Disponible en: http://www.esencialesparalavida.org/upload/file/acceso%20%20medicam%20alto%20coste%20PAO.pdf
11. Lage Dávila A. Global pharmaceutical development and access: critical issues of ethics and equity. MEDICC review. 2011;13(3):16-22.
12. World Health Organization/World Intellectual Property Organization/World Trade Organization. Promoting access to medical technologies and innovation. Intersections between public health, intellectual property and trade; 2013 [citado 12 de septiembre de 2013]. Disponible en: http://www.wto.org/english/res_e/booksp_e/pamtiwhowipowtoweb13_e.pdf
13. World Health Organization. Pharmaceutical production and related technology transfer. WHO; 2000 [citado 12 de septiembre de 2013]. Disponible en: http://www.who.int/phi/publications/Local_production_and_access_to_medicines.pdf
14. Organización Mundial de la Salud. Globalización y acceso a los medicamentos. Serie Economía de la salud y medicamentos, No. 07; 2000 [citado 16 de septiembre de 2013]. Disponible en: http://apps.who.int/medicinedocs/es/d/Jwhozip47s/
15. World Health Organization. Local production for access to medical products: developing a framework to improve public health. World Health Organization 2013 [citado 16 de septiembre de 2013]. Disponible en:
http://ictsd.org/downloads/2011/12/local_production_policy_framework1.pdf
16. World Health Organization. Local production of pharmaceuticals and related technology transfer in developing countries. World Health Organization; 2011 [citado 16 de septiembre de 2013]. Disponible en: http://unctad.org/en/PublicationsLibrary/diaepcb2011d7_en.pdf
17. Kaplan W, Laing R. Local production of pharmaceuticals: industrial policy and access to medicines; 2005 [citado 16 de septiembre de 2013]. Disponible en: http://www.who.int/medicines/technical_briefing/tbs/KaplanLocalProductionFinal5b15d.pdf
18. World Health Organization/United Nations Children's Fund/United Nations Programme on HIV/AIDS. Global update on HIV treatment 2013: results, impact and opportunities. World Health Organization; 2013 [citado 16 de septiembre de 2013]. Disponible en: http://apps.who.int/iris/bitstream/10665/85326/1/9789241505734_eng.pdf
19. Medicine Patent Pool. Estado de patentes de antirretrovirales. 2013 [citado 20 de septiembre de 2013]. Disponible en: http://www.medicinespatentpool.org/datos-sobre-patentes/estado-de-patentes-de-antirretrovirales/?lang=es
20. WIPO Re. Search Data Base [citado 20 de septiembre de 2013]. Disponible en: http://www.wipo.int/research/en/search/OrangeBook
21. Organización Mundial de la Propiedad Intelectual. Guía de la OMPI para la utilización de la información de patentes. Publicación de la OMPI N° L434/3(S); 2013 [citado 20 de septiembre de 2013]. Disponible en:
http://www.wipo.int/export/sites/www/freepublications/es/patents/434/wipo_pub_l434_03.pdf
22. Organización Mundial de la Propiedad Intelectual. PatentScope, Búsqueda y CLIR. Publicación de la OMPI Nº L434/7S; 2012 [citado 20 de septiembre de 2013]. Disponible en:
http://www.wipo.int/export/sites/www/freepublications/es/patents/434/wipo_pub_l434_07.pdf
23. Organización Mundial de la Propiedad Intelectual. Guía de Buenas Prácticas "Linking Innovation and Industrial Property". 2013 [citado 20 de septiembre de 2013]. Disponible en: http://www.patentsoffice.ie/PDF%20Documents/IP%20Good%20Practice%20Guide.pdf
24. Organización Mundial de la Propiedad Intelectual. Las patentes: fuente de información tecnológica; 2013 [citado: 20 de septiembre de 2013]. Disponible en: http://www.wipo.int/export/sites/www/freepublications/es/patents/434/wipo_pub_l434_02.pdf
25. Ortiz I, Escorsa E. Guía de buenas prácticas para la búsqueda de información en patentes. 2013 [citado 20 de septiembre de 2013]. Disponible en: http://pipra.fia.cl/media/7647/manual_patentes.pdf
26. Academia Europea de Patentes. Manual didáctico sobre patentes. AEP; 2009 [citado: 20 de septiembre de 2013]. Disponible en: http://www.oepm.es/cs/OEPMSite/contenidos/PatentKit/Manual.pdf
27. Amin T. How to conduct patent searches for medicines: a step-by-step guide. WHO; 2010 [citado 20 de septiembre de 2013]. Disponible en: http://apps.who.int/medicinedocs/documents/s17398e/s17398e.pdf
28. Milani B, Oh C. Methodology for patent searches on essential medicines in developing countries. PNUD; 2012 [citado 20 de septiembre de 2013]. Disponible en: http://www.undp.org/content/dam/undp/library/hivaids/English/Patent%20Information%20and%20Transparency.pdf
29. Infante M. Procedimiento para la vigilancia tecnológica en el ámbito universitario con el uso de las tecnologías de la web 2.0. La Habana: Instituto Superior Politécnico "José Antonio Echeverría". Tesis presentada en opción del Título de Máster en Informática Empresarial; 2011.
30. Delgado M, Arrebato-Agüero L. Diagnóstico integrado de la vigilancia tecnológica en organizaciones. Ingeniería Industrial. 2011;32(2):151-6.
31. Delgado M, Infante MB, Abreu Y, Infante O, Batista A, Martínez J. Vigilancia Tecnológica en una Universidad de Ciencias Técnicas. Ingeniería Industrial. 2011;1:69-75.
32. Morcillo P. Vigilancia e inteligencia competitiva: fundamentos e implicaciones. Rev Invest Gest Innov Tecnol. 2003;2(17):2-10.
33. Mignogna R. Competitive intelligence [Internet]. 1997 [citado 20 de septiembre de 2013]. Disponible en: http://www.chewy.gatech.edu/t2s/index/html
34. Sánchez J. Herramientas de Software para la práctica de la inteligencia competitiva en la Empresa. Madrid: Triz XXI; 2002.
35. Nosella A. Technological change and technology monitoring process: evidence from four Italian case studies. J Engin Technol Managm. 2008;25(4):321-37.
36. Vázquez R. Informe APEI sobre vigilancia tecnológica. Gijón, España: Informe APEI 4; 2009.
37. Núñez P. Guía metodológica para el estudio de las necesidades de formación e información de los usuarios o lectores. ACIMED. 1997;5(3):32-51.
38. AFNOR. Surveillance services and implementation services for a surveillance system. French Standar. France: AFNOR; 2011: XPX50-053:31.
39. AENOR. Gestión de la I+D+i: Sistema de Vigilancia Tecnológica e Inteligencia Competitiva. Norma Española. Madrid: UNE-166000. AENOR; 2011.
40. García B. Gestión y generación de conocimientos a partir de la información de patentes. Metodología. La Habana: Tesis presentada en opción del título de Máster en Gestión de la Propiedad Intelectual. OCPI; 2012.
41. García B, Delgado M, Infante M. Metodología para la generación y gestión del conocimiento para proyectos de i+d+i vista desde sus factores críticos. Rev Cubana Inform Cienc Salud. 2014;25(3):285-302.
]]> Recibido: 16 de octubre de 2014.
Aprobado: 10 de diciembre de 2014.
Dra. Beatriz Moraima García Delgado. Centro de Aplicaciones Tecnológicas y Desarrollo Nuclear (CEADEN). Miramar, La Habana, Cuba. Correo electrónico: beatrizgarcia@infomed.sld.cu
a Los ADPIC vinculan por primera vez los temas de propiedad intelectual al comercio.
]]> bEn su acápite 1 plantea: Reconocemos la gravedad de los problemas de salud pública que afligen a muchos países en desarrollo y menos adelantados, especialmente los resultantes del VIH/SIDA, la tuberculosis, el paludismo y otras epidemias.c Convenimos en que el Acuerdo sobre los ADPIC no impide ni deberá impedir que los Miembros adopten medidas para proteger la salud pública. En consecuencia, al tiempo que reiteramos nuestro compromiso con el Acuerdo sobre los ADPIC, afirmamos que dicho Acuerdo puede y deberá ser interpretado y aplicado de una manera que apoye el derecho de los Miembros de la OMC de proteger la salud pública y, en particular, de promover el acceso a los medicamentos para todos.
dExención Bolar: por la que se permitía a fabricantes de medicamentos genéricos producir y/o importar y utilizar ciertas cantidades de un producto patentado con el fin de realizar las pruebas necesarias para obtener la aprobación reglamentaria antes de la expiración de una patente. Canadá - Protección mediante patente de los productos farmacéuticos, WT/DS114/R, de 17 de marzo de 2000.
e De algunos países en vías de desarrollo.
fSegún el artículo 29.1 del Acuerdo sobre los ADPIC: “Los Miembros exigirán al solicitante de una patente que divulgue la invención de manera suficientemente clara y completa para que las personas capacitadas en la técnica de que se trate puedan llevar a efecto la invención, y podrán exigir que el solicitante indique la mejor manera de llevar a efecto la invención que conozca el inventor en la fecha de la presentación de la solicitud o, si se reivindica la prioridad, en la fecha de prioridad reivindicada en la solicitud”.
g FTO: Freedom to Operate.
h Esta metodología tuvo una larga evolución, comenzó como un proyecto piloto de la OMS (2005), con la asistencia técnica de la Oficina Europea de Patentes (EPO) y tuvo como objetivo desarrollar un método para la búsqueda de patentes de medicamentos esenciales en los países en desarrollo. Posteriormente (2008), el PNUD conjuntamente con la OMS y EPO, organizaron una consulta de expertos, que contó con la participación de representantes de: oficinas de patentes, autoridades regulatorias de medicamentos, instituciones de investigaciones y universidades, así como organizaciones de la sociedad civil, para proveer una asesoría técnica de la metodología. Basado en el mencionado proyecto, la Oficina Regional del Sureste de Asía y de la Región Occidental del Pacífico, publicó (2010) una breve guía para conducir las búsquedas de patentes.
iInternational non-proprietary names (INNs).
jLas llamadas marañas de patentes (patents tickets).