REPORT
Isolation and partial purification of a hemolytic sphingomyelin-inhibitable fraction from the sea anemone Anthopleura nigrescens
Aislamiento y purificación parcial de una fracción hemolítica inhibida por esfingomielina de la anémona marina Anthopleura nigrescens
]]>
Javier Alvarado1, Yeney Álvarez2, Lohans Pedrera2, Uris Ros2, María E Lanio2, Aisel Valle2, Carlos Álvarez2
1 Escuela de Ciencias Biológicas, Facultad de Ciencias Exactas y Naturales, Universidad Nacional. Apartado 86-300, Heredia, Costa Rica.
2 Centro de Estudio de Proteínas, Facultad de Biología, Universidad de La Habana, UH. Calle 25 No. 455 entre I y J, Vedado, CP 10400, La Habana, Cuba.
ABSTRACT
]]> Actinoporins are highly hemolytic pore-forming proteins with a molecular mass of around 20 kDa and high affinity for sphingomyelin-containing membranes. On the crude extract of the sea anemone Anthopleura nigrescens, hemolytic activity (HA) was detected. In order to identify the presence of pore-forming proteins similar to actinoporins in this anemone, the fractionation and analysis of its crude extract was carried out. The aqueous extract of the whole body was subjected to gel filtration chromatography on Sephadex G50 medium rendering three resolved peaks (P-I, P-II, and P-III) as measured by their absorbance at 280 nm. Functional characterization of the crude extract and chromatographic fractions was evaluated by HA against human red blood cells. The crude extract and two peaks (P-II and P-III) showed HA. Interestingly, the HA of the crude extract and P-II were specifically inhibited by small unilamellar vesicles of phosphatidylcholine: sphingomyelin (1:1). Both the crude extract and P-II revealed the existence of at least one protein band around 20 kDa by SDS-PAGE. The inhibition by sphingomyelin of the whole body extract HA and the localization of this property in P-II that could be associated with a protein of around 20 kDa suggest the presence of at least one novel actinoporin in A. nigrescens. Furthermore, this is the first report of a biochemical activity for this sea anemone. Work is in progress in order to purify and characterize the molecular entities responsible for this HA inhibited by sphingomyelin.
Keywords: actinoporin, Anthopleura nigrescens, sphingomyelin, hemolytic activity.
RESUMEN
Las actinoporinas son proteínas formadoras de poro, altamente hemolíticas, de aproximadamente 20 kDa, con una elevada afinidad por las membranas que contienen esfingomielina. En el extracto crudo de la anémona marina Anthopleura nigrescens se detectó actividad hemolítica (AH). Con el fin de identificar la presencia de proteínas formadoras de poros, similares a las actinoporinas, se fraccionó y analizó este extracto crudo. Se sometió a cromatografía de filtración en gel de Sephadex G50 medio, y se obtuvieron tres picos resueltos (P-I, P-II y P-III), según su absorbancia a 280 nm. La caracterización funcional del extracto y las fracciones cromatográficas se evaluaron por la AH frente a eritrocitos humanos. El extracto crudo y los picos II y III mostraron AH. Curiosamente la AH del extracto crudo y del pico II fueron inhibidas por vesículas unilamelares pequeñas de fosfatidilcolina: esfingomielina (1:1). Tanto el extracto crudo como P-II revelaron al menos una banda proteica de alrededor de 20 kDa, en SDS-PAGE. La inhibición por esfingomielina de la AH del extracto y la localización de esta propiedad en P-II, podría asociarse con una banda proteica de alrededor de 20 kDa, lo que sugiere la presencia de al menos una actinoporina en A. nigrescens. Este es el primer informe de una actividad bioquímica para esta anémona marina. Se continúa trabajando para purificar y caracterizar las entidades moleculares responsables de esta AH inhibida por esfingomielina.
Palabras clave: actinoporina, Anthopleura nigrescens, esfingomielina, actividad hemolítica.
INTRODUCTION
]]> Sea anemones are sessile animals that produce venom for defense from predators and capture of their preys. This secretion consists of an ensemble of molecules including phospholipases [1-4], sodium channel blockers [5], protease inhibitors [6], and pore-forming toxins called actinoporins [7-13]. Actinoporins are highly hemolytic, basic cysteine-less proteins with molecular weight around 20 kDa showing high affinity for membranes containing sphingomyelin; therefore, their hemolytic activity (HA) is characteristically inhibited when the toxin is pre-incubated with liposomes containing this phospholipid [11, 14-16]. These toxins have attracted the attention for their potential use in biomedicine and biotechnology, for instance, in the construction of immunotoxins against cancer cells [17, 18] or as a tool for protein-membrane interaction studies [19, 20].Recently, we described the presence of the sea anemone Anthopleura nigrescens on the Pacific coast of Costa Rica [21]. Here, we report the HA against human red blood cells (RBC) of the crude extract and two fractions obtained by molecular exclusion chromatography. The HA detected in the crude extract and in one partially purified fraction were inhibited by sphingomyelin, suggesting the presence of at least one novel actinoporin in A. nigrescens.
Recently, we described the presence of the sea anemone A. nigrescens on the Pacific coast of Costa Rica [21]. Here, we report the HA against human red blood cells (RBC) of the crude extract and two fractions obtained by molecular exclusion chromatography. The HA detected in the crude extract and in one partially purified fraction were inhibited by sphingomyelin, suggesting the presence of at least one novel actinoporin in A. nigrescens.
MATERIALS AND METHODS
Specimens were collected during low tide in a rocky area on the Pacific coast of Costa Rica (9º55′01.99′′N-84º 42′49.50′′W). Sea anemones were carefully detached from the rock, placed in plastic sample containers, and transported at 4 ºC ± 2 ºC to the laboratory. Crude extracts were obtained by mincing and homogenizing the whole animal body in distilled water (1:2, w/v). Protein concentration was determined according to Bradford [22] by 0.01 % (w/v) Coomassie Brilliant Blue G-250.
The crude extract (4.5 mg of protein) was applied onto a Sephadex G-50 medium (GE-Healthcare) column, equilibrated with 0.02 mol/L sodium phosphate buffer pH 7 at a flow rate of 27.5 cm/h. Fractions of 1 mL were collected and monitored at 280 nm for protein content estimation and HA determination.
The crude extract and fractions homogeneity were checked by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) using a 15 % polyacrylamide gel. Samples were dissolved in Laemmli buffer (10 % SDS, 0.02 M 2-mercaptoethanol, pH 6.8) [23] and heated at 100 °C for 10 min. Protein staining was carried out with Coomassie blue dye at 0.05 % (m/v). Molecular weight markers (8 to 220 kDa) ColorBurst™ (Sigma Aldrich) were used.
HA on RBC was evaluated turbidimetrically at 600 nm at room temperature (25 °C ± 2 °C) as previously described by Martínez et al. [24]. Briefly, the erythrocyte suspension was prepared using pooled fresh RBC, washed by centrifugation and resuspended in Tris-buffered saline (TBS; 145 mM NaCl, 10 mM Tris-HCl, pH 7.4). The cell suspension was diluted with TBS to an absorbance of 0.1 at 600 nm and the decrease in absorbance was followed in a microplate reader (MultiSkan FC, Thermo Scientific). The samples were two-fold serially diluted with TBS and the reaction started by adding the same volume of cell suspension to each well (200 μL final volume). The loss of turbidity was quantitatively related to the HA. Negative and positive controls were also run in parallel using RBC without the extract or fractions, and RBC with the actinoporin sticholysin II (St II) from Stichodactyla helianthus [9], respectively. HA was quantitatively calculated from a St II standard curve and expressed as protein nanogram equivalents.
Small unilamellar vesicles (SUV) of phosphatidyl-choline: sphingomyelin (PC:SM; 1:1) were used for the HA inhibition experiments. The crude extract and fractions that showed HA were incubated at room temperature for 30 min with SUV at different lipid: protein mass ratio (5, 2.5, 1.25, 0.6, 0.3) and in the absence of lipids. HA in incubated samples was determined as described above.
]]> The small unilamellar vesicles (SUV) of 1,2-dioleoyl-sn-glycero-3-phosphocholine (PC): egg SM (Avanti Polar Lipids), at 1:1 molar ratio, were prepared by thorough sonication of a multilamellar liposome suspension. For multilamellar vesicle preparation, the appropriate amounts of both lipids dissolved in chloroform: methanol solution 2:1 (v/v) were mixed and evaporated thoroughly at 40 °C. SUV were prepared by sonicating the multillamelar vesicle suspension in buffer (10 mM Tris-HCl, pH 7.4) with a probe ultrasonicator (Branson 450, Danbury, USA) as previously described [25]. Titanium particles released from the probe were removed by further centrifugation at 10 000 g for10 min at 22 °C. Phospholipid concentration was measured by determining inorganic phosphate according to Rouser et al. [26].
RESULTS AND DISCUSSION
The body total aqueous extract (1.5 mg/mL estimated on protein grounds) showed HA against RBC in the ng/mL concentration range (HC50 ≈ 100 ng/mL) similarly to Stichodactyla helianthus’s StI and StII (HC50 ≈ 30-45 ng/mL pure fractions) as reported by Lanio et al. [9].
In order to gain some insight into the nature of the proteins responsible for the HA, the effect of incubation with liposomes containing SM on their HA was studied. The HA of the crude extract was partiality inhibited upon incubation for 30 min with PC:SM (1:1) SUV (Figure 1A). Two orders of magnitude of protein concentration were necessary to recover the HA of the incubated fraction with SM in respect to the non-incubated fraction (Figure 1B). This suggested that the crude extract HA could be caused by at least one novel actinoporin.
The gel filtration chromatographic step of the total body extract rendered two defined peaks of HA (P-II and P-III) while peak I (P-I) was devoid of HA (Figure 2A). As evidenced by SDS-PAGE, P-II showed a band with a molecular weight of around 20 kDa (Figure 2B), a characteristic of the actinoporin family [16, 19, 20]. Peak III showed two bands with molecular weights between 60-100 kDa (Figure 2B). This result is surprising because these bands arise from the last fraction of the gel filtration chromatography (Figure 2A), where small molecules are expected to be found. Probably, such relatively high molecular weight proteins are somehow retained by the Sephadex matrix, and consequently are not properly resolved by gel filtration.
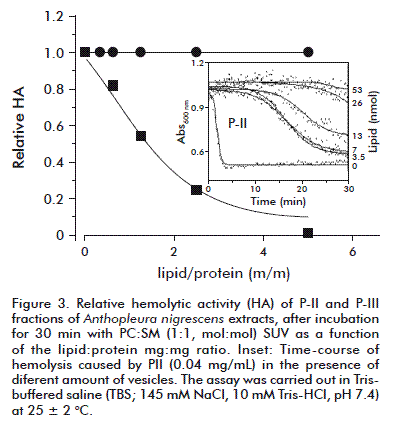
Upon incubation with sphingomyelin-containing vesicles, P-II showed a reduction of its HA that was dependent on the proportion of vesicle: protein assayed (Figure 3) and the time elapsed (inset to Figure 3). In fact, P-II HA inhibition was observed from a very low lipid: protein mass ratio (0.3) and it was completed when a ratio of 5 was achieved. Conversely, P-III HA was not affected by incubation with the same liposomes.
Altogether, the hemolytic activity, the inhibition by sphingomyelin of the whole body extract HA, and the localization of this property in P-II that could be associated with a protein of around 20 kDa, strongly suggests the presence of at least one novel actinoporin in the sea anemone A. nigrescens. Furthermore, this is the first report of a biochemical activity for this anemone. Work is in progress in order to purify and characterize the molecular entities responsible for this HA inhibited by sphingomyelin.
]]>
ACKNOWLEDGEMENTS
This work was supported by International Foundation for Science: IFS Grant F/5205-1, National University of Costa Rica: Project SIA 0279-10 -Ley de Pesca and Red Iberoamericana CYTED: BIOTOX-212RT0467.
REFERENCES
1. Pazos IF, Álvarez C, Gómez T, Lanio M. Caracterización enzimática de una fracción con actividad fosfolipasídica aislada de la anémona marina Stichodactyla helianthus. Revista Biología. 1993;7(2):90-6.
2. Romero L, Marcussi S, Marchi-Salvador DP, Silva FP Jr, Fuly AL, Stabeli RG, et al. Enzymatic and structural characterization of a basic phospholipase A(2) from the sea anemone Condylactis gigantea. Biochimie. 2010;92(8):1063-71.
3. Moran Y, Genikhovich G, Gordon D, Wienkoop S, Zenkert C, Ozbek S, et al. Neurotoxin localization to ectodermal gland cells uncovers an alternative mechanism of venom delivery in sea anemones. Proc Biol Sci. 2012;279(1732):1351-8.
4. Diochot S, Lazdunski M. Sea anemone toxins affecting potassium channels. Prog Mol Subcell Biol. 2009;46:99-122.
5. Honma T, Shiomi K. Peptide toxins in sea anemones: structural and functional aspects. Mar Biotechnol (NY). 2006;8(1):1-10.
6. Delfin J, Gonzalez Y, Diaz J, Chavez M. Proteinase inhibitor from Stichodactyla helianthus: purification, characterization and immobilization. Arch Med Res. 1994; 25(2):199-204.
7. Bernheimer AW, Avigad LS. Properties of a toxin from the sea anemone Stoichactis helianthus, including specific binding to sphingomyelin. Proc Natl Acad Sci USA. 1976;73(2):467-71.
]]>8. Kem WR, Dunn BM. Separation and characterization of four different amino acid sequence variants of a sea anemone (Stichodactyla helianthus) protein cytolysin. Toxicon. 1988;26(11):997-1008.
9. Lanio ME, Morera V, Alvarez C, Tejuca M, Gomez T, Pazos F, et al. Purification and characterization of two hemolysins from Stichodactyla helianthus. Toxicon. 2001; 39(2-3):187-94.
10. Bellomio A, Morante K, Barlic A, Gutierrez-Aguirre I, Viguera AR, Gonzalez-Manas JM. Purification, cloning and characterization of fragaceatoxin C, a novel actinoporin from the sea anemone Actinia fragacea. Toxicon. 2009;54(6):869-80.
11. Kohno Y, Satoh H, Iguchi A, Nagai H. Characterization of a new hemolytic protein toxin from the sea anemone Anthopleu-ra asiatica. Fish Sci. 2009;75(4):1049-54.
12. Monastyrnaya M, Leychenko E, Isaeva M, Likhatskaya G, Zelepuga E, Kostina E, et al. Actinoporins from the sea anemones, tropical Radianthus macrodactylus and northern Oulactis orientalis: Comparative analysis of structure-function relationships. Toxicon. 2010;56(8):1299-314.
]]> 13. Anderluh G, Sepčic K, Turk T, Maček P. Cytolytic proteins from Cnidarians- an Overview. Acta Chim Slov. 2011;58(4):724-29.14. Belmonte G, Pederzolli C, Macek P, Menestrina G. Pore formation by the sea anemone cytolysin equinatoxin II in red blood cells and model lipid membranes. J Membr Biol. 1993;131(1):11-22.
15. Valcarcel CA, Dalla Serra M, Potrich C, Bernhart I, Tejuca M, Martinez D, et al. Effects of lipid composition on membrane permeabilization by sticholysin I and II, two cytolysins of the sea anemone Stichodactyla helianthus. Biophys J. 2001;80(6):2761-74.
16. Anderluh G, Maček P. Cytolytic peptide and protein toxins from sea anemones (Anthozoa: Actiniaria). Toxicon. 2002;40(2):111-24.
17. Potrich C, Tomazzolli R, Dalla Serra M, Anderluh G, Malovrh P, Macek P, et al. Cytotoxic activity of a tumor protease-activated pore-forming toxin. Bioconjug Chem. 2005; 16(2):369-76.
18. Tejuca M, Anderluh G, Dalla Serra M. Sea anemone cytolysins as toxic components of immunotoxins. Toxicon. 2009;54(8):1206-14.
]]>19. Alvarez C, Mancheño JM, Martinez D, Tejuca M, Pazos F, Lanio ME. Sticholysins, two pore-forming toxins produced by the Caribbean Sea anemone Stichodactyla helianthus: their interaction with membranes. Toxicon. 2009;54(8):1135-47.
20. Alegre-Cebollada J, Onaderra M, Gavilanes JG, del Pozo AM. Sea anemone actinoporins: the transition from a folded soluble state to a functionally active membrane-bound oligo-meric pore. Curr Protein Pept Sci. 2007;8(6):558-72.
21. Acuña FH, Alvarado J, Garase A, Cortés J. First record of the sea anemone Anthopleura nigrescens (Cnidaria: Actiniaria: Actiniidae) on the Pacific coast of Central America. Mar Biodiv Rec. 2012;24(5):1-3.
22. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248-54.
23. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227(5259):680-5.
]]>24. Martinez D, Campos AM, Pazos F, Alvarez C, Lanio ME, Casallanovo F, et al. Properties of St I and St II, two isotoxins isolated from Stichodactyla helianthus: a comparison. Toxicon. 2001;39(10):1547-60.
25. Pazos IF, Alvarez C, Lanio ME, Martinez D, Morera V, Lissi EA, et al. Modification of sticholysin II hemolytic activity by free radicals. Toxicon. 1998;36(10):1383-93.
26. Rouser G, Fkeischer S, Yamamoto A. Two dimensional then layer chromatographic separation of polar lipids and determination of phospholipids by phosphorus analysis of spots. Lipids. 1970;5(5):494-6.
]]> Carlos Álvarez. Centro de Estudio de Proteínas, Facultad de Biología, Universidad de La Habana, UH. Calle 25 No. 455 entre I y J, Vedado, CP 10400, La Habana, Cuba. E-mail: calvarez@fbio.uh.cu. ]]>