RESEARCH
Application of a colorimetric CTLL-2 cell proliferation assay for the evaluation of IL-15 antagonists
Aplicación de un ensayo colorimétrico de proliferación celular en CTLL-2 para la evaluación de antagonistas de IL-15
]]>
Yunier Rodríguez-Álvarez1, Haydee Gerónimo-Pérez2, Hilda Garay-Pérez3, Jorge Castro-Velazco4, Gerardo García-Illera2, Alicia Santos-Savio1
1 Departamento de Farmacéuticos, Dirección de Investigaciones Biomédicas, Centro de Ingeniería Genética y Biotecnología, CIGB. Ave. 31 entre 158 y 190, Cubanacán, Playa, CP 11600, La Habana, Cuba.
2 Dirección de Control de la Calidad, CIGB. La Habana, Cuba.
3 Departamento de Química-Física, Dirección de Investigaciones Biomédicas, CIGB. La Habana, Cuba.
4 Bioterio, CIGB. La Habana, Cuba.
]]>
ABSTRACT
Dysregulated expression of Interleukin (IL)-15 has been associated to several autoimmune and inflammatory diseases. In this sense, some agents that inhibit IL-15 activity have been developed as possible drugs to treat these pathologies. We have been working in two strategies to inhibit IL-15 activity, a peptide (named P8) that binds to the alpha subunit of the IL-15 receptor (IL-15R) and a vaccine based on active immunization with human IL-15 (hIL-15). To measure the biological activity of the IL-15 antagonists we used the proliferation assay in CTLL-2, a cell line that depends on IL-2/IL-15 to proliferate. In the current work we defined the conditions of the assay to determine the half maximal inhibitory concentration (IC50) of the P8 peptide and the neutralizing titers of the sera from monkeys immunized with the anti-IL-15 vaccine. The specificity of the assay for IL-15 was documented using anti-IL-15 and anti-IL-2 specific antibodies. We also examined the specificity of the antagonists of IL-15 in presence of IL-2; neither the peptide nor the sera inhibited the cell proliferation induced by human IL-2. Finally, we evaluated the effect of commercial antibodies anti-hIL-15 and sera from mice immunized with hIL-15 on murine IL-15 (mIL-15). In this case, neither the antibodies nor the sera inhibited mIL-15; therefore, murine models are not useful to evaluate the effectiveness of anti-hIL-15 vaccine. The application of this assay allowed us to evaluate different strategies to inhibit the activity of IL-15 for its future development as drugs
Keywords: Interleukin-15, antagonist, peptide, serum, CTLL-2 cell line, proliferation assay.
RESUMEN
La expresión no regulada de la interleucina-15 (IL-15) se ha asociado con el desarrollo y patogénesis de varias enfermedades autoinmunes e inflamatorias. Se han desarrollado numerosos agentes que inhiben la actividad de la IL-15 como posibles fármacos para el tratamiento de estas patologías. Nosotros hemos trabajado en dos estrategias para inhibir la actividad de la IL-15, un péptido (llamado P8) que se une a la subunidad alfa del receptor de IL-15 y una vacuna basada en la inmunización activa con IL-15 humana (IL-15h). La actividad biológica de los antagonistas de IL-15 se midió mediante el ensayo de proliferación en CTLL-2, una línea celular que depende de IL-2/IL-15 para proliferar. En este trabajo se definieron las condiciones del ensayo para determinar la concentración inhibitoria media máxima (CI50) del péptido P8 y los títulos de neutralización de los sueros de monos inmunizados con la vacuna anti-IL-15. La especificidad del ensayo para la IL-15 se demostró usando anticuerpos específicos anti-IL-15 y anti-IL-2. También se examinó la especificidad de los antagonistas de IL-15 en presencia de IL-2, y se demostró que ni el péptido ni los sueros inhiben la proliferación celular inducida por IL-2 humana. Finalmente, se evaluó el efecto de anticuerpos comerciales anti-IL-15h y de sueros de ratones inmunizados con IL-15h sobre la IL-15 murina (IL-15m). Ni los anticuerpos ni los sueros inhiben la IL-15m, lo que indica que los modelos murinos no son útiles para evaluar la efectividad de la vacuna anti-IL-15h. Este ensayo permitió evaluar diferentes estrategias que inhiben la actividad de la IL-15 para su futuro desarrollo como fármacos.
Palabras clave: interleucina-15, antagonista, péptido, suero, línea celular CTLL-2, ensayo de proliferación.
]]>
INTRODUCTION
Interleukin-15 (IL-15) is a 14-15 kDa glycoprotein that belongs to the four-helix bundle cytokine family and was first characterized by its ability to substitute IL-2. Indeed, IL-15 shares with IL-2, β and γ subunits receptor but has a unique private α subunit (IL-15Rα) that is responsible for high-affinity binding [1]. IL-15 is essential to the development, function and survival of natural killer (NK) cells, NK T cells and memory CD8+ T cells [2, 3]. However, deregulated IL-15 expression has been demonstrated in patients with autoimmune inflammatory diseases, including rheumatoid arthritis, inflammatory bowel diseases, celiac diseases, psoriasis and sarcoidosis [4-10]. Therefore, various strategies were considered to target IL-15, its receptor or the molecules involved in IL-15-induced signaling for the treatment of such autoimmune diseases. In this sense, several agents that inhibit IL-15 activity have been developed, including soluble IL-15Rα, mutant IL-15 molecules, and antibodies specific for IL-15 or IL-2/15Rβ [11-14]. Also, we have been working in two strategies to inhibit IL-15 activity, a peptide that binds IL-15Rα [15] and a vaccine based on active immunization with hIL-15.
Cell proliferation assay is used to measure the biological activity for many cytokines and the effect of its antagonists on susceptible cells. In particular, CTLL-2 is a cell line dependent to IL-2/IL-15 to proliferate. This cell line expresses all three receptor subunits for IL-2 and IL-15, which is advantageous to evaluate approaches that target different receptor subunits for IL-15 [16]. Here we used a colorimetric method based on redox sensitive formazan (3-(4-5) dimethylthiazol-2yl)-2, 5-diphenyl tetrazolium bromide; MTT) because it is a simple and rapid procedure [17]. MTT is a tetrazolium salt that can be cleaved only by active mitochondria in metabolically active cells, and is hence applicable to almost any survival or proliferation assay in which living cells must be distinguished from the dead ones. The assay, which could be carried out in multiwell plates, also offers an advantage for testing a large number of drugs with good reproducibility [18].
To achieve our goal, first, we adjusted the CTLL-2 cell proliferation assay to define the conditions for evaluation of IL-15 antagonists using an international reference standard. The specificity of the assay was demonstrated using a neutralizing anti-IL-15 or anti-IL-2 antibody and IL-2 in dependence of experimental samples. In addition, the application of this assay allowed evaluating the neutralizing activity antibodies or sera generated with hIL-15 on mIL-15.
MATERIALS AND METHODS
]]> Cell line and reagentsThe CTLL-2 IL-2 dependent cell line was grown in RPMI-1640 (Gibco-BRL) containing 2 mM L-glutamine (GIBCO-BRL), 50 μg/mL gentamicin, 10 % heat-inactivated fetal bovine serum (PAA A21-701) and 10 ng/mL recombinant human IL-2 (R&D, 202-IL-010). Cells were incubated at 37 °C with 5 % CO2, 95 % humidity. CTLL-2 cells were harvested and used in log phase growth (cell passage 5 after thawing; cell viability: ≥ 95 %). Prior to use, the CTLL-2 cells were washed five times with RPMI-1640 medium (Gibco-BRL). The CTLL-2 cell bank was generated by the Biological Assays Laboratories from cells obtained from ATCC (TIB-214).
The International Standard reagent, Code: 86/504, was obtained from National Institute of Biological Standards and Controls (NIBSC, UK). Recombinant hIL-15 (Cat#247-IL), recombinant mouse IL-15 (Cat#447-ML), and anti-human IL-2 antibody (Cat#MAB202), anti-hIL-15 antibody (Cat#MAB647), anti-hIL-15 antibody (Cat#MAB247), anti-mIL-15 antibody (Cat#AF447) were from R&D systems, Inc. The Thiazolyl Blue Tetrazolium Bromide (MTT) (Cat#M2128-1G) was from Sigma-Aldrich.
Neutralizing sera
Sera were obtained after immunization with recombinant IL-15 using aluminium hydroxide or Montanide® ISA 51 as adjuvant. Monkeys and mice were immunized with 200 μg or 10 μg of antigen respectively. Immunization scheme include 3 immunizations every 15 days and blood samples were collected at the beginning (preimmune) and 7 days after each immunization. Sera were decomplemented for incubation at 56 °C for 30 min and were stored at -20 ºC until use.
Proliferation assay in CTLL-2 cell line
The bioassay was carried out in a 96-well cell culture cluster (Costar, Corning Inc., Corning, NY USA) at 5 × 103 CTLL-2 cells per well, previously washed five times with RPMI medium (Gibco, Invitrogen Corp., Carlsbad, CA, USA) without serum, and incubated in presence of two-fold serial dilutions (starting dilution 1:1000) of recombinant hIL-15 international standard (NIBSC) and the IL-15 from R&D in 100 μL total volume at 37 ºC and 5 % CO2. After 72 h MTT was added [17] and the plates were further incubated for 4 h. Finally, 100 μL of a solution containing 10 % SDS, 0.1 N HCL and 50 % isopropylalcohol were added per well. Plates were read at 578 nm on a Multiscan (Sensident Scan, Merck, Germany). An International Standard of IL-15 (NIBSC) was used as a reference. The potency (biological activity) was calculated using a statistical program of parallel lines developed in the Quality Control Direction of the CIGB, ParLin V.4.2 (register number: 1485-2004). One unit of activity is defined as the concentration of IL-15 required to induce the half maximal stimulation. This value is referred to as the effective dose at 50 % (ED50) and it was calculated using the ParLin V.4.2 program.
Effect of peptides and serum on proliferation of CTLL-2 cell line
To evaluate the neutralizing capacity of the P8 peptide or serum, two-fold serial dilutions of the peptide (starting concentration 500 μM) or heat inactivated sera (starting dilution 1:100) were performed in 96-well plates (Costar, Corning Inc., Corning, NY USA) in a volume of 30 μL of RPMI medium (Gibco, Invitrogen Corp., Carlsbad, CA, USA) supplemented with 10 % fetal bovine serum (Gibco). Then, previously washed CTLL-2 cells were added in amounts of 5 × 103 cells/ well in 50 μL. Afterwards, 300 pg/mL of hIL-15 or 20 IU/mL of human IL-2 or 800 pg/mL of mIL-15 in a volume of 20 μL was added to each well, and the plate was incubated for 72 h at 5 % CO2 and 37 °C. Proliferation was measured by MTT mitochondrial staining. Results are reported as a mean of triplicate experiments.
The percent of inhibition of IL-15 activity in the presence of P8 peptide was calculated by the equation:
]]>
Where:
OD sample: optical density in presence of P8 peptide; OD control: optical density in presence of non-related peptide.
The Curve Expert Program V.1.3.80 (http://www.curveexpert.net/) was used to calculate the neutralizing titers of sera from monkeys immunized with the anti-IL-15 vaccine.
RESULTS
Assay standardization and specificity
To adjust the CTLL-2 cell proliferation assay, a series of experiments were performed to determine the adequate cell seed density, the range concentration of IL-15 for a satisfactory dose response curve and the incubation time. We used an international standard from the NIBSC as reference and a commercial IL-15 from R&D as laboratory standard. The linearity ranges obtained were from 1 to 5 IU/mL for NIBSC standard as reported (Figure 1A) and 15-200 pg/mL for commercial hIL-15 (R&D). An IL-15 neutralizing antibody from R&D (MAB647) was used to evaluate assay specificity for IL-15. This antibody neutralized the IL-15 proliferation response in a concentration de-pendent manner (Figure 1B).
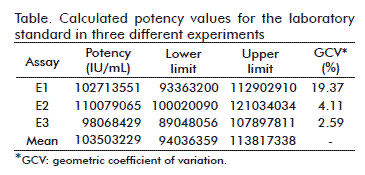
]]> Three different experiments were performed to determine the potency of IL-15 from R&D using as reference, an international standard from NIBSC. The obtained values showed a good repeatability with a geometric coefficient of variation (GCV) lower than or equal to 20 % and these values were between the established limits (Table). The calculated ED50 was 1.035 × 108 IU/mg which is very close to 108 IU/mg, the value reported for the commercial IL-15 [19-21].Evaluation of IL-15 antagonist peptides
In order to evaluate peptides that bind to IL-15Rα as inhibitors of IL-15 activity, first, we defined the experimental conditions. The volume was adjusted to 30 μL of peptide, 20 μL of IL-15 and 50 μL of cells at 5 × 105 cell/well. Two-fold serial dilutions of the peptide were performed to determine the dose range where the peptide is active. We chose 300 pg/mL as the set IL-15 concentration, considering that this dose corresponds to a point near to the proliferation curve plateau. Besides, 24, 48, and 72 h incubations were evaluated, the best results obtained after 72 h (data not shown). A satisfactory dose-response curve was obtained in a range from 500 to 2 μM of P8 peptide and the IC50 calculated was 130 μM. Also a good reproducibility for duplicate data was observed (Figure 2A).
Additionally, a fixed concentration of peptide was also evaluated against different amounts of IL-15, taking it as reference of a non-related peptide. An inhibitory effect of P8 peptide dependent of IL-15 concentration was observed in these experiments (Figure 2B). The inhibition percent calculated was 63.9 %, 61.1 % and 39.5 % for 150 pg/mL, 300 pg/mL and 600 pg/mL of IL-15, respectively.
The specificity of inhibitory effect of this peptide was assessed in presence of IL-2. The P8 peptide and an ovalbumin peptide (OVA peptide) as non-related peptide showed similar dose-response curve to IL-2 without inhibitory effect on IL-2 proliferative activity in CTLL-2. In contrast an anti-IL-2 antibody used as experimental control showed an inhibitory effect on the proliferation in dose dependent manner (Figure 3).
Evaluation of serum from animals immunized with IL-15
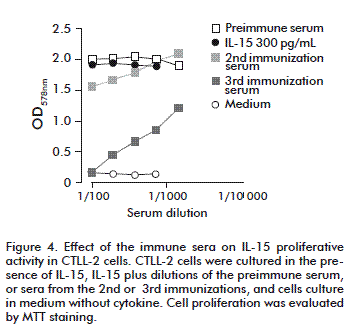
The CTLL-2 proliferation assay was applied to evaluate the neutralizing activity of sera from monkeys immunized with recombinant IL-15 obtained in our laboratory [22], which is structurally different to native IL-15. To measure the effect of sera we used the samples volume established for peptide evaluation, but in this case, the sera were pre-incubated with cytokine (IL-15 or IL-2, in dependence of the experiment) for 30 min before adding the cells. Sera from different times of immunization scheme (pre-immune, after 2nd and 3rd immunizations, respectively) were compared in order to know the number of immunization required to obtain neutralizing antibody response. These experiments showed that at least three immunizations are required as observed in figure 4.
]]>
CTLL-2 assays were also used to compare the effectiveness of two adjuvants on the development of a neutralizing anti-IL-15 response. In this case, different patterns were observed in the neutralization curves of sera from animals immunized with recombinant hIL-15 adjuvanted in Montanide® ISA 51 as compared to those of monkeys immunized with aluminum hydroxide as adjuvant (Figure 5). Specifically, animals immunized using Al(OH)3 showed similar neutralizing responses (Figure 5B). In correspondence, the values of neutralizing titers calculated by this assay for each animal were: 1:1600, 1:1600 and 1:3200 for animals immunized with Al(OH)3 as adjuvant and 1:400, 1:800 and 1:600 for those immunized with Montanide® ISA 51.
The specificity of inhibitory effect of serum to hIL- 15 was evaluated in presence of IL-2 (Figure 6). In this experiment only a specific antibody against IL-2 showed inhibitory effect on CTLL-2 proliferation induced by IL-2, none of the serum evaluated were able to neutralize IL-2 proliferative activity.
Finally, CTLL-2 assay allowed us to evaluate the neutralizing activity of antibodies or sera generated to hIL-15 against mIL-15. For this, the range of concentration was determined to get a satisfactory response curve for mIL-15. The linearity range was obtained from 390 pg/mL to 6.25 ng/mL. The 800 pg/mL concentration was selected to evaluate the neutralizing capacity of antibodies since it was near to the proliferation curve plateau (Figure 7).
Commercially available neutralizing antibodies anti-hIL-15 (MAB247 and MAB647) and serum from mouse immunized with hIL-15 were evaluated in the previously established conditions. In this experiment, we observed that neither the commercial antibodies anti-hIL-15 nor sera from mice immunized with hIL-15 inhibited the mIL-15. An anti-mIL-15 antibody was used as experimental control to evaluate the specifici-ty of the assay, which showed a dose-dependent inhibitory effect on mIL-15 (Figure 8 and Figure 9).
]]> DISCUSSION
It is essential to have optimized biological assays for preclinical studies of pharmaceutical molecules using cytokines as targets, which often use established cell lines that depend on the presence of particular cytokines for their continued growth and survival.
One of them, the CTLL-2 cell proliferation assay using the redox sensitive formazan MTT [17], is frequently used to determine IL-2 and IL-15 biological activity. In this paper, the CTLL-2 assay was adjusted to evaluate IL-15 inhibitors developed in our laboratory. We used a NIBSC standard to calibrate a laboratory standard reference and we tested the specificity of the assay using a neutralizing antibody against IL-15. CTLL-2 cells also proliferate in response to IL-2, in fact, the cells were maintained in culture medium supplemented with IL-2 before the experiments and several washes were required prior to performing the assay. The presence of neutralizing anti-IL-15 antibody in the assay showed that the proliferation of CTLL-2 cells was induced by IL-15. Also, the assay was demonstrated to have a good repeatability and reproducibility in a series of experiments as shown in the table.
To evaluate IL-15 antagonists developed in our laboratory, conditions such as sample volume, time of experiment and the order of samples application were established. In the case of the antagonist peptide P8 capable of binding to IL-15Rα [15], cells were pre-viously incubated with this peptide in order to give advantage to peptide/IL-15Rα binding because the affinity of IL-15/IL-15Rα complex is very high (10-11 M). This peptide exhibited an IC50 of 130 μM and showed a weak affinity to the target protein IL-15Rα compared to the high affinity IL-15/IL-15Rα complex. This is predictable because short peptides usually cannot hold the conformation needed for high-affinity biomolecular interaction. We chose an IL-15 dose of 300 pg/mL as set concentration, corresponding to a point near the proliferation curve plateau, in order to assess high-potency antagonists able to displace or inhibit IL-15/IL-15Rα binding.
The specificity of the effect of this peptide on IL-15 activity was determined in the presence of IL-2, a cytokine sharing certain functions with IL-15. It also has a similar structure and shares receptor β and γ subunits with IL-15, but, significantly, each cytokine has a specific alpha subunit [23]. This reason supports the specificity of the peptide that binds IL-15Rα for IL-15, as it was demonstrated in this work using CTLL-2 cell proliferation assay in the presence of IL-2. Furthermore, this result demonstrates that there is no toxicity associated to the P8 peptide on this cell line.
Another point of high interest was the applicability of this assay in the improvement and optimization of molecules derived from the P8 peptide. As previously reported, a more active peptide was identified with this assay, by studying specific single mutations on the P8 peptide [24].
In fact, the CTLL-2 proliferation assay was used to evaluate the sera from monkeys immunized with IL-15, in order to compare the immune response for each animal during the immunization scheme. A neutralizing response was only observed after the third immunization, providing the number of immunizations required to get a neutralizing response and its duration. These results are consistent to those previously describing that 3 or 4 immunizations per year were necessary to maintain a strong neutralizing antibody response [25].
Since modern vaccines based on recombinant antigens generally require adjuvants to generate adequate immune responses, either aluminum hydroxide or Montanide® ISA 51 were evaluated to induce a neutralizing antibody response to IL-15, to select the most effective adjuvant for an anti-IL-15 vaccine. Immunization with IL-15 formulated in each adjuvant generated antibodies neutralizing the native IL-15. Specifically, neutralizing antibody titers were higher in the group immunized with IL-15 on aluminum hydroxide, as shown in the CTLL-2 cell proliferation assay. This is also favored by the long experience on the use of aluminum hydroxide in humans, and its enhanced antibody responses [26]. Hence, we chose aluminum hydroxide as adjuvant for further immunizations with IL-15.
Furthermore, the specificity of sera from monkeys immunized with IL-15 in the presence of IL-2 was assessed. Both molecules are 43 % homologous in protein sequence but have a high structural homology. Nevertheless, it was shown that evaluated sera do not recognized human IL-2 and, therefore, do not inhibit the cytokine-induced proliferation. This remarkable result demonstrates the specificity of the antibodies for the immunized cytokine, in spite of both cytokines having the same receptor β and γ subunits and sharing some biological functions due to the redundant effect described for IL-15 and IL-2. The specificity of the neutralizing sera is essential to develop a vaccine targeting IL-15, since high titers of specific anti-cytokine antibodies induced by immunization could neutralize the local cytokine overexpression and inhibit its pathological effect [25, 27].
Finally, we optimized the conditions to evaluate the capacity of commercially available neutralizing antibodies and sera developed in mice against hIL-15 for mIL-15 neutralization, aimed to identify the possible use of murine models for preclinical evaluation of these IL-15 antagonists. CTLL-2 is a murine cell line; however, it was demonstrated by Eisenman et al. that these cells need more mIL-15 than hIL-15 to proliferate [28]. For current experiments, it was necessary to determine the range of doses to obtain a linear response. A dose of 800 pg/mL was set because it produces cell proliferation levels that support the evaluation of the effect of these antagonists. Neither the commercial neutralizing antibodies anti-hIL-15 nor sera against hIL-15 inhibited mIL-15. This represents a technical difficulty for the evaluation of neutralizing antibodies or the anti-hIL-15 vaccine in murine models. Any anti-cytokine vaccine requires antibodies generated through immunization also displaying neutralizing activity against the specific cytokine of the vaccinated animal species.
]]> In summary, our results demonstrate that the CTLL-2 cell proliferation assay is a very useful method to evaluate IL-15 antagonists. This assay can provide relevant information about antagonists’ activity, specificity and its possibility of evaluation in different animal models. Nevertheless, it requires further validation prior to its use as analytical tool on clinical trials.
ACKNOWLEDGEMENTS
The authors thank Prof. Elizabeth Díaz and Yolanda Gómez for their help in correcting language.
REFERENCES
1. Budagian V, Bulanova E, Paus R, Bulfone-Paus S. IL-15/IL-15 receptor biology: a guided tour through an expanding universe. Cytokine Growth Factor Rev. 2006;17(4):259-80.
2. Meazza R, Azzarone B, Orengo AM, Ferrini S. Role of common-gamma chain cytokines in NK cell development and function: perspectives for immunotherapy. J Biomed Biotechnol. 2011;2011:861920.
3. Kokaji AI, Hockley DL, Kane KP. IL-15 transpresentation augments CD8+ T cell activation and is required for optimal recall responses by central memory CD8+ T cells. J Immunol. 2008;180(7):4391-401.
4. Magyari L, Varszegi D, Kovesdi E, Sarlos P, Farago B, Javorhazy A, et al. Interleukins and interleukin receptors in rheumatoid arthritis: Research, diagnostics and clinical implications. World J Orthop. 2014;5(4):516-36.
5. Burska A, Boissinot M, Ponchel F. Cytokines as biomarkers in rheumatoid arthritis. Mediators of inflammation. 2014;2014:545493.
6. Yadav PK, Chen C, Liu Z. Potential role of NK cells in the pathogenesis of inflammatory bowel disease. J Biomed Biotechnol. 2011;2011:348530.
]]>7. Harris KM, Fasano A, Mann DL. Monocytes differentiated with IL-15 support Th17 and Th1 responses to wheat gliadin: implications for celiac disease. Clin Immunol. 2010;135(3):430-9.
8. Rashtak S, Murray JA. Review article: coeliac disease, new approaches to therapy. Aliment Pharmacol Ther. 2012;35(7):768-81.
9. McLInnes IB. Cytokine targeting in psoriasis and psoriatic arthritis: beyond TNFalpha. Ernst Schering Res Found Workshop. 2006(56):29-44.
10. Patterson KC, Hogarth K, Husain AN, Sperling AI, Niewold TB. The clinical and immunologic features of pulmonary fibrosis in sarcoidosis. Transl Res. 2012;160(5):321-31.
11. Waldmann TA, Conlon KC, Stewart DM, Worthy TA, Janik JE, Fleisher TA, et al. Phase 1 trial of IL-15 trans presentation blockade using humanized Mikbeta1 mAb in patients with T-cell large granular lymphocytic leukemia. Blood. 2013;121(3):476-84.
]]>12. Ferrari-Lacraz S, Zheng XX, Kim YS, Li Y, Maslinski W, Li XC, et al. An antagonist IL-15/Fc protein prevents costimulation blockade-resistant rejection. J Immunol. 2001;167(6):3478-85.
13. Villadsen LS, Schuurman J, Beurskens F, Dam TN, Dagnaes-Hansen F, Skov L, et al. Resolution of psoriasis upon blockade of IL-15 biological activity in a xenograft mouse model. J Clin Invest. 2003;112(10):1571-80.
14. Tinubu SA, Hakimi J, Kondas JA, Bailon P, Familletti PC, Spence C, et al. Humanized antibody directed to the IL-2 receptor beta-chain prolongs primate cardiac allograft survival. J Immunol. 1994;153(9):4330-8.
15. Santos A, Cabrales A, Reyes O, Gerónimo H, Rodríguez Y, Garay H, et al. Identification of an interleukin-15 antagonist peptide that binds to IL-15Ra. Biotecnol Apl. 2008; 25:320-24.
16. Giri JG, Ahdieh M, Eisenman J, Shanebeck K, Grabstein K, Kumaki S, et al. Utilization of the beta and gamma chains of the IL-2 receptor by the novel cytokine IL-15. EMBO J. 1994;13(12):2822-30.
]]>17. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65(1-2):55-63.
18. Scherliess R. The MTT assay as tool to evaluate and compare excipient toxicity in vitro on respiratory epithelial cells. Int J Pharm. 2011;411(1-2):98-105.
19. Wright JF. Bioassays and biological stability. Dev Biol Stand. 1999;97:73-80.
20. Mire-Sluis AR, Gerrard T, Das RG, Padilla A, Thorpe R. Biological assays: their role in the development and quality control of recombinant biological medicinal products. Biologicals. 1996;24(4):351-62.
21. Mire-Sluis AR, Page L, Thorpe R. Quantitative cell line based bioassays for human cytokines. J Immunol Methods. 1995;187(2):191-9.
]]>22. Santos A, Morera Y, Araña M, Ferrero J, Moro A, García J, et al. Obtaining Biologically Active IL-15 in Escherichia coli. Biotecnol Apl. 2000; 17: 221-224.
23. Giri JG, Kumaki S, Ahdieh M, Friend DJ, Loomis A, Shanebeck K, et al. Identification and cloning of a novel IL-15 binding protein that is structurally related to the alpha chain of the IL-2 receptor. EMBO J. 1995;14(15):3654-63.
24. Savio AS, Acosta OR, Perez HG, Alvarez YR, Chico A, Perez HG, et al. Enhancement of the inhibitory effect of an IL-15 antago¬nist peptide by alanine scanning. J Pept Sci. 2012;18(1):25-9.
25. Zagury D, Burny A, Gallo RC. Toward a new generation of vaccines: the anti-cytokine therapeutic vaccines. Proc Natl Acad Sci USA. 2001;98(14):8024-9.
26. Coffman RL, Sher A, Seder RA. Vaccine adjuvants: putting innate immunity to work. Immunity. 2010;33(4):492-503.
]]>27. Zagury D, Gallo RC. Anti-cytokine Ab immune therapy: present status and perspectives. Drug Discov Today. 2004;9(2):72-81.
28. Eisenman J, Ahdieh M, Beers C, Brasel K, Kennedy MK, Le T, et al. Interleukin-15 interactions with interleukin-15 receptor complexes: characterization and species specificity. Cytokine. 2002;20(3):121-9.
Received in February, 2014.
Accepted in December 2014.
Yunier Rodríguez-Álvarez. Departamento de Farmacéuticos, Dirección de Investigaciones Biomédicas, Centro de Ingeniería Genética y Biotecnología, CIGB. Ave. 31 entre 158 y 190, Cubanacán, Playa, CP 11600, La Habana, Cuba. E-mail: yunier.rodriguez@cigb.edu.cu.
]]>