ARTÍCULO ORIGINAL
Cardiac cellular actions of quebrachidine, an indole alkaloid isolated from Rauwolfia viridis Roem et Schult.
]]>
Acciones celulares cardíacas de la quebrachidina, un alcaloide indólico aislado de Rauwolfia viridis Roem et Schult.
Julio L. Álvarez,I Guy VassortII
IDoctor en Ciencias. Laboratorio de Electrofisiología. Instituto de Cardiología y Cirugía Cardiovascular. Ciudad de La Habana, Cuba. ]]> IIDoctor en Ciencias. Unité de Recherches de Physiopathologie Cardiovasculaire, U-637 INSERM. CHU Arnaud de Villeneuve, Montpellier, France.
INTRODUCTION: the search for new drugs with safer therapeutic profiles in Cardiology is still a need and natural products, particularly from plants, constitute an excellent source of new compounds.
OBJECTIVE: to study the cardiac cellular actions of quebrachidine an indole alkaloid, extracted from the roots of Rauwolfia viridis R et S, known as Quebrachidine, which is structurally related to the antiarrhythmics ajmaline and prajmaline.
METHODS: several complementary experimental approaches to evaluate the effects of quebrachidine on the electrophysiological and contractile properties of cardiac tissues and cells were used.
RESULTS: quebrachidine increased the ventricular fibrillation threshold in anaesthetized rabbits. It decreased the maximum rate of depolarization and increased the duration of the ventricular action potential in different species. These actions were accompanied by a positive inotropic effect over a broad concentration range and were consistent with the increase in Ca2+ currents recorded in single ventricular cardiomyocytes.
CONCLUSIONS: the present results demonstrate that quebrachidine keeps the antiarrhythmic profile of ajmaline and prajmaline but also demonstrates a net positive inotropic action on cardiac tissues predictive of better therapeutic safety margin. Our results suggest that ajmalan-like molecular structures could provide a sound basis for the search of effective antiarrhythmics with positive inotropic effect.
Key words: quebrachidine, ajmaline, prajmaline, indole alkaloids, Rauwolfia, antiarrhythmics, natural products.
Palabras clave: quebrachidina, ajmalina, prajmalina, alcaloides indólicos, Rauwolfia, antiarrítmicos, productos naturales.
]]>
INTRODUCTION
Cardiac arrhythmias are a leading cause of death in patients suffering from cardiac diseases and there is still a need to search for safe and efficient treatments.1 Recently, new therapeutic approaches have emerged but for most of them there is a long road to go before their successful and safe application.2
Despite the discouraging results with antiarrhythmic drugs in large randomized placebo-controlled clinical studies carried out late in the eighties and the early nineties,3-5 several compounds are still used in antiarrhythmic therapy alone or in combination with implantable cardioverter defibrillators6 and the search for new targets and more specific drugs still goes on.7 As for most of our life needs, natural products, particularly from plants, constitute an excellent source of new compounds that can often give substantial contribution to drug innovation by providing novel chemical structures (and/or mechanisms of action) with potential therapeutic properties. In this sense, the antiarrhythmic properties of indole alkaloids obtained from the roots of Rauwolfia serpentina, are known since the pioneering work of Arora and Madam.8 These authors were the first to characterize the pharmacological properties of ajmaline (ajmalan-17, 21-diol) later introduced in clinics by Kleinsorge.9 Since then, several structural derivatives having an ajmalan nucleus, have been characterized and employed in clinics.10
It is considered that these compounds exert their antiarrhythmic action by decreasing the fast inward Na+ current INa in a voltage- and use-dependent manner, thus slowing conduction and increasing the effective refractory period (for a review see 10). Among the ajmaline derivatives, prajmaline (prajmalium, 17R, 21a-dihydroxy-4-propylajmalinium) shows the longer half time and the higher potency of action, thus offering a low toxicity. Another peculiarity of prajmaline is that at therapeutic levels (» 100 ng/mL) the drug exerts no negative inotropic effect.10 At the cellular level, this could be explained by the fact that prajmaline increases the L-type Ca2+ current (ICaL) in single cardiac cells.11,12 This is an important property since the CAST studies3,4 have suggested that the increased mortality in patients treated with antiarrhythmics, could be well related to their negative inotropic effect, particularly flecainide and encainide. It is to note that ajmaline is currently used as a diagnostic challenge to disclose the full-blown electrocardiographic pattern of Brugada syndrome in family members to support the diagnosis of this lethal inherited cardiac arrhythmia.13
No quantitative structure-activity relationship exists for alkaloids with an ajmalan nucleus although early in the sixties, Bonati and co-workers exposed some fundamental results about ajmaline-like structures with antiarrhythmic actions.14,15 Other structurally related alkaloids like vincristine and vinblastine are known to induce positive chronotropic and inotropic effects and antiarrhythmic activity in cultured cardiac cells.16
With the aim to further characterize the cardiac cellular actions of indole alkaloids with an ajmalan nucleus, we performed experiments with quebrachidine (ajmalan-16-carboxylic acid-19,20 didehydro-1-demethyl-17-hydroxy-methyl ester) a molecule closely related to ajmaline but with a higher lipid solubility, comparable to that of prajmaline. The results show that quebrachidine retains the Na+ current inhibition properties of its congeners while it displays a significant positive inotropic effect.
METHODS ]]>
Isolation and purification of quebrachidine: quebrachidine (ajmalan-16-carboxylic acid-19,20 didehydro-1-demethyl-17-hydroxy-methyl ester; see figure 1) was kindly provided by Dr. J. Martínez. It was isolated and purified from the roots of Rauwolfia viridis Roem et Schult as previously described.17,18 At pH 7.4 the relative lipophylicity of quebrachidine/ajmaline is 4.24, determined by the n-octanol/water partition coefficients (sodium mono-di-phosphate/NaOH buffer) while that of prajmaline/ajmaline is 3.29 (Martínez J, personal communication). Quebrachidine was first dissolved at acidic pH to get a 10 mmol/L stock solution (stored at 4 ºC) and directly added to the physiological solution at the desired concentrations.
Electrocardiography in anaesthetized rabbits: rabbits were anaesthetized with sodium pentobarbital (30 mg/kg) and ventilated through a tracheotomy with a volume-adjusted respirator. The electrocardiographic leads I, II and III were simultaneously recorded. The left femoral vein was cannulated for infusion of solutions. The heart was exposed through a mid thoracotomy and a pair of platinum wires was fixed above the left ventricular epicardium for stimulation. The "ventricular fibrillation threshold" (VFT) was estimated using the following stimulation protocol: 30 pulses of 2 ms duration were applied, at a cycle length of 20 ms, with increasing intensities. The fibrillation threshold was considered to be the lowest current intensity (in mA) that induced six or more sustained ventricular responses once finished the stimulation. The value was only taken into account if it was replicated at least two times. We took advantage that with the rabbit model, ventricular fibrillation was spontaneously resumed in more than 90 % of trials. Animals were allowed to stabilize for 30 minutes before beginning the experiments.
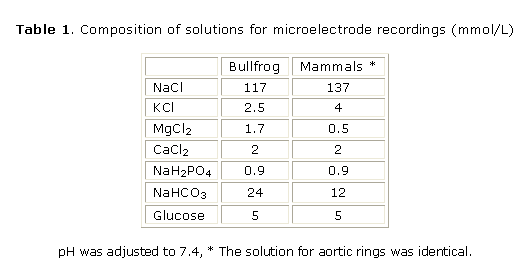
Electrophysiological and mechanical recordings in multicellular cardiac preparations: mammals (rat, rabbit and guinea pig) were anaesthetized with sodium pentobarbital (30 mg/kg) and bullfrogs (Rana catesbeiana) were decapitated and pithed. The hearts were rapidly removed and dissected in a well oxygenated physiological solution at room temperature (21-23 °C; see Table 1 for composition of solutions). Action potentials using high-resistance (10-20 MW) microelectrodes and contractions using a force transducer and stainless steel hooks were recorded from right ventricular papillary muscles (or from small frog ventricular strips) fixed to the bottom of a 2.5 mL recording chamber continuously perfused (10 mL/min; 35 °C or room temperature for frogs preparations) as previously described19. Stimulation (2 ms, twice the threshold) was achieved by field electrodes and the stimulation frequency was 75/min and 12/min for mammalian and frog preparations respectively.
]]>
Patch-clamp experiments on isolated cardiomyocytes
a) Cell dissociation: experiments were carried out on single atrial and ventricular cells dissociated from bullfrog hearts and on single ventricular cardiomyocytes form rat, rabbit and guinea-pig hearts. Frog cardiomyocytes were enzymatically dissociated according to the method developed in our laboratories.20,21 Cardiomyocytes from mammalian hearts were enzymatically isolated according to Alvarez et al.12 (see Table 2 for the composition of solutions). Yields of viable elongated cells were 40-60 % for mammalian hearts and > 80 % for frog hearts. Isolated mammalian cardiomyocytes were kept in physiological solution (Ca2+ = 1 mmol/L) at room temperature (21-23 EC) and used within 6-8 hours. Frog cardiomyocytes were placed in a refrigerator (10 EC) and could be used for as long as 24 hours.
b) Recording of ionic currents: the methods for whole-cell patch-clamp recording were essentially the same as described before.12,20 Extracellular solutions contained 20 mmol/L CsCl (instead of KCl; see table 2) to inhibit K+ currents. For recording Ca2+ currents tetrodotoxin (TTX) was used to inhibit the Na+ current (INa) at 3 µmol/L for frog cells and 50 µmol/L for mammalian cells. When INa was recorded (only in frog ventricular cardiomyocytes) the extracellular solution contained 50 % NaCl (substituted by choline chloride + 10 µmol/L atropine) and 3mmol/L CoCl2 to block the L-type Ca2+ current (ICaL). Patch-clamp pipettes had resistances of 2 - 3 MW for bullfrog cardiomyocytes or 0.9 - 1 MW for mammalian cardiomyocytes and were filled with a solution containing 120 mmol/L CsCl to ensure complete blockade of K+ currents (Table 2). Pulse generation and data acquisition were done using a computer and the ACQUIS1 software (version 2.0; CNRS License; France) with a LabMaster DMA interface (Scientific Solutions; Solon, OH, USA). Membrane capacitance (Cm) was estimated by applying 2-mV, 20-ms hyperpolarizing voltage-clamp pulses from the holding potential. Capacitive spikes were fitted to a single exponential and Cm was calculated according to:
Cm= tm . I0 / Vm (1 - Iss / I0) ]]>
where tm is the membrane time constant, I0 is the peak capacitive spike, Iss the steady state current at the end of the 20 ms pulse and Vm = 2 mV. Ionic currents were normalized to Cm to obtain current density (pA/pF).
The holding membrane potential was set at -100 mV in frog cardiomyocytes and -80 mV in mammalian ones. For routine monitoring of ICaL the membrane was depolarized to 0 mV during 200 ms every 4 s. The T-type Ca-current (ICaT) was only studied in frog atrial cells and was routinely evoked by a 200-ms depolarization to -50 mV where no interference with ICaL exists.11,19,20 Recordings of INa were accomplished in frog ventricular cells with 50 ms pulses to 0 mV every 4 s. Ca+ and Na+ currents to voltage relationships and their respective availability curves were estimated according to standardized pulse protocols.11,19,20 All patch-clamp experiments were conducted at room temperature (21-23 EC).
Force measurements in aortic rabbit rings: Abdominal aortic rings, about 3-mm width were dissected from rabbits and rats under animal anaesthesia, fixed to the bottom of a 3-mL perfusion chamber and to a force transducer with stainless steel hooks and perfused (10 mL/min) with physiological solution (table 1) at 35EC. A resting tension of 800 mg was applied and the rings were stabilized for one hour in these conditions. Contractions were induced by a solution with high K (isotonic, 140 mmol/L, without endothelial activity) or with norepinephrine (10 µmol/L).
Results were analyzed by a paired Student's t-test and are expressed as means and standard error of the means. They were considered to be statistically significant at p< 0.05.
RESULTS
Effects of quebrachidine on Ventricular Fibrillation Threshold (VFT): VFT in anaesthetized rabbits was estimated as described in Methods. In control condition VFT was 30.4 ± 4.2 mA (N = 8). In the absence of pharmacokinetic data for quebrachidine, we decided to test concentrations of 0.3 and 3 mg/kg (infused consecutively with an interval of 15 min) which roughly correspond to circulating concentrations of 1 - 2 and 30 - 60 µmol/L. VFT was significantly (p < 0.05) increased by 55 ± 12 % at 0.3 mg/kg and 62 ± 18 % at 3 mg/kg respectively (N= 8). The RR interval, however, was not significantly modified. From a control value of 260 ± 12 ms it changed to 268 ± 13 ms at 0.3 mg/kg and to 283 ± 20 ms at 3 mg/kg. ]]>
Effects of quebrachidine on action potential and force of contraction: action potential characteristics under control condition are summarized in table 3. Figure 2 (A - D) summarizes the effects of different quebrachidine concentrations on maximum rate of depolarization (dV/dt), action potential duration (measured at 20 % and 80 % of repolarization, D20 and D80, respectively) and force of contraction of frog ventricular strips and mammalian right ventricular papillary muscles. Quebrachidine decreased dV/dt in a concentration-dependent manner. Experimental data were fitted to a Hill function and estimated IC50 for dV/dt inhibition were 0.8, 2.8, 29 and 9.8 µmol/L for frog, rat, guinea pig and rabbit, respectively. Figure 2 also shows that quebrachidine increased D20 and D80 over the whole concentration range studied in the different ventricular preparations although without a clear-cut concentration-dependency. However, maximal increases in D20 and D80 were obtained at 1 µmol/L concentration in all species except frog ventricular strips in which maximal increase in D80 was obtained at 0.01 µmol/L. Quebrachidine induced a clear-cut positive inotropic effect in frog, guinea pig and rabbit ventricular preparations with maximal increases in force of contraction at 1 - 10 µmol/L. In rat ventricle however, the positive inotropic effect was noticeably only at 0.001 µmol/L. In four rabbit papillary muscles, the positive inotropic effect of 10 µmol/L quebrachidine was additive to that of 1 µmol/L isoproterenol (b-adrenergic agonist) or 1 µmol/L of the "Ca2+-agonist" BAY K 8644 (not shown). This action of quebrachidine was not prevent by propranolol (1 µmol/L) or prazosin (1 µmol/L). Resting potential and overshoot of action potential were not affected by quebrachidine at any concentration in any of the studied species. All effects of quebrachidine on action potential and contraction were readily reverted upon washout with control physiological solution.
Effects of quebrachidine on Na+ current of single bullfrog ventricular cardiomyocytes: in an attempt to obtain reliable voltage control during the flow of INa, experiments were performed only on frog ventricular cardiomyocytes which display a low INa density in low-Na+ extracellular solution11 (see Methods). Under these conditions peak inward INa was maximal at -20 mV with a density of 7.1 ± 0.6 pA/pF and a time for half inactivation (t50%) of 3.3 ± 0.2 ms (N= 8). Quebrachidine induced a concentration-dependent decrease in INa with an IC50 of 8.3 µmol/L (Fig. 3A). However, t50% was not significantly affected. The quebrachidine-induced decrease in INa was both voltage- and frequency-dependent. Voltage-dependent block was assessed at 10 µmol/L concentration. Current-to-voltage relationships and availability curves of INa were shifted by » 10 mV in the hyperpolarizing direction (Fig. 3 B and C). Potential for half-availability was shifted by 10.5 ± 1.8 mV in the hyperpolarizing direction and the slope factor increased from 4.67 ± 0.3 mV to 6.54 ± 0.2 mV (N= 8). In partially depolarized cells (holding potential of -70 mV) on which different quebrachidine concentrations were applied, the IC50 for INa inhibition was decreased to 0.65 µmol/L. As mentioned above, INa block by quebrachidine (10 µmol/L) was also frequency-dependent (Fig. 3D) with a small tonic or first pulse decrease of INa of 15.6 ± 6.2 % of control (. 28 % of total block) and a fast use-dependent decrease which represent an additional decrease of 40.2 ± 2.2 % (» 72 % of total block) at a holding potential of -100 mV. The effects of quebrachidine on INa were reverted upon washout with normal physiological solution.
Effects of quebrachidine on Ca2+ currents of single cardiomyocytes: we have previously reported that when a frog atrial (but not ventricular) cell is depolarized from a holding potential of - 100 mV, two distinct Ca currents can be evoked, ICaT and ICaL.11,20,21 Their activation characteristics permitted us to monitor changes in peak ICaT without significant contamination of ICaL. Since there were no differences in membrane capacitance (Cm) and ICaL density in atrial and ventricular cells results for ICaL were pooled. No attempts were done to check for ICaT in guinea pig and rabbit ventricular myocytes since in our hands, less than 30 % of these cells exhibited both ICaL and ICaT. Table 4 summarizes the characteristics of calcium currents under control conditions in the four species studied and figure 4 shows that quebrachidine induced an increase in both ICaT and ICaL in bullfrog cardiomyocytes and in ICaL in guinea pig and rabbit ventricular myocytes over a broad concentration range.
Quebrachidine was less effective on ICaL in rat ventricular myocytes. The effects of quebrachidine on ICaL were voltage-dependent since current-to-voltage relationships and availability curves for Ca2+ currents were shifted to more negative potentials. At 10 µmol/L concentration, potential for half availability of ICaL was shifted by about 5 mV in the hyperpolarizing direction in all species studied except rat ventricular myocytes in which it was not significantly modified (5.8 ± 0.9 mV in frog; 5.2 ± 1.0 mV in guinea pig and 4.7 ± 0.8 mV in rabbit; N³ 4). Times for half-inactivation of Ca2+ currents were, however, not modified by quebrachidine. The effects of quebrachidine on Ca2+ currents were reverted upon washout with normal physiological solution. ]]>
Effects of quebrachidine on smooth muscle contractile activity: Quebrachidine had no effect on KCl- and norepinephrine-induced contraction of rat aortic rings at any of the concentrations studied (N= 5). However, in rabbit aortic rings, quebrachidine induced a relaxing effect of KCl-induced contraction which was not concentration-dependent. At quebrachidine concentrations of 10, 30 and 100 µmol/L aortic contraction was significantly reduced to 65 ± 12 %, 56 ± 18 % and 64 ± 14 % of control (N= 5). No effects of quebrachidine were observed on the norepinephrine-induced contraction of rabbit aortic rings.
DISCUSSION
The main conclusion of this paper is that quebrachidine, like its congeners ajmaline and prajmaline displays a Class 1 antiarrhythmic action by inhibiting in a concentration-, voltage- and use-dependent manner the fast inward Na+ current INa. Moreover, like prajmaline, quebrachidine induces a positive inotropic effect at concentrations that significantly decreased INa. This effect is be attributable to an increase in Ca2+ current and could counterbalance the negative inotropy generally seen with compounds that reduce the intracellular Na+ concentration by inhibiting INa.
Quebrachidine is one and three orders of magnitude less potent than ajmaline and prajmaline, respectively in inhibiting INa with corresponding IC50s in bullfrog ventricular cells of 8.3 µmol/L (present results), 0.7 µmol/L and 0.01 µmol/L.10,11 It has been concluded that the difference in potency between ajmaline and prajmaline could be related to the higher liposolubility of prajmaline.10 The contrary holds for quebrachidine which is more liposoluble than prajmaline but much less potent on INa. This makes difficult to draw any conclusion from a (quantitative) structure-activity relationship analysis. At present, there is no structural data of the possible receptor(s) site(s) for quebrachidine within the Na+ channel that could shed some light to the mode of action of quebrachidine and other ondole alkaloids at the molecular level. However, the voltage- and strong use-dependent quebrachidine block of INa suggests that like ajmaline and prajmaline, quebrachidine is an inactivated state channel blocker.10-12 This characteristic inhibitory action on INa, together with the relatively high IC50 for normally polarized cells, suggests that quebrachidine could be an antiarrhythmic with a good safety profile (less side effects). More preclinical trials are of course needed to confirm this assertion. The inhibitory action of quebrachidine on dV/dt (an indirect estimate of INa) of action potential of different mammalian species indicate that it is a Na+ channel blocker. However, the estimated IC50 were variable: 2.8, 29 and 9.8 µmol/L for rat, guinea pig and rabbit, respectively and were inversely related to the dV/dt value under control condition (table 3) suggesting that INa inhibition could be related to Na+ channel density. The difference between IC50s obtained for INa in single bullfrog cardiomyocytes (8.3 µmol/L) and dV/dt in bullfrog ventricular strips (0.8 µmol/L) could be easily explained by the voltage-dependent action of quebrachidine since the resting membrane potential was more negative in single cells (holding potential of -100 mV) than in bullfrog ventricular strips (» -85 mV). In fact, when single bullfrog cardiomyocytes were depolarized (holding potential= -70 mV) the IC50 decreased to 0.65 µmol/L a value that is fully comparable with the IC50 obtained in ventricular strips.
Action potential duration was increased by quebrachidine in the four species we studied. Particularly, action potential duration at the plateau level (D20) was markedly increased by this indole alkaloid. This effect can be well explained by the increase in Ca2+ currents under the action of quebrachidine (see below). However, actions on other repolarizing currents (e.g. decrease in K+ currents) can not at present be ruled out. At concentrations over 10 µmol/L both ajmaline and prajmaline are reported to inhibit a delayed outward rectifier K+ current in nerve22 as well as the transient outward current Ito in rat ventricular cardiomyocytes (Alvarez JL; unpublished results).
Perhaps the most interesting result is that quebrachidine, together with its INa inhibitory action, displayed a net positive inotropic effect in bullfrog, guinea pig and rabbit ventricular preparations. A less marked effect was observed in rat. The positive inotropic effect is similar to that previously reported for prajmaline10-12 and seems to be independent of adrenergic receptor activation since it was not prevented by propranolol or prazosin and was additive to the positive inotropic effect of isoproterenol. While this action can be explained by an increase in Ca2+ currents (see below) the receptor site within the Ca2+ channels need to be elucidated. Our results suggest that quebrachidine does not interact with the dihydropyridine receptor site since its positive inotropic action is additive to that of BAY K 8644 a dihydropyridine with "Ca2+ agonist" properties. In fact, quebrachidine does not interact with the dihydropyridine (PN 200-110) or fenilalkylamine (D 888) sites within the L-type Ca2+ channel (Escande D, personal communication).
Nevertheless, in sharp contrast with most antiarrhythmics (for review see reference 10), quebrachidine increases both T- and L-type Ca2+ currents, and like prajmaline11,12 this action was voltage-dependent thus sharing similarities in their mechanisms of action with dihydropyridine Ca2+ agonists.23 However, inactivation time courses of ICaT and ICaL were not affected by quebrachidine. While the increases in Ca2+ currents were large enough to account for the positive inotropic action of this alkaloid, we can not rule out interactions of this molecule with other structures implicated in the control of the excitation-contraction coupling in the cardiac cell (e.g. the sarcoplasmic reticulum Ca2+ channel) that could contribute to the positive inotropic effect. No attempts were done to quantitatively correlate the positive inotropic effect with the increase in ICaL in different species. It is to note however, that where an increase in ICaL was found a consistent positive inotropic effect was evident. In this sense it is to note that in rat ventricle where the positive inotropic effect was weak or absent, there was almost no effect on ICaL. ]]>
Another interesting property of quebrachidine is its poor effect on vascular smooth muscle. No effect was observed on rat aortic rings while a limited relaxing action was achieved in rabbit aortic rings only at high concentrations.To conclude, quebrachidine is an interesting molecule since its electrophysiological profile is similar to that of the well-known and structurally related antiarrhythmics ajmaline and prajmaline while its significant positive inotropic effect in cardiac tissues suggest that it could be devoid of the adverse side-effects of most antiarrhythmic drugs. Our results also confirm that indole alkaloids from Rauwolfia sp. constitute an excellent source of new compounds with potential therapeutic properties.
ACKNOWLEDGMENTS
We wish to express our gratitude to Lic. M. Arpa and Lic. G. Garrido who collaborated in some preliminary experiments and to Dr. J. Martínez for providing quebrachidine. The skillful technical assistance of M. Carrabeo with the anaesthetized rabbits is greatly acknowledged.
]]>
REFERENCES
1. Jordan PN, Christini DJ. Therapies for ventricular cardiac arrhythmias. Critical Rev Biomed Engin. 2005;33:557-604.
2. Cho HC, Marbán E. Biological therapies for cardiac arrhythmias: can genes and cells replace drugs and devices? Circ Res. 2010;106:674-85.
3. The CAST Investigators I. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med. 1989; 321:406-12.
4. The CAST Investigators II. Effect of the antiarrhythmic agent moricizine on survival after myocardial infarction. N Engl J Med. 1992;327:227-33.
5. Waldo AL, Camm AJ, deRuyter H, Friedman PL, MacNeil DJ, Pauls JF, et al. Effect of d-sotalol on mortality in patients with left ventricular dysfunction after recent and remote myocardial infarction. The SWORD Investigators. Survival With Oral d-Sotalol. Lancet. 1996;348:7-12.
6. Nattel S, Carlsson L. Innovative approaches to anti-arrhythmic drug therapy. Nature Rev Drug Discovery. 2006;5:1034-49.
7. Roden DM, Kannankeril PJ, Darbar D. Arrhythmia pharmacogenomics: methodological considerations. Curr Pharm Des. 2009;15:3734-41.
8. Arora RB, Madan BR. Antiarrhythmics VI. Ajmaline and serpentine in experimental cardiac arrhythmias. J Pharmacol Exp Therap. 1956;117:62-7.
9. Kleinsorge H. Klinische Untersuchungen über die Wirkungsweise des Rauwolfia-alkaloids bei Herzrhythmusstörungen insbesondere der Extrasystolie. Med Clin. 1959;54:409-16.
10. Alvarez JL, Vassort G. Prajmalium. Cardiovasc Drugs Revs. 1993;11:385-410.
11. ________________. Dual action of prajmalium on the Ca-current in frog isolated cardiomyocytes. J Mol Cell Cardiol. 1991;23:627-38.
12. Alvarez JL, Rubio LS, Garrido G. Vassort G. Prajmalium, an antiarrhythmic with positive inotropic effect: Mechanism of action in rabbit single cardiomyocytes. J Cardiovasc Pharmacol. 1992;20:43-9.
13. Yap YG, Behr ER, Camm AJ. Drug-induced Brugada syndrome. Europace. 2009;11:989-94.
14. Bonati A, Bocchia A. Derivati dell'ajmalina. Nota I. Il Farmaco Ed Sc. 1963;18:84-93.
15. Bonati A, Bombardelli E. Derivati dell'ajmalina. Nota II. Il Farmaco Ed Sc. 1963;18:851-63.
16. Lampidis TJ, Kolonias D, Savaraj N, Rubin RW. Cardiostimulatory and antiarrhythmic activity of tubulin-binding agents. Proc Natl Acad Sci USA. 1992;89:1256-60.
17. Martínez JA, Rodríguez MR, Dehesa MA, Machuá M. Alcaloides de Rauwolfia. IX. Alcaloide de la corteza de la raíz de R. viridis Roem. et Schult. Rev Cubana Quim. 1988;4:43-51.
18. Martínez JA, Bello A, Montero O, Fuente V. Bases débiles minoritarias de la raíz de Rauwolfia viridis Roem et Schult. Rev Cubana Farm. 1997;31:188-91.
19. Alvarez JL, Dorticós F, Morlans J. Changes in electrical and mechanical activities of rabbit papillary muscle during hypoxic perfusion. J Physiol (Paris). 1981;77:807-12.
20. Alvarez JL, Vassort G. Properties of the low-threshold Ca current in frog atrial cardiomyocytes. A comparison with the high-threshold Ca current. J Gen Physiol. 1992;100:519-45.
21. Alvarez JL, Rubio L, Vassort G. Facilitation of T-type Ca- current in bullfrog atrial cells: voltage-dependent relief of a G-protein inhibitory tone. J Physiol (L). 1996;491(2):321-34.
22. Khodorov BI, Zhaborovskaya LD. Blockade of sodium and potassium currents in the node of Ranvier by ajmaline and N-propyl ajmaline. Gen Physiol Biophys. 1983;2:233-68.
23. Bechem M, Schramm M. Electrophysiology of dihydropyridine calcium agonists. In: Morad M, editor. The calcium channel: structure, function and implications; 1988. p. 63-70.
Recibido: 26 de abril de 2010. ]]> Aprobado: 23 de junio de 2010.
Dr. Julio L. Álvarez. Laboratorio de Electrofisiología. Instituto de Cardiología y Cirugía Cardiovascular. Edificio Asclepios. 17 y Paseo, Vedado, CP 10400. Ciudad de La Habana, Cuba. Teléf.: 8360831. E-mail: alvarezj@infomed.sld.cu ]]>