Oral monitoring of a pediatric patient during chemotherapy treatment
Monitoreo oral de un paciente pediátrico durante el tratamiento de quimioterapia
Isabella Lima Arrais Ribeiro,I Ana Maria Gondim Valença,II Paulo Rogério Ferreti Bonan,II Fabíola Galbiatti de Carvalho CarloII
I Departament of Statistic, Federal University of Paraiba, João Pessoa, Brazil.
II Department of Clinic and Social Dentistry, Federal University of Paraiba, João Pessoa, Brazil.
]]>
Oral side effects must be expected during cancer treatment on pediatric patients. Monitoring side effects on oral cavity of antineoplastic therapy is desirable but sometimes performed without criteria. The purpose of this article is to describe an oral monitoring in an male with Hodgkin lymphoma during chemotherapy treatment using an Oral Assessment Guide. An 11-yr-old male was assisted during all treatment of chemotherapy against Hodgkin's lymphoma in the dental sector of a hospital of reference of João Pessoa, Paraíba, Brazil. The Oral Assessment Guide was applied by a calibrate examiner and was observed the emergence of ulcerative lesions on the labial mucosa emerged on two different periods (D15- primary cycle; D15-second cycle) and the major values of oral mucositis were verified in D1 e D15 periods of second cycle of chemotherapy. Monitoring oral side effects during antineoplastic therapy could prevent severe oral complications and avoid to associate systemic complications.
Key words: Hodgkin lymphoma, pediatric cancer, oral mucositis.
Los efectos secundarios orales se manifiestan durante el tratamiento del cáncer en los pacientes pediátricos. El monitoreo de los efectos secundarios en la cavidad oral de la terapia antineoplásica es deseable, pero a veces se realiza sin criterios. El propósito de este artículo es describir un monitoreo oral en un niño con linfoma de Hodgkin durante el tratamiento de quimioterapia, empleando una guía de evaluación oral. Un niño de 11 años de edad fue asistido durante todo el tratamiento de quimioterapia contra un linfoma de Hodgkin en el sector odontológico de un hospital de referencia de João Pessoa, Paraíba, Brasil. La guía de evaluación oral fue aplicada por un examinador calibrado. Se observó la aparición de lesiones ulcerosas en la mucosa labial que aparecieron en dos períodos diferentes (D15- primer ciclo; D15-segundo ciclo) y los valores más altos de la mucositis oral se verificaron en períodos D1 e D15 del segundo ciclo de quimioterapia. El seguimiento de los efectos secundarios orales durante la terapia antineoplásica podría prevenir las complicaciones orales graves y evitar complicaciones sistémicas asociadas.
Palabras clave: linfoma de Hodgkin, cáncer pediátrico, mucositis oral.
INTRODUCTION
Patients with leukemia or lymphoma undergoing chemotherapy frequently develop alterations of the oral cavity.1 A direct effect of the drug on the oral mucosa or the associated myelosuppressive action are the two main mechanisms associated with side effects such as mucositis, dry mouth, bleeding, bone alterations, teeth disorders, pain and infections.1-3 An ongoing oral assessment must be performed to promote the early identification of oral complications.
A previous instrument used developed by Nebraska University, the Oral Assessment Guide (OAG) showed as a comprehensive, accurate tool for assessing oral integrity in children with cancer during and after chemotherapy.1 The OAG is a guide of oral evaluation initially proposed by Eilers, Peterson and Berger in 19884 and modified for Cheng et al. (2004)5 to assess changes in the oral mucosa resulting from antineoplastic treatment using chemotherapy. This instrument allows to evaluate 8 items, according to the scales of impaired oral health, with assigned values 1-3 for each item, as follows: 1 for conditions where it is checked normality; 2, for the verification of mild to moderate changes in relation to the epithelial integrity or function and 3 for a severe impairment. At the end, the total checked for mucositis varies from 8 to 24, without a cutoff between these values for the estimation of mucositis.
]]> The purpose of this article is to describe an oral monitoring in an 11 year-old male with Hodgkin lymphoma during chemotherapy treatment using a modified Oral Assessment Guide.
DESCRIPTION OF CASE
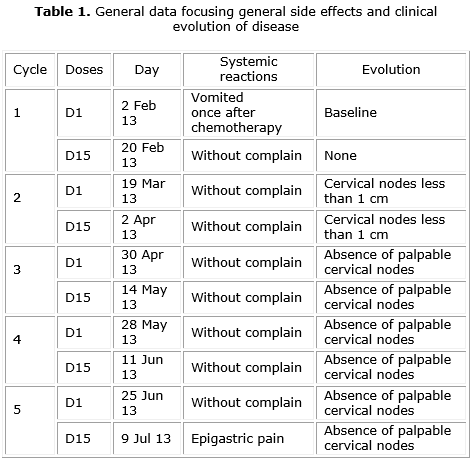
An 11 year-old, male, non caucasian patient was referred to reference hospital on Paraiba state, Brazil, with initial complaint of a swelling on the right supraclavicular with progressive growth. Lymph node biopsy was taken revealing Hodgkin Lymphoma with positiveness to EBV and sparse immunostaing to CD30. Therapy with chemotherapy administration was instituted following the protocol: 26 mg of Doxorubicin- IV for 30 min (D1 and D15), 10 mg of Bleomycin - IV for 30 min (D1 and D15); 6 mg of Vimblastine (IV push) (D1 and D15); 375 mg of Dacarbazine - IV for 120 min (D1 and D15) with a total of 5 cycles. Moreover, it was prescribed Sulfamethoxazole + Trimethoprim suspension orally for 12/12 h, 3 times per week in continuous use. The general recorder of patient evolution during chemotherapy is described on table 1.
At diagnosis, we evaluated the oral cavity and DMFT was equal to 3, due to the presence of two teeth restored without caries (permanent maxillary right second molar and primary mandibular right first molar), with restored before the antineoplastic treatment and a restored tooth with dental caries (primary mandibular right second molar). Need to improve daily oral hygiene was percept, which has been shown and monitored throughout treatment.
During the treatment, OAG was applied by a calibrate examiner during D1 and D15, inside the cycles, and the obtained values are described on table 2. On this index, we assigned a value of 1 to 3, according to the progressive morbidity.
During the assessments, ulcerative lesions on the labial mucosa emerged on two different periods (12 Mar 13 and 2 Apr 13) (Fig.). These mucositis events were coincident with higher levels of OAG. Palliative treatment for these two episodes of mucositis was the application of mucolaser (ECCO Fibras e Dispositivos/Brazil; n/s – 040401; model – BM0004A; Power – 120 Mw; Doses – 7 J/cm2; Time doses – 33 sec.) with clinical improvement.
]]>
DISCUSSION
Oral Assessment Guide and oral hygiene care regimen could be clinically useful in improving oral assessment and hygiene in children with cancer during and after chemotherapy.1 Oral complications could be inflammatory and ulcerative conditions and meant each category achieved a score of 2 or a total score greater than 9.1 In this case, mucositis events were coincident with higher levels of OAG. Oral vigilance is desirable to avoid major complications. To illustrate this, an interview and oral examination was conducted on 150 pediatric cancer patients receiving standard dose chemotherapy showed a relative high prevalence of chemotherapy-induced oral mucositis and oral infections. The chemotherapeutic antimetabolites were the most frequently associated with oral complications than other types of chemotherapy.2
The mucositis induced by chemotherapy occurs because during the primary damage response phase is initiate a series of interacting biological events as the activation of a number transcription factors, such as NF-KB, Wnt, P-53 and their associated cronical pathways.5-7 Each cycle of chemotherapy incurs in increased risk to develop mucositis.8-10
Thus, monitoring the oral health of the patient, and the use of palliative treatments are needed to prevent injuries that might compromise the pediatric patients in antineoplastic therapy of cancer.6-8 Such monitoring can be done using the OAG, which allows the monitoring of different locations where mucositis can occur in the oral cavity.11,12
Monitoring oral side effects during antineoplastic therapy could prevent severe oral complications and avoid to associate systemic complications and we recommended OAG, which proved to be efficient on detect mucositis upset and other oral alterations on oral cavity caused by chemotherapy.
BIBLIOGRAPHIC REFERENCES
1. Chen CF, Wang RH, Cheng SN, Chang YC. Assessment of chemotherapy-induced oral complications in children with cancer. J Pediatr Oncol Nurs. 2004;21(1):33-9.
2. Jensen SB, Peterson DE. Oral mucosal injury caused by cancer therapies: current management and new frontiers in research. Journal of Oral Pathology & Medicine. 2014;43(2):81-90.
3. Lopes IA, Nogueira DN, Lopes IA. Oral manifestations of chemotherapy in children from a cancer treatment center. 2012;12(1):113-9.
4. Eilers J, Berger AM, Peterson MC. Development, testing, and application of the oral assessment. Oncol Nurs Forum. 1988;15:325-30.
5. Cheng KKF, Chang AM, Yuenc MP. Prevention of oral mucositis in paediatric patients treated with chemotherapy: a randomised crossover trial comparing two protocols of oral care. European Journal of Cancer. 2004;40:1208–16.
6. Dhillon J, Kalra GK, Mathur V. Oral health in children with leucemia. Indian Journal of Palliative Care. 2012;18(1):12-8.
7. Kutob AF, Gue S, Revesz T, Logan RM, Keefe D. Prevention of oral mucositis in children receiving cancer therapy: A systematic review and evidence-based analysis. 2013;49(2):102-7.
8. Barbosa AM, Ribeiro DM, Caldo-Teixeira AS. Conhecimentos e práticas em saúde bucal com crianças hospitalizadas com câncer. Ciência & Saúde Coletiva. 2010;15(1):1113-22.
9. Sonis ST. The Epidemiology and Risk Assessment of Mucositis. Oral mucositis. 2012:15-24.
10. Sonis ST. Mucositis: The impact, biology and therapeutic opportunities of oral mucositis. Oral Oncol. 2009;45(12):1015-20.
11. Cheng KKF, Lee V, Li CH, Goggins W, Thompson DR, Yuen HL, et al. Incidence and risk factors of oral mucositis in paediatric and adolescent patients undergoing chemotherapy. Oral Oncology. 2011;47(3):153–62.
12. Valéra M-C, Noirrit-Esclassan M, Pasquet M, Vaysse F. Oral complications and dental care in children with acute lymphoblastic leukaemia. Journal of Pathology & Medicine. 2014;43(8):122-6.
Recibido: 10 de mayo de 2014.
Aprobado: 4 de enero de 2015.
Isabella Lima Arrais Ribeiro. Posgraduate program in Decision Models and Health, Departament of Statistic. University city s/n. João Pessoa, Brazil. ]]>
Correo electrónico: isabella_arrais@yahoo.com ]]>