Gastroprotective effects of D-002 (beeswax alcohols) and Lyprinol® on experimentally-induced gastric ulceration in rats
Efectos gastroprotectores del D-002 (alcoholes de cera de abejas) y el Lyprinol® sobre la úlcera gástrica experimental en ratas
MSc. Yazmin Ravelo Calzado, Dra. C. Vivian Molina Cuevas, Dra C. Daisy Carbajal Quintana, Dra. C. Rosa Más Ferreiro, Dra C. Zullyt Zamora Rodríguez
Centro de Productos Naturales, (CPN), Centro de Investigaciones Científicas (CNIC). La Habana, Cuba.
]]>
ABSTRACT
Introduction: D-002, a mixture of beeswax alcohols, has been effective in osteoarthritis models and for reducing osteoarthritis symptoms. Unlike the classic anti-inflammatory drugs, D-002 elicits gastroprotective rather than gastrotoxic effects. Lyprinol, used for ameliorating inflammation and arthritic symptoms, improves gastrointestinal dysfunction symptoms in osteoarthritis subjects. Both D-002 and Lyprinol inhibit cyclooxygenase and 5?lipoxygenase activities, and have been similarly effective for reducing inflammation experimentally.
Objective: to compare the effects of D-002 and Lyprinol on gastric mucosa of normal and experimentally-induced ulcer rats.
Methods: ulcer indexes were measured in normal rats and in rats with ethanol or pylorus ligation-induced ulcers, in which gastric volume and mucus secretion were also measured. Normal rats were randomized into a vehicle control, one acetic salicylic acid (150 mg/kg), three D-002, three Lyprinol groups; rats with ethanol-ulcers into a vehicle control, three D-002 and three Lyprinol-treated groups; and the experiment on pylorus ligation included a negative control and eight pylorus-ligated groups: one vehicle control, three D-002, three Lyprinol, one omeprazole 10 mg/kg. In all cases, D-002 and Lyprinol (50, 200 and 400 mg/kg) were given orally.
Results: unlike D-002 and Lyprinol (50-400 mg/kg), acetic salicylic acid increased ulcer indexes and the incidence of ulcers versus the vehicle control. Single oral doses of D-002 (50-400 mg/kg) or Lyprinol (200 and 400 mg/kg) decreased significantly (p<0.01) and in a similar way ulcer indexes versus the ethanol-positive control. D-002 and Lyprinol (50-400 mg/kg) lowered significantly (p<0.01) and comparably ulcer indexes in rats with pylorus ligation versus the positive controls. D-002 (200 and 400 mg/kg) decreased gastric volume and increased gastric mucus secretion versus the positive control whereas only Lyprinol 400 mg/kg increased the gastric mucus secretion but without modifying the gastric volume. Omeprazole significantly reduced ulcer index (p<0.05) and gastric volume (p< 0.01), with no change in mucus secretion.
Conclusion: D-002 and Lyprinol did not show gastrotoxic effects and similar efficacy in protecting against ethanol and pylorus ligation-induced gastric ulceration in rats
Keywords: D-002, Lyprinol, ethanol ulcers, pylorus ligation, gastroprotection.
RESUMEN
Introducción: el D‒002, una mezcla de alcoholes de la cera de abejas, efectivo en modelos de osteoartritis y para reducir los síntomas de la misma. A diferencia de los medicamentos antiinflamatorios clásicos el D‒002 produce efectos gastroprotectores más que efectos gastrotóxicos. El Lyprinol, usado para disminuir la inflamación y los síntomas artríticos, mejora los síntomas de disfunción gastrointestinal en sujetos con dicha enfermedad. D‒002 y Lyprinol inhiben las actividades de cyclooxigenasa y 5‒lipooxigenasa, y son similarmente efectivos para reducir la inflamación en modelos experimentales. ]]>
Objetivo: comparar los efectos del D‒002 y el Lyprinol sobre la mucosa gástrica de ratas normales y de ratas con úlcera gástrica inducida experimentalmente.Palabras clave: D‒002, Lyprinol, úlcera gástricas por etanol, ligadura del píloro, gastroprotección.
INTRODUCTION
Inflammation is a normal host response against deleterious stimuli. Acute inflammation involves short-term responses to pro-inflammatory injuries and further repair, but if this mechanism fail it may evolves to chronic inflammation, which has been linked with long-standing diseases, including osteoarthritis.1 The inflammatory response involves the increased production of arachidonic acid from membrane phospholipids, whichis then metabolized by cyclooxygenase (COX) and lipoxygenase (LOX) pathways to produce prostaglandins (PG), thromboxanes, prostacyclins, and leukotrienes (LT). 2
Currently non selective non-steroidal anti-inflammatory drugs (NSAIDs) and COX-2 inhibitors, very effective to treat inflammation and associated pain, are among the most prescribed drugs worldwide. Basically, NSAIDs and COX-2 inhibitors display their anti-inflammatory effects by inhibiting the COX pathway, but such a mechanism also supports some of their relevant drug-related adverse effects, like gastric damage and bronchospasm linked with COX-1 inhibition, and cardiovascular adverse side effects caused by COX2 inhibition.3 NSAIDs-induced gastric damage, a relative limitation for their clinical use, results from the curtailed production of gastroprotective PG and the displacement of the arachidonic acid metabolism towards the LOX pathway, which increases the synthesis of pro-inflammatory and gastrotoxic LT, so that increased concentrations of LTB4 have been found on the walls of NSAID-induced gastric ulcers, which attract leukocytes and contribute to cause stomach ulceration,4 enhancing the gastrotoxicity due to the PG deficit.3
]]> Keeping in mind the extended use of NSAIDs for vascular prevention, which is the case for acetic salicylic acid (ASA), and inflammation-related diseases, like osteoarthritis, the incidence of NSAIDs-related gastric damage and complications have been increasing, a trends predicted to continue due the increase on life expectancy, so that special recommendations for their use in the elderly have been reported.5 This background supports the search of effective and safer anti-inflammatory substances, like dual inhibitors of COX and 5-LOX, which cut PG synthesis and provide anti-inflammatory effects, but also prevent the switch to an increased LT production by inhibiting 5-LOX, thus reducing the occurrence of gastric adverse effects.6 The inhibition of LTB4 synthesis, therefore, seems to be a relevant target of anti-inflammatory drugs devoid of gastrotoxicity.7D-002, a mixture of six beeswax alcohols with triacontanol as the main component, has been effective for lowering experimental inflammation8 and OA symptoms.9 D-002 was also found to inhibit both COX and 5-LOX activity, 10and to lower LTB4 levelsin the pleural exudates of rats with carrageenan-induced pleurisy.7Unlike the classic anti-inflammatory drugs, however, D-002 has shown to cause gastroprotective, rather than gastrotoxic effects, in experimental11,12 and clinical studies.13 Although the dual inhibition of COX and 5-LOX may support the lack of D-002 gastrotoxicity; this mechanism alone cannot explain the gastroprotective effect of D-002, which involves antioxidant effects on the gastric mucosa and increased gastric mucus secretion.11,12
Lyprinol, trade mark of a lipid extract from green-lipped mussel (GLM) (Perna canaliculus), inhibits COX and 5-LOX enzymes and reduces the production of LTB4. Lyprinol has been used for the symptomatic relief of inflammatory conditions, including arthritis, not only without causing the adverse effects of NSAIDs, but even improving symptoms of gastrointestinal dysfunction in OA subjects.14
A previous study found that D-002 and Lyprinol were similarly effective for lowering the oedema in the model of carrageenan-induced pleurisy,15 but their effects on gastric ulceration had not been compared yet.
This study compares the effects of single oral doses of D-002 and Lyprinol on gastric ulceration in rats.
METHODS
ANIMALS
Male Sprague Dawley rats (250-300g) from the National Center for Laboratory Animal Production (CENPALAB, Havana, Cuba) were used for the study and adapted for 7 days to the following conditions: temperature (22-23 oC), relative humidity (55-60 %) and 12 hours dark/light cycles. Food and water were freely supplied. The animals had fasted for 24 h prior to the experiments.
The experiments were performed in accordance with the care and use of experimental animals prescribed by the Cuban Guidelines for the care of laboratory animals and the Cuban Code of Good Laboratory Practice (GLP), after obtaining the approval of the Institutional Board for animal use.The study protocol and animals use were approved prior to the study by the Institutional Animal Ethics Committee (No 035/2011).
]]> ADMINISTRATION AND DOSAGEThe batch of D-002, supplied by the Plants of Natural Products (National Center for Scientific Research, Havana City, Cuba) was used after corroborate its quality criteria. Batch composition, assessed with a validated gas chromatographic method,16 was as follows: 1-tetracosanol (5 %), 1-hexacosanol (10.2 %), 1-octacosanol (14 %), 1-triacontanol (34.21 %), 1-dotriacontanol (24.24 %) and 1-tetratriacontanol (3.03 %). Purity (total content of these alcohols) was 90.7 %.
Lyprinol soft gel capsules (Blackmores, Australia, batch: 252918) used for the experiments contained 50 mg of the lipid extract from GLM diluted in olive oil and Vitamin E. GLM contain free fatty acids, mainly omega-3 polyunsaturated fatty acids (PUFAs), such as eicosapentaenoic acid (18 %) and docosahexanoic acid (15%), palmitic acid (16 %), cholesterol (≈30 %), sterol esters, triglycerides, and polar lipids, as reported. 17 The content of the capsules was directly drawn with a syringe and used in the experiments.
Omeprazole (lote OE513187) from Laboratorios NOVATEC (La Habana, Cuba), was dissolved in acacia gum/water vehicle (10 mg/mL) and then used in the experiments. D-002 and Lyprinol were suspended in 2 % Tween 65/water, respectively, while ASA (from the Cuban Pharmaceutical Industry, Havana, Cuba) was dissolved in acacia gum/water vehicle (10 mg/mL). All treatments (vehicle, D-002, Lyprinol, Aspirin and Omeprazole) were administered orally by gastric gavage (5mL/kg).
EXPERIMENTAL DESIGN
Three experiments were performed for comparing the effects of single oral doses of D-002 and Lyprinol on gastric ulceration in rats. The first experiment investigated the potential gastro-toxic effects of D-002 and Lyprinol in normal rats, while the two other series compared the effects of both substances on ethanol and pylorus ligation-induced ulcers in rats by assessing their effects on ulcer index.
EFFECTS ON THE GASTRIC MUCOSA OF NORMAL RATS
Rats were randomized into eight groups (10 rats per group): a vehicle control, three treated with D-002 (50, 200 and 400 mg/kg), three treated with Lyprinol (50, 200 and 400 mg/kg) and one with aspirin (150 mg/kg).
All treatments were administered for seven days. After the last administration, rats were fasted for 24 h with free access to water and then they were sacrificed by using an overdose of thiopental anaesthesia (80 mg/kg). The stomachs were immediately removed for quantifying the lesions.
EFFECTS ON ETHANOL INDUCED GASTRIC ULCERS IN RATS
]]> Seven groups of 10 rats each one were treated with the control vehicle; D-002 (50, 200 and 400 mg/kg) or Lyprinol (50, 200 and 400 mg/kg) one hour prior to oral administration of ethanol (60 % ethanol; 1 mL/200 g).18 The animals were sacrificed after one hour under an overdose of thiopental anesthesia and their stomachs were washed with cold saline and removed for examining the ulcer index microscopically.EFFECTS ON PYLORUS LIGATION INDUCED GASTRIC ULCERS IN RATS
Rats were randomized into nine groups of 10 rats each one, a negative control and eight pylorus-ligated groups that were orally administered with the vehicle (positive control), D-002 (50, 200 and 400 mg/kg), Lyprinol (50, 200 and 400 mg/kg) or omeprazole 10 mg/kg, respectively.
After one hour, the rats were anaesthetized with thiopental (40 mg/kg, i.p), the abdomen was cut open, the pyloric end was ligated without damaging the blood supply and the abdominal cavity was closed. The rats were anaesthetized and sacrificed four hours after pylorus ligation, the stomachs were removed and opened along the greater curvature and the ulcer indexes were measured. The volume of gastric juice, the mucus weight/stomach weight ratio and the concentrations of total hydroxyperoxides (THP) in gastric mucus were also measured.
EVALUATION OF GASTRIC ULCERATION
The lesions in the gastric mucosa were examined macroscopically using magnification 3x. Ulcer indexes were determined as the sum of the lengths of the whole gastric lesions (in mm). Two independent, blinded observers performed the observations and measurements of lesion lengths. 19
GASTRIC JUICE VOLUME
The gastric contents were collected and centrifuged at 3000 rpm-1 for 10 min. The supernatant was used for determining the gastric volume.
MUCUS WEIGHT/STOMACH WEIGHT RATIO
Stomachs were weighed in Mettler Toledo balance, opened along the greater curvature and then the mucus was scraped gently with a scalpel and weighed immediately in the Mettler Toledo balance (model: AG204, manufacturer: DeltaRange®, precision: 0.0001g). The amount of mucus was expressed as the mucus weight/stomach weight ratio.
]]> MEASUREMENT OF LIPID PEROXIDATIONThe stomachs were washed with normal saline, cut into small pieces and homogenized in a Potter-Elvehjem glass homogenizer in ice cold 0.15 M KCl to obtain 20 % homogenates used for the determination of THP,20 a marker of lipid peroxidation.In brief 0.1 mL of the homogenatewas allowed to react with 0.9 mL of Fox reactive (88 mg of butylated hydroxytoluene, 7.6 mg of orange xylenol, 9.8 mg of ammonium sulphate were dissolved in a 9:1 mixture of methanol:sulphuric acid). This mixture was incubated at 37 ºC for 30 min and then cooled. This mixture was centrifuged at 2400xg for 10 min and the absorbance of the supernatant was measured at 560 nm. The concentrations of THP were calculated from a standard calibration curve generated with cumene hydroperoxide. THP values were expressed as mol/mg of tissue protein.
STATISTICAL ANALYSES
Comparisons among groups were done with the Kruskal Wallis test, paired comparisons between each treated and control groups with the Mann-Whitney U test and categorical data (ulcer frequency) with the Fisher’s Exact Probability test. Statistical significance was chosen for α=0.05. Data were processed with the Statistics Software for Windows (Release 6.1 Stat Soft Inc, Tulsa OK, USA).
RESULTS
EFFECTS ON THE GASTRIC MUCOSA OF NORMAL RATS
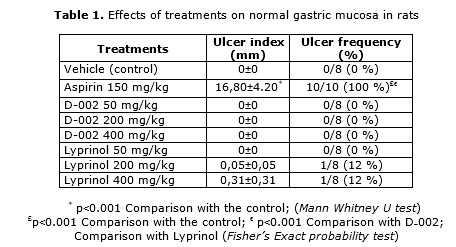
No rat from the vehicle control group, but all treated with aspirin 150 mg/kg exhibited gastric ulceration, so that both the ulcer index and the frequency of rats with gastric ulcers were significantly higher in the aspirin group as compared to the vehicle control. By contrast, no rat treated with D-002 (50˗400 mg/kg), and only two treated with Lyprinol at 200 and 400 mg/kg, respectively, displayed gastric ulcers. In consequence, ulcer index and frequency of animals with gastric ulceration in these two groups were similar to those of the control group (Table 1).
EFFECTS ON ETHANOL-INDUCED GASTRIC ULCERS
]]> Pre-treatment of rats with single oral doses of D-002 (50-400 mg/kg) decreased significantly and markedly (38.8% to 71.0 %) the ulcer index induced by ethanol as compared to the positive control (table 2). In turn, Lyprinol 200 and 400 mg/kg, not 50 mg/kg, reduced significantly and markedly (67.5 % and 73.7 %) the ulcer index in this model. The reductions of ulcer index with both substances were similar.EFFECTS ON PYLORUS LIGATION INDUCED GASTRIC ULCERS
Table 3 summarizes the effects of treatments on pylorus ligation-induced gastric damage in rats. Oral acute administration of D-002 (50˗400 mg/kg) produced significant decreases (56 ˗63 %) of the ulcer index as compared with the positive control group, and statistically similar reductions (52˗ 64 %) were found with Lyprinol (50˗400 mg/kg) (table 3). Also, all doses of both substances (50, 200 and 400 mg/kg) reduced significantly THP concentrations in the mucus (44, 59 and 63 % with D˗002; respectively; 22, 34 and 40 % with Lyprinol, respectively), but D-002 was more effective than Lyprinol for lowering this variable across all the dose range. Also, D-002 (200 and 400 mg/kg) decreased the gastric volume (34 and 39 %, respectively) and increased gastric mucus secretion versus the positive control; meanwhile only the highest dose of Lyprinol (400 mg/kg) increased significantly the gastric mucus secretion but without modifying the gastric volume. Omeprazole 10 mg/kg significantly reduced ulcer index (60 %), gastric volume (50 %) and mucus THP values (93 %), but unchanged mucus secretion.
DISCUSSION
Inflammation of the gastric mucosa may affect its own defensive protection, which may lead to gastric mucosal ulceration. Effective management of gastric ulceration, which results from the imbalance between gastroprotective and aggressive factors, would primarily depends on the reduction of the aggressive factors, the reinforcement of protective factors or a combination of both.21
Gastric ulcers may arise frequently due to the chronic use of external aggressive factors, such as NSAIDs, stress, smoking, and alcohol consumption.21 Nowadays, drug-induced damage to the gastrointestinal tract has become a health problem, in great part due to the extensive use of NSAIDs, 3 which could increase dramatically if its use for chemoprotection against different cancers is expanded.22
In this study, it was demonstrated that the repeated oral administration of D-002 or Lyprinol (50-400 mg/kg) did not induce gastric ulceration in rats, differently from aspirin (150 mg/kg), which produced evident ulcers in all treated rats, as expected. Beyond this lack of gastrotoxic effects caused by either D-002 or Lyprinol, both substances (50 – 400 mg/kg) demonstrated to be similarly effective for protecting against gastric ulceration induced by ethanol or pylorus ligation, this study being the first one reporting such effect for Lyprinol (Entrez PubMed, up to April 2014).
We found that the decrease of pylorus ligation-induced gastric ulceration produced by both D-002 and Lyprinol was accompanied by a reduction of lipid peroxidation (assessed by THP levels). The gastroprotective effect of D-002, not of Lyprinol, on pylorus ligation-induced ulceration also involves a decrease of gastric volume and an increase of gastric mucus secretion, in agreement with previous findings found in other ulcer models11 and Omeprazole, a classic proton pump inhibitor used as reference drug, was able to reduce the ulcer index, the gastric volume, and THP concentrations, in agreement with its antisecretory, antiapoptoticand antioxidant effects.23 These results agree with those of other authors,24 but diverge of those of pantoprazole, which was ineffective in this model.25
]]> The gastric ulceration produced by pyloric ligation is caused by the increased accumulation of gastric acid and pepsin, leading to auto-digestion of gastric mucosa. Defensive factors like the mucus are known to protect the submucosal layers from the back-diffusion of hydrogen ions. Administration of D-002 and the highest doses of Lyprinol significantly increased mucus secretion, which accounts for a reinforcement of the defensive factors acting on the gastric mucosa. D-002 also significantly reduced the gastric volume. Both D-002 and Lyprinol significantly decreased the concentrations of THP in mucosa homogenates when compared to the control group of animals. The antioxidant effect of D-002 on the gastric mucosa is consistent with previous reports, 12 meanwhile the effect on lipid peroxidation of Lyprinol on this target is documented for the first time and must be corroborate for further studies. D-002, however, was more effective than Lyprinol regarding for lowering the extent of lipid peroxidation in gastric homogenates. The decrease on lipid peroxidation could play an important role in the protection of the gastric mucosa, as oxidative damage is accepted to be a common factor in the pathogenesis of gastric ulceration.26Indeed, dual inhibition of D-002 and Lyprinol on COX and LOX pathways could be beneficial in limiting inflammation when compared to the conventional NSAIDs; and the joint blockade of both pathways may also lower the incidence of gastric ulceration.
CONCLUSION
This study demonstrates that instead to be gastrotoxic, oral administration of D-002 and Lyprinol similarly protected against ethanol and pylorus ligation-induced gastric ulceration.In pylorus-ligated rats, D-002 and Lyprinol increased mucus secretion but D-002 displayed the greatest reduction of lipid peroxidation. Taken together, these data merit further research and enhance our understanding of the gastric safety of two dual inhibitors of COX and 5-LOX enzymes, namely D-002 and Lyprinol, which seem to reinforce some defensive factors on the gastric mucosa.
REFERENCES
1. Berenbaum F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthritis Cartilage 2013; 21: 16–21.
2. Gajraj NM. Cyclooxygenase-2 Inhibitors. Anesthesia & Analgesia 2003; 96: 1720-1738.
]]>3. Takeuchi K. Pat hogenesis of NSAID-induced gastric damage: importance of cyclooxygenase inhibition and gastric hypermotility. World J Gastroenterol 2012; 18:2147-2160.
4. Hudson N, Balsitis M, Everitt S, Hawkey CJ. Enhanced gastric mucosal leukotriene B4 synthesis in patients taking non-steroidal anti-inflammatory drugs. Gut 1993; 34: 742–747.
5. Harirforoosh S, Asghar W, Jamali F. Adverse effects of nonsteroidal antiinflammatory drugs: an update of gastrointestinal, cardiovascular and renal complications. J Pharm Pharm Sci 2013;16: 821-847.
6. Landa P, Kutil Z, Temml V, Malik J, Kokoska L, Widowitz U, et al. Inhibition of in vitro leukotriene B4 biosynthesis in human neutrophil granulocytes and docking studies of natural quinones. Nat Prod Commun 2013; 8:105-108.
7. Altavilla D, Minutoli L, Polito F, Irrera N, Arena S, Magno C, et al. Effects of flavocoxid, a dual inhibitor of COX and 5-lipoxygenase enzymes, on benign prostatic hyperplasia. Br J Pharmacol 2012; 167: 95-108.
]]>8. Carbajal D, Molina V, Valdés S, Arruzazabala ML, Más R, Magraner J. Anti-inflammatory activity of D-002: an active product isolated from beeswax. Prostagl Leukotr Essent Fatty Acids 1998; 59: 235-238.
9. Rodríguez I, Mendoza S, Illnait J, Mas R, Fernández JC, Fernández L, et al.Effects of D-002, a mixture of beeswax alcohols, on osteoarthritis symptoms: a randomized placebo-controlled study. IOSR J Pharmacy 2012, 2:1-9.
10. Pérez Y, Oyarzábal A, Ravelo Y, Más R, Jiménez S and Molina V. Inhibition of COX and 5-LOX enzymes by D-002 (beeswax alcohols). Current Top Nutraceutical Research 2014, 12 (1/2): 13-18.
11. Carbajal D, Molina V, Noa M, Valdes S, Arruzazabala ML, Aguilar A, et al. Effects of D-002 on gastric mucus composition in ethanol induced ulcer. Pharmacol Res 2000; 42: 329-32.
12. Molina V, Valdés S, Carbajal D, Arruzazabala ML, Menéndez R, Mas R. Antioxidant effects of D-002 on gastric mucosa of rats with experimentally-induced injury. J. Med. Food. 2001; 4: 79-83.
]]>13. Illnait J, Rodríguez I, Molina V, Mendoza S, Mas R, Fernández L, et al. Effects of D-002 (beeswax alcohols) on gastrointestinal symptoms and oxidative markers in middle-aged and older subjects. Lat Am J Pharm 2013; 32: 166-174.
14. Coulson S, Butt H, Vecchio P, Gramotnev H, Vitetta L. Green-lipped mussel extract (Perna canaliculus) and glucosamine sulphate in patients with knee osteoarthritis: therapeutic efficacy and effects on gastrointestinal microbiota profiles. Inflammopharmacology 2013; 21:79-90.
15. Ravelo Y, Molina V, Pérez Y, Oyarzábal A, Jiménez S, Mas R. Anti-oedema effects of D-002 and Lyprinol on the carrageenan-induced pleurisy in rats. Int J Pharm Scienc Review Res 2013; 23(1): 24-28.
16. González V, Marrero D, Sierra R, Velázquez C, Vicente R. Nuevo método por Cromatografía Gaseosa Capilar para el análisis del ingrediente activo D-002. Revista CENIC Ciencias Químicas 2008; 39: 123-124.
17. Murphy KJ, Mooney BD, Mann NJ, Nichols PD, Sinclair AJ. Lipid, FA, and sterol composition of New Zealand green lipped mussel (Perna canaliculus) and Tasmanian blue mussel (Mytilus edulis). Lipids 2002; 37:587–595
18. Zengil H, Onuk E, Ercan ZS, Tker RK. Protective effect of iloprost and UK 38 485 against gastric mucosal damage induced by various stimuli. Prostagl, Leukotr Med 1987; 30: 61-67.
19. Ohara A, Sugiyama S, Hoshino H, Hamajima E, Goto H, Tsukamoto Y, et al. Reduction of adverse effects of indometacin by anti-allergic drugs in rat stomachs. Arzneim-Forsch Drug Res 1992; 42 (II): 9.
20. Jiang Zy, Hunt JV, Wolff SP. Detection of lipid hydroperoxides using the FOX method. Anal Biochem 1992; 384-389.
21. Tytqat GN. Etiopathogenic principles and ulcer disease classification. Dig Dis 2011; 29: 454-458.
22. Mikulec CD, Rundhaug JE Simper MSLubet RA,Fischer SM. The chemopreventive efficacies of nonsteroidal anti-inflammatory drugs: the relationship of short-term biomarkers to long-term skin tumor outcome. Cancer Prev Res. 2013; 6: 675-85.
23. Biswas K, Bandyopadhyay U, Chatoopadhyay I, Varadaraj A, Ali E, Ranajit K, et al. A novel anti oxidant and antiapoptotic role of omeprazole to block gastric ulcer through scavenging of hydroxyl radical. J Biol Chem 2003; 278: 10993-11001.
24. Livingston Raja NR, Sundar K. Psidium guajava Linn confers gastro protective effects on rats. Eur Rev Med Pharmacol Sci 2012;16: 151-156.
25. Dae-Kwon Bae, >Dongsun ParkSun Hee LeeGoeun YangYun-Hui Yang, et al. Different antiulcer activities of pantoprazole in stress, alcohol and pylorus ligation-Induced ulcer models. Lab Anim Res 2011; 27: 47–52
26. Bindu S, Mazumder S, Dey S, Pal C, Goyal M, Alam A, et al. Non-steroidal anti-inflammatory drug induces proinflammatory damage in gastric mucosa through NF-κB activation and neutrophil infiltration: Anti-inflammatory role of heme oxygenase-1 against non-steroidal anti-inflammatory drug. Free Radic Biol Med 2013; 65: 456-67.
Recibido: 23 de enero de 2015.
Aprobado: 15 de febrero de 2015.
Vivian Molina Cuevas . Centro de Productos Naturales, (CPN), Centro de Investigaciones Científicas (CNIC). Calle 198 e/ 19 y 21 Atabey, Playa, La Habana, Cuba. Teléfono: 2714225, 2714200, 2714212 Correo electrónico: vivian.molina@cnic.edu.cu
]]>