Stability of 50mg-Beeswax alcohols tablets (Abexol®) packed in high-density polyethylene flasks
]]> ]]>
Estabilidad de tabletas con 50 mg de alcoholes de cera de abejas (Abexol®) en frascos de polietileno de alta densidad
Dr.C. Víctor Luis González-Canavaciolo, MSc. Roxana de la Caridad Sierra-Pérez, Lic. Carmen Luisa Morales-Rico, Ing. Roxana Vicente-Murillo, Dr.C. Eduardo Antonio Rodríguez-Leyes, Dr.C. David Marrero-Delange
Centro de Productos Naturales del Centro Nacional de Investigaciones Científicas. La Habana, Cuba.
]]>
ABSTRACT
Introduction: beeswax alcohols, consisting in a reproducible mixture of six primary fatty alcohols from 24 to 34 carbon atoms purified from Apis mellifera beeswax, has been shown to produce antioxidant and gastroprotective effects. This substance is used to manufacture Abexol® 50 mg tablets, the finished form used in clinical studies and in routine practice.
Objective: to determine the stability of 50 mg‒Beeswax alcohols tablets (Abexol®) packed in high‒density polyethylene flasks.
Methods: samples from three batches of Abexol® 50 mg tablets packed in white high density polyethylene flasks (Rainbow & Nature, Sydney, Australia) were put into cardboard boxes and kept under climatic conditions of the zone IV (30±2 ºC, 70±5 % of relative humidity) for five years.
Results: all parameters tested were within specifications throughout the whole study: appearance (white round tablets with intact surfaces), average weight (initial average weight±7,5 %), total content of the six fatty alcohols (50±3.75 mg), disintegration time (<15 min), hardness (˃3 kg/cm2) and microbiological content (≤1 000 bacteria/g and ≤100 fungi/g, absence of E. coli,S. aureus, Pseudomonas, Salmonella and C. albicans). This result is consistent with the data of a previous stability study of Abexol® 50 mg tablets manufactured in Cuba and packed in blisters of polyvinyl chloride and aluminum.
Conclusions: the results of the present study support that Abexol®50mg tablets packed in white high density polyethylene flasks and stored at the conditions of the climatic zone IV have a shelf life of 5 years.
Keywords: beeswax alcohols, Abexol, fatty alcohols, stability, tablets.
RESUMEN
Introducción: los alcoholes de la cera de abejas constituyen una mezcla reproducible de seis alcoholes grasos primarios de 24 a 34 átomos de carbono purificados de la cera de Apis mellifera. Esta sustancia, con efectos antioxidantes y gastroprotectores, es empleada para la elaboración de las tabletas de Abexol® con dosis de 50 mg de alcoholes, forma farmacéutica terminada utilizada en los ensayos clínicos y en la práctica de rutina. ]]>
Objetivo: determinar la estabilidad de las tabletas con 50 mg de alcoholes de cera de abejas (Abexol®) en frascos de polietileno de alta densidad.
Métodos: muestras de tres lotes de tabletas de Abexol® con dosis de 50 mg envasadas en frascos de polietileno de alta densidad ( Rainbow & Nature, Sydney, Australia) se pusieron en cajas de cartón y se mantuvieron en las condiciones de la zona climática IV (30±2 ºC, 70±5 % de humedad relativa) durante cinco años.
Resultados: los parámetros evaluados se mantuvieron dentro de sus especificaciones de calidad durante todo el estudio: apariencia (tabletas blancas redondas con superficies enteras), peso promedio (inicial±7,5 %), contenido total de los seis alcoholes grasos (50±3,75 mg), tiempo de desintegración (<15 min), dureza (˃3 kg/cm2) y contenido microbiológico (≤1 000 bacterias/g y ≤100 hongos/g, ausencia de E. coli, S. aureus, Pseudomonas, Salmonella y C. albicans). Este resultado es consistente con los datos de un estudio previo de estabilidad de tabletas de Abexol® con dosis de 50 mg, fabricadas en Cuba y envasadas en blísteres de cloruro de polivinilo y aluminio.
Conclusiones: los resultados del presente estudio sustentan que las tabletas de Abexol® con dosis de 50 mg, envasadas en frascos de polietileno de alta densidad y almacenadas en las condiciones de la zona climática IV, presentan un tiempo de vida útil de cinco años.
Palabras clave: alcoholes de la cera de abejas, Abexol, alcoholes grasos, estabilidad, tabletas.
INTRODUCTION
Beeswax alcohols (BWA) (formerly coded as D-002) is an active substance purified from the beeswax of Apis mellifera, mainly composed by a mixture of saturated linear fatty alcohols from 24 to 34 carbon atoms in reproducible proportions.1 BWA has been shown to produce antioxidant and gastroprotective effects in pharmacological and clinical studies.2-4 Since 1999, BWA (Abexol®) 50mg tablets packed in polyvinyl chloride (PVC) /aluminum blisters have been marketed in Cuba as an antioxidant nutritional supplement.
Stability studies have demonstrated the high intrinsic stability of BWA (active substance and tablets),5-6 consistent with the low reactivity of the fatty alcohols that compose this substance. No degradation products were found under conditions of thermolysis, hydrolysis, oxidation, and photolysis. 5 Six months accelerated studies conducted at 40 °C and 75 % of relative humidity (RH), and three years stability studies performed under the conditions of the climatic zones II and IV also proven the high stability of the active substance and of the tablets packaged in polyethylene bags and PVC/aluminum blisters, respectively, as containers.5 These results corroborated those of the differential scanning calorimetry study, which demonstrated the high thermal stability of BWA up to 200 °C and its null interaction with the excipients used in the tablet formulation.6
]]> BWA has been approved as functional food or as a supplement in some Asian countries. While originally BWA tablets were packed in PVC/aluminum blisters, these new markets have demanded their packing in high density polyethylene (HDPE) flasks, following a common current tendency for this kind of products. Previous data, however, were not enough to extrapolate the stability of BWA tablets packed in blisters to flasks. In light of these issues, the aim of the present work was to determine the shelf life of BWA tablets in their new primary container.
METHODS
REAGENTS AND CHEMICALS
BWA active substance was provided by the National Centre for Scientific Research (Cuba). Fatty alcohols reference standards and N-methyl, N-trimethylsilyltrifluoroacetamide (MSTFA) were purchased from Sigma (USA). The analytical grade hexane and chloroform were purchased from Merck (Germany).
EQUIPMENTS
The gas chromatographic (GC) analysis was performed by using an Agilent 6890N gas chromatograph (USA) equipped with flame ionization detector, a Chemstation software, version 3.03.01, and an HP-5 fused-silica capillary column (30 m x 0,25 mm id, 0,25 µm film-thickness, Agilent, USA). Analytic balance (Mettler Toledo, Switzerland), dry thermostat (Lab-Line Instr., USA), Monsanto hardness tester and disintegrator tester ( PharmaTest, Germany) were also used.
STABILITY STUDIES
Samples of the industrial batches 51265, 51266 and 52058, consisting of BWA 50 mg tablets packed in white HDPE bottles with snap hinge top caps of the same material and tamper evident closures (90 tablets/bottle) were supplied by Rainbow & Nature (Sydney, Australia). Bottles were packed into cardboard boxes and kept under climatic zone IV conditions: 30±2 ºC and 70±5 % RH for five years. Studies followed the regulations issued by the International Conference on Harmonization7,8 and by the regulatory Cuban agency CECMED.9
The assayed parameters, tested at initial time (t=0) and every 12 months, should be within the quality specifications of these tablets, unpublished data stated in the chemical pharmaceutical part of the Dossier submitted to the regulatory agencies that approved this product. Such parameters were the following (limits and number of replies in parentheses): appearance (white round tablets with intact surfaces, assayed with 20 tablets), average mass (initial average mass±7,5 %, assayed with 20 tablets), total content of the six fatty alcohols (50±3,75 mg, assayed with 20 tablets, n=3), disintegration time (<15 min, n=6), hardness (˃ 3 kg/cm2, n=10) and microbiological content (≤1 000 bacteria/g and ≤100 fungi/g, absence of E. coli, S. aureus, Pseudomonas, Salmonella and C. albicans, assayed with 100 tablets). The content of the six high molecular weight alcohols was assayed by a validated stability-indicating GC method that uses 1-eicosanol as internal standard.10 The total content of the six alcohols, determined at different times, were compared to baseline values with the t Student test (p=0.05). The other previously mentioned parameters were assayed by USP methods.11
]]> PREPARATION OF SAMPLE SOLUTION AND GC ASSAYTwenty tablets were weighed and the tablets were transferred into dry mortar. Tablets powder equivalent to 25 mg of BWA was quantitatively transferred to a test tube were 3 mL of Internal standard solution (0,4 mg 1-eicosanol/mL in chloroform) and 2 mL chloroform were added. Test tube was heated at 65 ºC for 15 minutes, with occasional shaking. This solution was hot filtered through Whatman 1 filter paper to another test tube, and 3 mL n-hexane was added. The tube was heated at 65 ºC for 3 minutes, with occasional shaking. Afterwards, the tube was let to stand at 25 ºC for 5 minutes and the solution was filtered through Whatman 1 filter paper. A 0,5 mL aliquot was transferred to a 4 mL vial with screw cap, were 0,05 mL MSTFA and 0,5 mL chloroform was added. The vial was heated at 65 °C for 15 minutes. Samples were prepared by triplicate.
A volume of 0,5 µL of the sample solution was injected in the GC equipment in splitless mode. The oven was heated from 70 °C (1 min) to 200 °C (at 30 °C/min), and from 200 to 320 °C (at 8 °C/min) with 15 min isothermal at the final temperature. Hydrogen was used as carrier gas (at 1 mL/min), and detector and injector temperature was 320 °C.
RESULTS
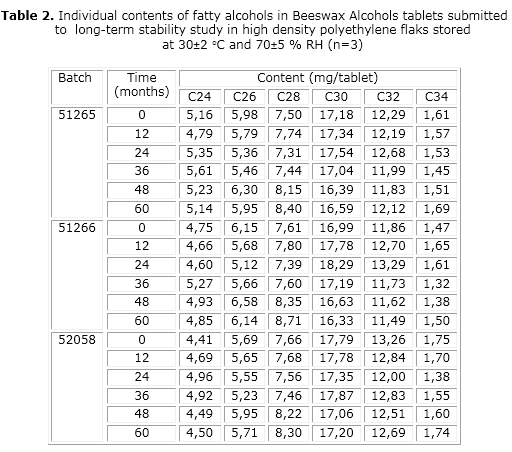
No individual out of specification values were observed for assayed parameters during all the study. Average mass, hardness, disintegration time and BWA content (table 1), as well as organoleptic characteristics and microbiological content, were within specifications. On its side, the individual contents of the six fatty alcohols, determined by gas chromatography (table 2 and fig.), matched with the initial contents, keeping the relative proportions that define BWA’s identity. Likewise, the small variations in BWA’s content were within the permitted dispersion of the analytical method (≤2 %).9 Data analysis show that the mean content of BWA in the tablets packed in HDPE flasks and stored for 5 years under 30±2 ºC/70 ±5 % RH did not differ statistically from the initial content.
DISCUSSION
This long-term stability study support the high stability of Abexol tablets, which did not undergo evident degradation after 60 months at the conditions of the climatic zone IV. Although disintegration time and hardness (tablet breaking force) increased during the study, final values agreed the specifications limits of these parameters; therefore, bioavailability of BWA should not be affected after 60 months of tablet storage at these conditions. This behavior evidences a cohesion increase of the tablet components on aging under high-humidity conditions, which has been too observed by other authors.12
]]> The high stability observed in this study is consistent with the result of a stability study of these tablets conducted in Australia in conditions of the climatic zone II (Rainbow & Nature LTD, Sydney, Australia, 2011, unpublished data). Additionally, these results represent new evidences of the high stability of this product, coherent with those of the stability study of BWA tablets manufactured in Cuba and packed in PVC /aluminum blisters,5 and with a previous study by Differential Scanning Calorimetry that demonstrated the high thermal stability of the active substance and its null interaction with the excipients.6Long-term stability studies, so-called real-time stability studies, define the shelf life of a product. The results of this study support that Abexol® (BWA) 50 mg tablets packed in white high density polyethylene flasks and stored at the conditions of the climatic zone IV have a shelf life of 5 years.
REFERENCES
1. González V, Sierra R, Magraner J, Rodríguez-Leyes E. Método para la determinación por cromatografía de gases del contenido de D-002, una nueva sustancia biologicamente activa purificada de la cera de abejas. Rev CENIC Cien Quim. 2006;37(1):21-2.
2. López E, Illnait J, Molina V, Oyarzábal A, Fernández L, Pérez Y, et al. Effects of D-002 (beeswax alcohols) on lipid peroxidation in middle-aged and older subjects. Latin Am J Pharm. 2008;27:695-703.
3. Pérez Y, Oyarzábal A, Mas R, Molina V, Jiménez S. Protective effect of D002, a mixture of beeswax alcohols against indomethacin-induced gastric ulcers and mechanism of action. J Nat Med. 2013;67(1):182-9.
]]>4. Illnait J, Rodriguez I, Molina V, Mendoza S, Mas R, Fernández L, et al. Effects of D-002 (beeswax Alcohols) on gastrointestinal symptoms and oxidative markers in middle-aged and older subjects. Lat Am J Pharm. 2013;32:166-74.
5. González VL, Sierra R, Marrero D, Rodríguez EA. Estudios de estabilidad del D002, mezcla de alcoholes grasos purificados de la cera de abejas, ingrediente activo y tabletas (3 años). Rev Cub Quím. 2011;XXIII(3):46-51.
6. González VL, Marrero D, Sierra R, Martínez L. Estudios organolépticos, fisicoquímicos microbiológicos e interacción con excipientes farmacéuticos de un extracto purificado de cera de Apis mellifera. Rev Cub Farm. 2010;44(3):354-9.
7. European Agency for the Evaluation of Medicinal Products. ICH Q1AR Stability Testing Guideline: Stability Testing of New Drug Substances and Products, London: EAENP; 2000.
8. Grimm W. Extension of the International Conference on Harmonization Tripartite Guideline for Stability Testing of New Drug Substances and Products to Countries of Climatic Zones III and IV. Drug Develop & Indust Pharm. 1998;24:313-25.
]]> 9. CECMED. Requerimientos de los estudios de estabilidad para el registro de productos farmacéuticos nuevos y conocidos. Centro de Control de Medicamentos, Equipos y Dispositivos. , Regulación 23; 2000.10. Marrero D, González VL, Sierra R, Rodríguez EA, Vicente R, Velázquez C. Validación de una nueva metodología analítica por CG capilar para la determinación de D002 en tabletas con dosis de 50 mg. Rev Cub Quím. 2009;XX(3):27-31.
11. The United States Pharmacopeial Convention. United Stated Pharmacopoeia 33 and National Formulary 28. Supplement 1. (Monograph on CD-ROM). Rockville, MD: USPC, Inc; 2010.
12. Chowhan ZT, Amaro A. The Effect of Low- and High-Humidity Aging on the Hardness, Disintegration Time and Dissolution Rate of Tribasic Calcium Phosphate-Based Tablets. Drug Dev Ind Pharm 1979;5(6):545-562.
Recibido: 16 de julio de 2014. ]]> Aprobado: 1 de mayo de 2015.
Víctor Luis González-Canavaciolo. Centro de Productos Naturales. Centro Nacional de Investigaciones Científicas. Calle 198, s.n., e/ 19 y 21, Atabey, Playa, La Habana, Cuba. Teléfono: 271 42 25. Correo electrónico: victor.gonzalez@cnic.edu.cu
]]>