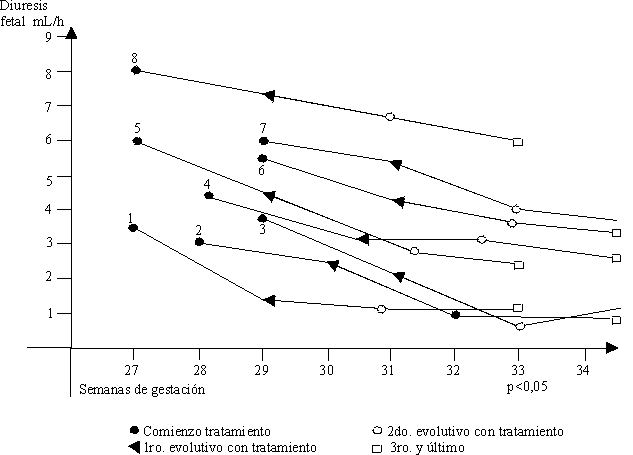
RESUMEN: Se estudiaron 8 embarazadas con polihidramnios, 4 de ellas con síntomas de amenaza de parto pretérmino o disnea ligera que fueron tratadas con indometacina oral (100 mg/ diarios por 7 d en 3 ciclos). El tiempo del diagnóstico y comienzo de la terapia fue de 27 ± 3 semanas. Se realizó biometría fetal semanalmente, así como ecocardiografía para valorar constricción del ductus, no se demostró que ocurriera en los casos tratados. Se encontró en los 8 casos que el 62,5 % nació después de las 37 semanas, con 3 bajo peso (37,5 %) y 1 de ellos presentaba crecimiento intrauterino retardado. El índice de bolsones se normalizó entre el 50 y 95 percentil en 6 de los casos después del tercer ciclo de tratamiento. Se realizó el estudio seriado de vejiga fetal por ecografía antes y después de la terapia con indometacina observándose una declinación significativa de la diuresis fetal. En 3 de los 8 casos se pudo determinar la causa del hidramnios. Hubo una muerte anteparto en un gemelar y otra neonatal. Se recomienda esta terapia con indometacina en los hidramnios con un seguimiento estricto de los parámetros expuestos para la evolución.
Descriptores DeCS: POLIHIDRAMNIOS/ quimioterapia; INDOMETACINA/ administración & dosificación; INDOMETACINA/ uso terapéutico.
El embarazo con polihidramnios, por sus múltiples causas, ha sido considerado como un riesgo, donde poco podemos hacer los obstetras como no sea tener una conducta expectante y una vigilia del embarazo, cuya evolución en muchos casos no es favorable por el riesgo que existe de parto pretérmino, crecimiento intrauterino retardado, rotura de las membranas ovulares y otras.
La regulación del fluido de líquido amniótico ha sido poco conocida, demostrándose un papel importante de la orina fetal, evidenciándose que una anuria resulta en oligohidramnios.1-4
Ha sido en los últimos tiempos discutido el tratamiento con indometacina en estos casos de hidramnios en períodos tempranos como factor que causa disminución de la orina fetal. Kirshan y otros han encontrado relación entre la poliuria y el hidramnios,2,5,6 no así en casos con diuresis normal con tratamiento de indometacina, posiblemente por un aumento de la hormona antidiurética (ADH) fetal aunque está por comprobarse el efecto que sobre la reabsorción del líquido amniótico por parte del lecho alveolar ejerce la indometacina. Pritchard sostiene que podría aumentar la reabsorción, mientras que Ketterman y Liggens argumentan un incremento de la frecuencia respiratoria.7-12
Nosotros hemos querido realizar este estudio preliminar en 8 pacientes con polihidramnios diagnosticado clínica y ecográficamente, algunos de los cuales ya presentaban sintomatología discreta cuantificándose algunas variables de interés.

Se le realizó a estas pacientes un ultrasonido antes de comenzar el tratamiento y descartar algún tipo de malformación fetal. ]]>
Se comenzó posteriormente el tratamiento con indometacina oral 25 mg cada 6 h es decir 100 mg diarios durante 7 d; se descansa una semana sin administrar el medicamento y se realiza una biometría fetal y una ecocardiografía. De no existir constricción del ductus se continúa la terapia durante 7 d más hasta realizar 3 ciclos con tratamiento.En cada examen ecográfico evolutivo se determinó el diámetro biparietal, la longitud del fémur, las circunferencias cefálica y abdominal, el índice de los bolsones de líquido amniótico, el peso fetal y el volumen de la vejiga fetal en 2 tomas con diferencia de 1 h para determinar la diuresis fetal.
Se buscó además la vía del nacimiento, el peso al nacer y el sexo, así como la evolución del recién nacido. Todos los resultados fueron tabulados y se evaluó estadísticamente por prueba de Student para significación de p < 0,05.
Como conclusión tenemos que la diuresis fetal disminuyó en los casos tratados con indometacina, siendo estadísticamente significativa. El índice de los bolsones de líquido amniótico disminuyó después de la terapia en el 75 % de los casos. En el 62,5 % de las pacientes se produjo el nacimiento después de las 37 semanas y hubo 3 recién nacidos con bajo peso.
| | ||||
| No. de casos | | ]]> 29 | | |
| Diuresis | | | | |
| 1 | | | ]]> 1,5 | |
| 2 | | | | |
| 3 | | | | |
| 4 | ]]> 4,5 | | | |
| 5 | | | | |
| 6 | | | ]]> 4,5 | |
| 7 | | | | |
| 8 | | | | |
| X | ]]> 5,0 | | | |
| Paciente | | ]]> TG término tratamiento | | | | | | |
| | | |||||||
| | ]]> 27 | | | | | | | |
| | | ]]> 335 381 | | | | | | |
| | ||||||||
| | | | ]]> 36 | | | | | |
| | | | ||||||
| | ]]> 284 | | | | | | | |
| | | ]]> 33 | | | | | | |
| | | | ]]> Cesárea | | | | | |
| | | | | | ]]> 2 570 | | | |
| | | |||||||
| | | | | ]]> Cesárea | | | | |
Subject headings: POLYHYDRAMNIOS/drug therapy; INDOMETHACIN/administration & dosage; INDOMETHACIN/therapeutic use.
1. Lind T, Kendale A, Ilytten FE. The role of the fetus in the formation of amniotic fluid. Br J Obstet Gyneacol 1972;289-98.
2. Campbell L, Wladimiroff IW. The antenatal measurement of fetal urinary production. Obstet Gyneacol Br Commow 1973;80:680-6.
3. Cantor B, Tyler T, Nelson RM. Oligohydramnios and transient neonatal anuria. A possible association with the maternal use of prostaglandin synthetase inhibitors. J Reprod Med 1988;24:220-3.
4. Quenam JT, Thompson W, Whilfield CR. Amniotic fluid volumen in normal pregnancy. Am J Obstet Gynecol 1972;114:34-8.
5. Kirshan B. Fetal urine output in hydramnios. Obstet Gynecol 1989;73:240-2.
6. Cabrol D, Landesman R, Muller J. Treatment of polyhydramnios with prostaglandin synthetase inhibidor. Am J Obstet Gynecol 1987;157:422-6.
7. Anderson R, Beil T, Mc Donald D. Prostaglandins: effects and blood pressure renal blood flow, sodium and water excretion. Kidney Int 1976;10:205-15.
8. Leyberth HW, Rasche W, Hackenthal R. Effect of prolonged indomethacin therapy on renal funtion and selected vasoactive hormone in very low weight infants. J Pediatr 1983;103:979-84.
9. Kirshon B, Mari G, Moise K. Indomethacin therapy in the treatment of symptomatic polyhydramnios. Obstet Gynecol 1992;75: 202-5.
10. Sosa A. Patología del líquido amniótico. En: Ultrasonografía y clínica embrio-fetal. Editorial Tatum, 1993; t18:277-86.
11. Moise KJ, Kirshon B, Cano LE. Placental transfer of indomethacin in human pregnancy. Am J Obstet Gynecol 1990;162:549-60.
12. Philan UP, Park VW. Polyhydramnios and perinatal outcome. J Perinatal 1992; 10:347-50.Recibido: 4 de junio de 1998. Aprobado: 30 de septiembre de 1998. ]]>
Dr. Luis Raúl Martínez González. Hospital Docente Ginecoobstétrico "Justo Legón Padilla". Pinar del Río, Cuba.1 Especialista de II Grado en Ginecoobstetricia y Ultrasonografía.
2 Especialista de I Grado en Ginecoobstetricia. ]]>