The effect of erythropoietin on creatine phosphokinase levels during ischemia reperfusion injury in rats
Efecto de la eritropoyetina en niveles de creatine fosfoquinasa durante daño por isquemia reperfusión en ratas
Constantinos Tsompos,I Constantinos Panoulis,II Konstantinos Toutouzas, III George Zografos,III Apostolos PapaloisIV
I Department of Obstetrics & Gynecology, Mesologi County Hospital, Etoloakarnania, Greece.
II Department of Obstetrics & Gynecology, Aretaieion Hospital, Athens University, Greece. ]]>
ABSTRACT
Objective: to examine the effect of erythropoietin testing on rat model and particularly the ischemia reperfusion protocol.
Methods: experimental study of 40 rats weighing 247.7 g as average. The beneficial effect or non-effectiveness of the erythroproietin molecule on the blood creatine phosphokinase levels was biochemically studied. It was measured 60 min (groups A and C) and 120 min (groups B and D) after reperfusion with no administration of erythropoietin in groups C and D.
Results: Erythropoietin administration significantly increased the creatine phosphokinase levels to 3586.05 IU/L (1873.115 IU/L-5298.985 IU/L;p= 0.0001). This finding was in accordance with the result of paired t-test (p= 0.0001). Reperfusion time significantly increased the CPK levels to 557.35 IU/L (-1513.284 IU/L-2627.984 IU/L; p= 0.5890), also in accordance with paired t-test (p= 0.4661). The interaction of erythropoietin administration and reperfusion time significantly increased the creatine phosphokinase levels to 1988.282 IU/L (918.2646 IU/L-3058.299 IU/L; p= 0.0006).
Conclusions: Erythropoietin administration, reperfusion time and their interaction generally increase short-term effects on blood creatine phosphokinase after ischemia reperfusion injury.
]]>
Keywords: erythropoietin, creatine phosphokinase levels, reperfusion.
RESUMEN
Objetivo: examinar el efecto del test de eritropoyetina en un modelo de rata, particularmente el protocolo de isquemia reperfusión.
Métodos: estudio experimental en el que se usaron 40 ratas con un peso medio de 247,7 g. Se estudió bioquímicamente el beneficio o el no efecto de la molécula de eritropoyetina en la creatín fosfokinasa sanguínea. Esta se midió en dos momentos: 60 min después de la reperfusión (grupos A y C) y 120 min después de esta (grupos B y D). No se administraron eritropoyetina en los grupos A y B, contrario a los grupos C y D.
Resultados: la administración de eritropoyetina aumentó significativamente la creatín fosfokinasa sanguínea a 3586,05 UI/L (1873,115 IU/L-5298,985 UI/L; p= 0,0001), de acuerdo también con el test pareado t-test (p= 0,0001). El tiempo de reperfusión incrementó significativamente la creatín fosfokinasa sanguínea a 557,35 UI/L (-1513,284 UI/L-2627,984 UI/L; p= 0,5890), de acuerdo con el test aplicado t-test (p= 0,4661). La interacción de la administración de eritropoyetina y el tiempo de reperfusión elevó significativamente los niveles de creatín fosfokinasa sanguínea a 1988,282 UI/L (918,2646 IU/L-3058,299 IU/L; p= 0,0006).
Conclusión: la administración de eritropoyetina, el tiempo de reperfusión y su interacción, generalmente incrementa los efectos a corto plazo en la creatín fosfokinase sanguínea después del daño por isquemia reperfusión.
Palabras clave: eritropoyetina, creatín fosfoquinasa, reperfusión.
INTRODUCTION
]]> Tissue ischemia and reperfusion (IR) remain one of the main causes of permanent or transient damage with serious implications on adjacent organs and certainly on patients' health. The use of erythropoietin (Epo) is a well established knowledge for many years. However, even if important progress has been made, satisfactory answers have not been given yet to fundamental questions, such as, by what velocity this factor acts, when should it be administered, and in which dosage. The particularly satisfactory action of Epo in stem blood cells recovery has been noted in several performed experiments. Since a careful literature search (PubMed-Medline) was conducted, it was realized that this certain factor has been tried in experiments. However, just few relative reports were found, not covering completely this particular object of action velocity. Also, a lot of publications addressed trial of other similar molecules of growth factors to which the studied molecule also belongs to.The aim of this study was to examine the effect of Epo drug on rat model and particularly in an IR protocol. The beneficial effect or non-effectiveness of that molecule were studied by measuring blood creatine phosphokinase levels (CPK). This experimental study was approved by Scientific committee of Ippokrateion General Hospital, Athens University, and by Veterinary Address of East Attiki Prefecture under 3693/ november 12th, 2010 & january 14/10th, 2012 decisions. Institutional and national guide for the care and use of laboratory animals was followed.
METHODS
Experimental groups
This experimental study was laid out at the Exprerimental Research Center of ELPEN Pharmaceuticals Co. Inc. S.A. at Pikermi, Attiki. All settings needed for the study including of consumables, equipment and substances used, were a courtesy of that S. A. Albino female Wistar rats were used in accordance with accepted standards of humane animal care. They spent in laboratory 7 days before the experiment with easy access to water and food. The experiment was acute, that is, the animal usage was completed by following experimental set of times without awakening and preservation of the rodents. They were randomly assigned to four experimental groups (10 animals in each group).
- Group A: ischemia for 45 min and afterwards reperfusion for 60 min.
- Group B: ischemia for 45 min and afterwards reperfusion for 120 min.
]]> - Group C: ischemia for 45 min and afterwards immediate Epo intravenous (IV) administration and reperfusion for 60 min.- Group D: ischemia for 45 min and afterwards immediate Epo IV administration and reperfusion for 120 min.
The Epo molecule dose was 10 mg/Kg body weight of animals.
The experiment was beginning by prenarcosis and general anesthesia administration in animals. Their electrocardiogram and acidometry were continuously monitored. The vessels concerning blood supply were prepared so as their flow to be excluded by forceps. After exclusion, the protocol of IR was applied, exactly as described in experimental groups. The molecules were administered at the time of reperfusion, through inferior vena cava (catheterization had been preceded at experiment beginning, after general anesthesia establishment). The CPK levels measurement was performed at 60 min of reperfusion (groups A and C) and 120 min of reperfusion (groups B and D).
Protocol
Rats underwent general anesthesia by initial intramuscular (IM) administration of 0.5 cc compound, which constituted of 0.25 cc xylazine, [20 mg/cc, 25 cc] and 0.25 cc ketamine hydrochloride [1000, 100 mg/cc, 10 cc]. Before initiation of laparotomy, 0.03 cc butorphanol [10 mg/cc, 10 cc] anesthesia was administered subcutaneously (SC). Continuous oxygen supply was administered during whole experiment performance. Ischemia was caused by clapping inferior aorta over renal arteries for 45 min after laparotomic access was achieved. Reperfusion was induced by removing the clapping and reestablishment of inferior aorta potency. Forty (40) female Wistar albino rats were used of mean weight 247.7 g [Std. Dev: 34.99172 g], with min weight ≥ 165 g and max weight < 320 g. Rats' weight could be potentially a confusing factor, e.g. fatter rats to have greater blood CPK levels. This suspicion will be investigated.
Model of ischemia-reperfusion injury
Control groups: 20 control rats of mean weight 252.5 g [Std. Dev: 39.31988 g] were subjected to ischemia for 45 min and then reperfusion was induced.
]]>- Group A: reperfusion which lasted 60 min concerned 10 controls rats of mean weight 243 g [Std. Dev: 45.77724 g], mean CPK levels 2170.3 IU/L [Std. Dev: 817.926 IU/L].
- Group B: reperfusion which lasted 120 min concerned 10 controls rats of mean weight 262 g [Std. Dev: 31.10913 g], mean CPK levels 3107.6 IU/L [Std. Dev: 2360.911 IU/L].
Erythropoietin group: 20 rats of mean weight 242.9 g [Std. Dev: 30.3105 g] suffered by ischemia for 45 min and then reperfusion in the beginning of which 10 mg Epo/kg body weight were IV administered.
- Group C: reperfusion which lasted 60 min concerned 10 Epo rats of mean weight 242.8 g [Std. Dev: 29.33636 g], mean CPK levels 6136.3 IU/L [Std. Dev: 2973.109 IU/L].
- Group D: reperfusion which lasted 120 min concerned 10 Epo rats of mean weight 243 g [Std. Dev: 32.84644 g], mean CPK levels 6313.7 IU/L [Std. Dev: 3826.46 IU/L].
RESULTS
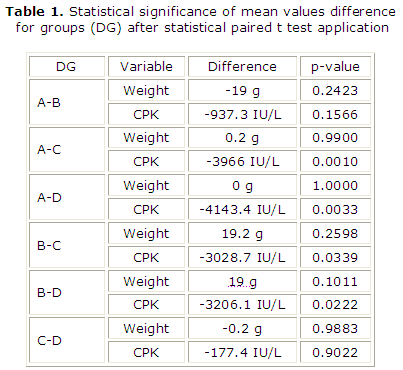
Every weight rats group initially was compared with other one from 3 remained groups applying statistical paired t-test (table 1). Any emerging significant difference among CPK levels, will be investigated whether owed in the above mentioned probable weight correlations. Every CPK rats group was also compared with other one from 3 remainder groups applying statistical paired t-test (table 1).
]]>
Applying generalised linear models (GLM) with dependant variable, the predicted CPK values and independent variables the erythropoietin administration or no, the reperfusion time and their interaction, results in: Epo administration significantly increased the CPK levels by 3586.05 IU/L [1873.115 IU/L-5298.985 IU/L] (p= 0.0001). This finding was in accordance with the result of paired t-test (p= 0.0001). Reperfusion time non-significantly increased the CPK levels by 557.35 IU/L [-1513.284 IU/L-2627.984 IU/L] (p= 0.5890), also in accordance with paired t-test (p= 0.4661). However, Epo administration and reperfusion time together significantly increased the CPK levels by 1988.282 IU/L [918.2646 IU/L-3058.299 IU/L] (p= 0.0006). The table 2 sums up concerning the alteration influence of Epo in connection with reperfusion time.
Inserting the rats weight as independent variable at GLM, a very significant relation turns on CPK levels (p= 0.0144), so as to further investigation is needed. The predicted CPK values, adjusted for rats' weight were calculated. Afterwards, the predicted CPK values of each one from 4 rats groups were compared with other one from 3 remainder groups applying statistical paired t-test. The table 3 sums up concerning the increasing influence of Epo in connection with reperfusion time.
DISCUSSION
Many clinical situations can prove how CPK levels are influenced by ischemic cases. Gonçalves et al subjected1 rats to ischemia for 30 min. There was a significant increase in plasma CPK levels of control rats than treated ones by ornithine α-ketoglutarate at the end of 30 minutes reperfusion period. Jebeli et al randomized2 patients with impaired left ventricular function [(LVEF) < 35 %] undergoing on-pump coronary artery bypass graft CABG to receive either milrinone or placebo saline (50 + 0.5 μg/kg/min) respectively. CPK serum levels, occurrence of myocardial ischemia or infarction were significantly higher in placebo group (p< 0.05). Sala et al compared3 patients with ventricular tachycardia (VT) occurring within the first 4 days of an acute phase of myocardial infarction (AMI) with control ones. The profile of patients with AMI, hospitalized in coronary care unit, who will likely suffer from VT is, more frequent occurrence of previous AMI, more frequent by 3.95-fold CPK-MB levels peak > 300 IU/L, more frequent by 11.23-fold ischemia previous to VT and 8.75-fold greater mortality rate than that in control group (p< 0.001 for all).
Serruys et al assigned4 patients after successful completion of their first percutaneous coronary intervention (PCI) randomly to receive treatment with fluvastatin 80 mg/d, or matching placebo at hospital discharge for 3 to 4 years. Major adverse cardiac events (MACE)-free survival time was significantly longer in the fluvastatin group (p= .01). There were no instances of CPK elevations 10 or more times the upper limit of normal. Savchuk et al caused5 short-term myocardial ischemia which increased the activity of total blood CPK by 57.3 + 11.7 mE/mL (p< 0.001) in dogs. Coronary blood flow disturbances experiments showed changes in enzymes activity.
Yang et al studied6 the effects on the glycerol-induced rhabdomyolysis, one of the causes of acute renal failure which significantly increased blood CPK levels in rats. Ten minutes after rhabdomyolysis, post-treatment with rhEPO (300 U/kg) decreased blood CPK levels and the markers of kidney injury 48 h after glycerol administration. Popov et al examined the single-nucleotide polymorphism (SNP) rs1617640 in the promoter of the Epo gene in patients who underwent7 cardiac operation with cardiopulmonary bypass (CPB). However, an association between the TT genotype and acute renal replacement therapy (p= 0.03), and serum CPK-MB increase (p= 0.03) was observed after operation. They suggest that the risk allele (T) 36 % homozygous genotype distribution of SNP rs1617640 in the promoter of the Epo gene plays a role in renal dysfunction development of after CPB operation. Patients with the TT risk allele required more frequent acute renal replacement therapy. Wu et al induced8 endotoxin shock by IV injection of Escherichia Coli lipopolysaccharide (20 mg/kg) in conscious rats. Endotoxin shock significantly increased blood CPK levels after sepsis. Epo (300 U/kg, IV administration) further increased the CPK markers of organ injury after LPS. Cho et al assigned9 healthy male volunteers to 1 of 2 dose (50 IU/kg or 100 IU/kg) of 3 formulations: an albumin-free rhEPO (Aropotin) and 2 existing rhEPO formulations (Espogen, Recormon) with albumin. After a single 50 or 100 IU/kg dose for Aropotin/Espogen, and for Aropotin/Recormon, the most frequent adverse events (AEs) were 25 % occurrences of elevated serum CPK levels in the Recormon 100-IU/kg group over 32 hours. Groopman found zidovudine toxic10 to myeloid and erythroid precursors in bone marrow with proliferative capability of these progenitor cells reduced ~ 60 %; at concentrations of zidovudine near those associated with the optimal antiviral effect in vitro. Alleviation of zidovudine hematologic toxicity includes the use of hematopoietic growth factors, such as Epo. Anemia, leukopenia and myopathy, also appears to be time and dose dependent. Patients often exhibit an associated elevation in CPK level.
Erythropoietin administration, reperfusion time and their interaction have increasing short- term effect on CPK levels. From clinical situations seems that Epo itself exerts a restorating influence on CPK release on time, during IR injury. The predicted results, adjusted for rats' weight as more reliable, show that this CPK levels rise is not significant yet. This means that the difference of CPK levels from baseline ones is indeed smaller that it seems. So the restorating effect of Epo is really much more powerful than it was believed.
]]> Acknowledgment
This study was funded by Scholarship by the Experimental Research Center ELPEN Pharmaceuticals (ERCE), Athens, Greece. The research facilities for this project were provided by the aforementioned institution.
REFERENCES
1. Gonçalves ES, Rabelo CM, Prado Neto AX. Effect of short-term ornithine alpha-ketoglutarate pretreatment on intestinal ischemia-reperfusion in rats. Acta Cir Bras. 2011;26 (Suppl 1):2-7.
2. Jebeli M, Ghazinoor M, Mandegar MH. Effect of milrinone on short-term outcome of patients with myocardial dysfunction undergoing coronary artery bypass graft: A randomized controlled trial. Cardiol J. 2010;17(1):73-8.
3. Sala MF, Bárcena JP, Rota JI. Sustained ventricular tachycardia as a marker of inadequate myocardial perfusion during the acute phase of myocardial infarction. Clin Cardiol. 2002 Jul;25(7):328-34.
]]>4. Serruys PW, de Feyter P, Macaya C. Fluvastatin for prevention of cardiac events following successful first percutaneous coronary intervention: a randomized controlled trial. JAMA. 2002 Jun 26;287(24):3215-22.
5. Savchuk VI, Vinogradov AV, Pozin VM. Activity of total serum creatine phosphokinase and its MB fraction in reversible coronary blood flow disorders in dogs in a chronic experiment. Kardiologia. 1980 Jan;20(1):58-60.
6. Yang FL, Subeq YM, Chiu YH. Recombinant human erythropoietin reduces rhabdomyolysis-induced acute renal failure in rats. Injury. 2012 Mar;43(3):367-73.
7. Popov AF, Schulz EG, Schmitto JD. Relation between renal dysfunction requiring renal replacement therapy and promoter polymorphism of the erythropoietin gene in cardiac surgery. Artif Organs. 2010 Nov;34(11):961-8.
8. Wu WT, Hu TM, Lin NT. Low-dose erythropoietin aggravates endotoxin-induced organ damage in conscious rats. Cytokine. 2010 Feb;49(2):155-62.
]]>9. Cho SH, Lim HS, Ghim JL. Pharmacokinetic, tolerability, and bioequivalence comparison of three different intravenous formulations of recombinant human erythropoietin in healthy Korean adult male volunteers: an open-label, randomized-sequence, three-treatment, three-way crossover study. Clin Ther. 2009 May;31(5):1046-53.
10. Groopman JE. Zidovudine intolerance. Rev Infect Dis. 1990 Jul-Aug;12(Suppl 5):S500-6.
Recibido: 17 de febrero de 2014.
Aprobado: 17 de abril de 2014.
]]> Tsompos Constantinos. Department of Obstetrics & Gynecology. Mesologi County Hospital. Nafpaktou street. Mesologi 30200. Etoloakarnania. Greece. Tel: 00302631360237. Fax: 2106811215. Email: constantinostsompos@yahoo.com