HUMORAL RESPONSE TO DIFFERENT VACCINATION SCHEMES AGAINST CLASSICAL SWINE FEVER (CSF) SUCCESSIVELY APPLIED DURING AN OUTBREAK OF THE DISEASE
RESPUESTA HUMORAL A DIFERENTES ESQUEMAS DE VACUNACIÓN CONTRA PESTE PORCINA CLÁSICA (PPC) APLICADOS SUCESIVAMENTE DURANTE UN FOCO ACTIVO DE LA ENFERMEDAD
María Irian Percedo*, P. Alfonso*, María Teresa Frías**, Heydi Díaz de Arce**, Maritza Barrera**, O. Fonseca*, Sara Castell**
Grupos de Epizootiología* y Virología Animal**, Centro Nacional de Sanidad Agropecuaria (CENSA), Apartado 10, San José de las Lajas, La Habana, Cuba. Correo electrónico: percedo@censa.edu.cu ]]>
ABSTRACT
Vaccination against classical swine fever (CSF) is an essential measure for its control in endemic areas, and the Chinese Strain has been used with success in already disease free countries. Nevertheless the complexity of the factors interacting on the epidemiology and control of CSF, when the expected success is not reached with the vaccination of premises, doubts always fall on the vaccine used and the vaccination scheme applied In consecuences, many changes in vaccination scheme by the breeders are frecuently observed whitout positive influence in CSF control. To clarify the CSF recurrence in a herd vaccinated by a different scheme as the one recommended in the national control program, serological samples were taken to different animal groups in three moments. Sample I, to evaluate the response to the vaccination scheme decided by producers (litters on 5 and 40 days); and samples II and III, to know its behaviour after the implementation of the scheme recommended by official veterinary service (litters at weaning, at 33 days). The presence of titres =1:50 in all litters at 3 days of age (Sample I), indicated that primo-vaccination at 5 days is not correct. The absence of protective titres on 22% of the animals (9/41), belonging to the groups with clinical cases of the disease, at only 23-30 days from the second doses, confirmed that the double vaccination scheme at 5 and 40 days was not correct. Although at 33 days of age a risk of vaccinating in the presence of high titres of passive antibodies (=1:50) in 65% of the pigs was present (Sampling II), it was verified that at 37 post-vaccination days all pigs showed high neutralizing antibodies titres (Sampling III). This result, together with the clinical behaviour and humoral response of pigs on pre- fattening demonstrated that primo-vaccination at 33 days was adequate and contributed to the control of clinical manifestation of the diseases in the herd.
Key words: classical swine fever; antibodies; vaccination; swine, serology; focus
RESUMEN
La vacunación contra la peste porcina clásica (PPC) es una medida esencial para su control en áreas endémicas, y la cepa china ha sido empleada con éxito en países ya libres de la enfermedad. No obstante la complejidad de los factores que interactúan en la epidemiología y el control de la PPC, cuando no se obtiene el éxito esperado con la vacunación de los rebaños, las dudas siempre recaen en el producto vacunal y su esquema de aplicación. Es así como muchos criadores hacen cambios en los esquemas de vacunación sin que se observen influencias favorables en el control de la enfermedad. Para esclarecer la recurrencia de la enfermedad en un rebaño donde se estaba aplicando un esquema de vacunación diferente al recomendado en el programa de control vigente, se realizaron muestreos serológicos a diferentes grupos de animales en tres momentos: Muestreo I, para evaluar la respuesta al esquema de vacunación orientado por los productores (crías a 5 y 40 días); y Muestreo II y III, para conocer su comportamiento tras la implementación del esquema recomendado por el servicio veterinario oficial (crías al destete, a los 33 días). La presencia de títulos =1:50 en todas las crías a los tres días de nacidas (Muestreo I), indicaron que no era correcta la primovacunación a los cinco días. También la ausencia de títulos protectores en el 22 % de los cerdos (9/41), pertenecientes a los grupos con casos clínicos de la enfermedad, a solo 23-30 días de aplicada la segunda dosis, corroboró que el esquema de vacunación doble a los 5 y 40 días era incorrecto. Aunque a los 33 días de edad se tuvo el riesgo de vacunar en presencia de altos títulos de anticuerpos pasivos (=1:50) en el 65 % de los lechones (Muestreo II), se constató que a los 37 días post-vacunación (DPV) todos los cerdos tenían títulos altos de anticuerpos neutralizantes (Muestreo III). Este resultado, junto al comportamiento clínico y respuesta humoral posterior de los cerdos en preceba, demostraron que la primovacunación a los 33 días fue adecuada y que contribuyó al control de la manifestación clínica de la enfermedad en el rebaño.
Palabras clave: peste porcina clásica; anticuerpos; vacunación; cerdos; serología; foco
]]>
INTRODUCTIONVaccination against Classical Swine Fever (CSF) is an essential measure for its control in areas where the disease is endemic (1; 2), and vaccination with the Chinese Strain is recognized by its effectiveness and contribution to the disease eradication in several countries (3).
In Cuba, CSF was under control through vaccination program among other sanitary measures. There was an epizootic silence for more than 25 years, until its re-emergency in 1993, due to the unfavourable economical situation affecting the sanitary stability of the national swine herd, which in parallel produced an increase of the backyard breeders with the well-known breaches on bio- safety.
Until now, several human and financial resources have been used without the corresponding success on the control of the disease. That is why, the different situations presented as evidence of the problems affecting the control of the disease are always analyzed. Nevertheless, in spite of the complexity of the factors interacting on the epidemiology of the disease and its control, when CSF is introduced or re-emerge on systematically vaccinated herd, doubts falls always on the vaccine or the applied vaccination scheme, in a manner that there is always breeders trying with vaccine schemes different to that recommended by the sanitary authorities.
In this work, the humoral response to the vaccination against CSF obtained by an early immunization scheme in an outbreak of the disease and also the behaviour after the use of recommended by the control program in force used in the country were evaluated.
Therefore, in a CSF re- emerging case where a vaccination program not adjusted to the indications of
the Prevention and Control National Program for CSF (4) was applied, the humoral response to the vaccination schema used was evaluated comparing with the response obtained after the application of the scheme legally in force.
MATERIALS AND METHODS
Experimental design ]]>
In a herd of 450 sows, previously recovered from a CSF outbreak, the animals were maintained under vaccination scheme oriented by the producers (Chinese Strain; LABIOFAM, S.A) to the litters at 5 and 40 days born, to the sows 4 times a year and every 6 months to the sires, as part of the measures for the control of the disease. In order to clarify the recurrence of the disease, a serological test was carried out in the different animal groups (Technological Sections, S) (Sampling I), and the vaccination sheme recommended by official veterinary services was implemented, among another sanitary measures. Sows and litters at post-weaning time (33 days) were vaccinated; sires were vaccinated every six month, as usual.In Sampling, researches regarding 18 pregnant sows between 8 and 11 pregnancy weeks at the final stage of gestation; two groups of 20 litters each at 3 and 33 days of age; 20 and 21 swine of 4 and 5 pre-fattenings sections, were carried out.
After vaccination sheme change, for monitoring the passive immunity of the litters before primovaccination, a sampling was done (Sampling II) using two groups of 10 sows, with two piglets each, at 15 and 33 days post partum, respectively. To evaluate the response to the new vaccination scheme, another serological sampling (Sampling III) was done four months later in six groups of animals (49) in different pre-fattening and fattening technological sections (S). In all cases, blood was obtained through vein.
Antibody detection
Peripheral blood samples without anticoagulant were taken to determine the presence of neutralizing antibodies against classical swine fever virus (CSFV), by neutralizing peroxidase linked assay (NPLA). Assays were done as the technical specification (5) with PK 15 cells on 96 wells plates and 100 DICT 50 of Margarita Strain (6). Sera were evaluated at titres of 1:25 and 1:50 (Sampling I and II). At Sampling III, the humoral response of the young pigs (pre-fattening) at 30 days post-vaccination (PVD) was evaluated at titres from 1:32 to 1:256; and screening test was carried out to the remaining samples at 1:50 dilution as a valid criterion of protection for vaccinated animals (7).
RESULTS AND DISCUSSION
Many questions arise from the introduction or recurrence of CSF in herds, mainly when producers try to fulfil the established measures to prevent and control the disease in endemic areas. When failure on the control occurs, many doubts falls on the quality of vaccines and the effectiveness of the application method, due to the great importance of both aspects.
The national lapinized Chinese strain vaccine, contains 100 protective infectious doses (pid) per animal, recommended by the World Organization for Animal Health (OIE) and its quality is verified by the State Control Laboratory.
Neutralizing antibody titres higher than 32 are accepted as protective after applying the modified life vaccines (7). As vaccinal antigens are capable to present themselves to the immune system through exogenous and endogenous way, they efficiently activate both humoral and cellular immunity which is traduced in a good correlation between the antibody production and Interferon-ã (8; 9). ]]>
The presence of titers =1:50 in every litter at 3 days of age, and the 80% decrease (16/20) at only 28 days after primo-vaccination, demonstrated that the vaccinated scheme in use was unsuitable (5 and 40 days of age) (Table1).The results demonstrated that sows were correctly vaccinated and transfered adequate antibody levels to the litters. Thus, if the litters were vaccinated at 5 days of age, the existent high passive antibody titers would neutralize the vaccinal antigen.
It is generally accepted that passive antibody titers higher than 32 significantly affect the induction of
protecting immunity through vaccination with modified live strains, causing vaccine failures (10; 11; 12).
At the same time, it is recognized that in endemic areas, specially during an active focus of the disease with a high viral circulation, sows previously vaccinated received a constant booster (9).This situation is a problem, because contributes to the increase of titers which are being transferred passively to the litters, probably also conducting to vaccinal failures if this is not taken into account when applying the primo-vaccination.
The Chinese Strain vaccine is capable of inducing complete protection if it is applied at the right moment, when it cannot be interfered with the colostrum antibodies. Thus, in challenged vaccinated pigs an inhibition of both humoral and cellular immunity has been observed, in the same proportion as the higher passive titers at vaccination; and when it occurs, it is corroborated that the 50% of pigs eliminates viruses after challenge (12).
Primo-vaccination in the presence of high passive antibody titres as the cause of vaccination failures and the existence of swine apparently healthy eliminating virus for long periods are considered the main causes of the increasing of focuses in areas where CSF is endemic and vaccination is systematic, as it occurred in Mexico and Thailand during the last years (13; 9).
In the studied herd, an evidence of vaccination failures has occurred due to the presence of CSF in pigs from 9 weeks of birth (pre-fattening Section 4 and 5), where the 22% of the sampled animals (9/41) did not protect titres (=1:50), only at 23 and 30 days after finishing the two-dose vaccination scheme, respectively.
These results helped to the producers to support the proposal to modify the vaccination scheme, starting to apply primo-vaccination at 33 days, at weaning, taking advantage for vaccinating the mothers at the same time.
In Sampling II (Table 2), the two 15 day-pigs with titres of 1:25 belonged to the same litter, indicating that the mother could not transferred a good immunity through its colostrum. Concerning pigs with 33 days of age, the results also indicated the effect of the mother, as one of them had her two pigs negative and other sow had litters with titres of 1:25. These sows were destined to sanitary slaughter after parturition, due to their former poor reproductive performance and as part of the herd sanitation. In general, the low titres obtained in 7 of the litters could be influenced by the deficient immunological status of the sows in which several factors such as nutritional, management or infectious causes could be determined. ]]>
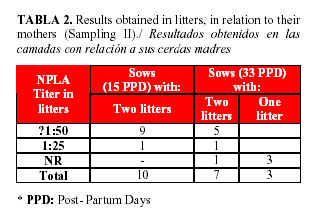
As it was demonstrated, the 33% (7/20) of the litters at 33 days of age (Sampling II, Table 1), did not have protective titres, evidencing the maintaining of a high infection pressure in the herd, even when applying the rest of the sanitary measures. However, vaccinating at that moment was also a risk, due to the presence of high passive antibody titres (=1:50), in 65% of the remaining pigs.
The demonstration of high titres at 37 DPV in all vaccinated 33 days of age pigs (Table 3), together with the gradual decreasing of disease cases in the herd in successive months indicated that the decision of vaccinating at that moment was correct to stop the outbreak.
As in endemic areas, the natural exposure to CSFV can occasionally occur; the sows systematically vaccinated can increase the antibody titres without clinical signs of the disease, and the transfer to their offspring produce interferences on the induction of an active immune response at the vaccination moment.
These findings and others indicating that passive antibody titres higher than 128 were necessary for conferring protection against certain low virulence CSF isolates from Thailand, have motivated the insistence on the necessity to monitore the immunological status of the herds since vaccination failed in inducing protection, particularly in endemic areas where vaccination was overused and pigs were vaccinated at very low age (9). Thus even if many factors could influence on unsuccessful vaccination programs, the difficulties arising from the vaccination of pigs with maternal antibodies are considered one of the most important factor (13; 9).
The high titres observed at 37 days post-vaccination (PVD) in all pigs vaccinated at 33 days of age (Table 3), contrasted with the ones in the animals of similar age (Section 5, Sampling I), only after 30 days from the biphasic vaccination (5 and 40 days), where the 19 % of the animals did not have protective titres (Table 1). Similar favourable results were observed in the following sections on pre-fattening and fattening, although in Section 7 only the 50% of the pigs showed protective titres (Sampling III), in which other processes could influence producing vaccination failures.
It was also pointed out that for vaccinating pigs after 5 weeks of age developed a higher quantity of neutralizing antibodies and Interferon- ã producing specific cells against CSF than the ones vaccinated at 3 weeks (14), because it was recognized that the swine immune system was not completely mature until 4 weeks of age (15). Thus, under outbreak conditions the animals were vaccinated very young with two doses, and the second dose was applied as a booster at 30 days, it was recommended to interrupt this practice as soon as possible (9).
When CSF is introduced in a free country or zone where vaccination is not applied, it is clear that the early vaccination of litters can be the solution for the emergency, until it could be guaranted an adequate protection of the litters through the vaccination of the sows. But in endemic areas, with a systematic vaccination of the herds, in which CSF is not usually present in litters or adult animals, but in pigs after weaning on transition to the fattening, it is evident that the most important problems are failures in the consolidation of post-vaccination active immunity.
With the aim to stop the known losses produced by CSF, producers make mistakes such as the early vaccination of the litters and the indistinctly revaccination of any category. The bad consequences of the wrong use of a vaccine can be harmful as they do not use it on a control strategy.
Another aspect causing disagreements is the frequency of vaccinating sows. In this sense, results referred on a field assay were interesting, the majority of the vaccinated sows were sero-positive between 2,5 and 3 years after vaccination, and less than half of the ones vaccinated under ¨real¨ field conditions were sero- positive a year later (16). ]]>
In practice, special care most be taken on the vaccination of the breeders. In relation to the place where the vaccine is applied and the type of needle used, precaution has to be taken in applying the injection in the muscle (not in the subcutaneous fat layer) in which the length of the needle is important.On big animals, it is recommended to apply the vaccine in the lateral-cervical region behind the ear using needle 1-1.5 x 18, with the precaution not to remove it quickly after applying the product to avoid the reflux of the liquid (4).
It is considered that the change on the vaccination scheme, together with the rest of the sanitary measures strictly applied, decisively contributed to the gradual herd recovering, demonstrated by the not-appearance of new sick animals and the improvement of the bio-productive indicators during the period evaluated.
ACNKNOWLEDGES
To Drs. Yanelis Díaz, Roberto Pérez, Roberto González and María Reyes, as well as to all technicians and workers of the swine premise, for the cooperation in the sampling and data collection in the field.
REFERENCES
1. Moennig V. Introduction to classical swine fever virus, disease and control policy. Vet Microbiol. (2000);73:93-102.
2. Morilla GA, Carvajal MA. La fiebre porcina clásica endémica en México. Ciencia Veterinaria. (2004); 9-2003-4, 166-190. Rev. 22/2/2008 http://www.rlc.fao.org/prior/segalim/animal/ppc/plan /paises/pdf/mexico.pdf
3. Espuña E. Inmunización frente a la peste porcina clásica. Porci. Aula Veterinaria. 1994;22:45-56.
4. Instituto de Medicina Veterinaria (IMV). Programa Nacional de Prevención y Control de la peste porcina clásica. 2005. Imprenta MINAG.
5. World Organization for Animal Health. OIE. Terrestrial Manual. Chapter 2.8.3. Classical swine fever (hog cholera). 2008; 1098-1000.
6. Ganges L, Barrera M, Díaz de Arce H, Vega A, Núñez JI, Sobrino F, Frías MT. Antigenic, biological and molecular characterization of the Cuban CSFV isolate "Margarita". Rev Salud Anim. 2007;29(3):182-192.
7. Terpstra C, Wensvoort G. The protective value of vaccineinduced neutralising antibody titres in swine fever. Vet Microbiol. 1988;16:123-128.
8. Suradhat S, Intrakamhaeng M, Damrongwata-napokin S. The correlation of virus-specific interferon-gamma production and protection against classical swine fever virus infection. Vet Immunol Immunopathol. 2001;83:177-189.
9. Suradhat S, Damrongwatanapokin S, Thanawong-nuwech R. Factors critical for successful vaccination against classical swine fever in endemic areas. Vet Microbiol. 2007;119:19.
10.Suvintrakorn K, Hanveeraphon A, Pariyakanok V. Efficacy of lapinized swine fever vaccine in piglets born from immune sows. J Thai Vet Med Assoc. 1993;23:93-103.
11.Parchariyanon S, Tantaswasdi U, Pinyochon W, Methiyapun P. Immunity against swine fever vaccine. II. Immunity against swine fever vaccine in piglets and protection level of maternal immunity in piglets before vaccination. J Thai Vet Med Assoc. 1994;45:37-45.
12.Suradhat S, Damrongwatanapokin S. The influence of maternal immunity on the efficacy of a classical swine fever vaccine against classical swine fever virus, genogroup 2.2, infection. Vet Microbiol. 2003;92:187-194.
13.Morilla GA. A decade of learning to control CSF in endemic areas. 17 th Cong. IPVS (Int. Pig Vet. Socc.), 2002. Iowa, USA.
14.Suradhat S, Damrongwatanapokin, S. Factors that influenced an effectiveness of classical swine fever vaccine: a case study. Thai J Vet Med. 2002;32 (Suppl):163-172 (in Thai).
15.Povey RC, Carman PS. Technical basis of vaccination. En: Pastoret, P.-P., Blancou, J., Vannier, P., Verschueren, C. (Eds.),Veterinary Vaccinology. Elsevier Science B.V., Amsterdam. 1997. pp. 519-580.
16.Van Bekkum JG. Experience in the Netherlands with the lapinised so-called Chinese (C) strain of vaccine. Eradication of classical swine fever in Hungary. In: Proceedings of the CEC Seminar on Hog Cholera/Classical Swine Fever and African Swine Fever. EUR 5904 EN, Hannover. 1977. pp. 379-391.
(Recibido 15-3-2009; Aprobado 15-5-2009) ]]>