Mass spectroscopy investigation of the effect of gamma irradiation
on the mean value of the number of ethoxy groups in the TRITON X-100
Estudio del efecto de la radiación gamma en elvalor medio del número de grupos etoxilados
del surfactante TRITÓN X-100
Gilmer Valdés Díaz1, Simón Rodríguez Calvo1, Aurora Pérez Gramatges1, Manuel Rapado Paneque2, Francisco Alberto Fernandez Lima1, Cassia Ribero R. Ponciano3, Enio Frota da Silveira3
1Instituto Superior de Tecnologías y Ciencias Aplicadas (InSTEC), Ave. Salvador Allende, esq. Luaces, Plaza, Ciudad de La Habana, Cuba
2Centro de Centro de Aplicaciones Tecnológicas y Desarrollo Nuclear (CEADEN), Calle 30 No. 502 e/ 5ta Ave. y 7ma, Playa, Ciudad de La Habana, Cuba ]]>
3Departamento de Física, Pontifícia Universidade Católica, Rio de Janeiro, Brasil
gvaldes@instec.
ABSTRACT
The effect of gamma radiation from a 60Co source on the structure of a nonionic surfactant, namely
TRITON X-100, was investigated. Three main regions can be distinguished in the behavior of the mean value of ethoxy groups with an increase in the absorbed dose. However just a slightly decrease on this mean value was obtained when the dose range from 0 to 70 kGy.
RESUMEN
El efecto de la radiación gamma proveniente de una fuente de 60Co en la estructura del surfactante no iónico Tritón X-100 fue investigado. Tres regiones principales pueden ser distinguidas en el comportamiento del valor medio del número de grupos etóxidos al aumentar la dosis. Sin embargo, el resultado global encontrado fue una pequeña variación en este valor medio al cambiar la dosis entre 0 y 70 KGy.
Key words: gamma radiation, cobalt 60, radiation doses, mass spectroscopy, sterilization, radiation dose distributions, polymers
INTRODUCTION ]]>
Studies on radiation chemistry in micellar systems is a relatively new field due to the fact that these systems can resemble biological tissues, and be therefore used in different pharmaceutical applications. A classical example of the biological importance of these systems is the study of lung surfactant solutions, which are used as a model to determine the type and extent of possible radiation effects in this organ [1,2]. Micellar systems have also being used in the encapsulation of drugs and labelled compounds, such as radiopharmaceuticals for therapy and diagnosis in medical applications.
For example, injectable delivery systems, such as polymeric microspheres, and carriers used in gene therapy, such as amphiphilic aggregates (liposomes and niosomes), are intended for intravitreal administration and have to meet the pharmacopoeia requirements of sterility. In the pharmaceutical field, one of the applications of ionizing radiation is the final sterilization of these biodegradable materials intended for parenteral use. A minimum absorbed dose of 25 kGy is regarded as adequate for the purpose of sterilizing pharmaceutical products without providing any biological validation [3].
Gamma-radiation as a form of electromagnetic radiation, characterized by high penetration at a very low dose rate, can modify the performance of irradiated drug delivery systems, prolonging the peroxidative radiolitic mechanism due to the exposure time. However, the degradation products generated can significantly alter the aqueous microenvironmental conditions, e.g. H+ concentration within the system.
The effects of ![]() -irradiation on polymer microspheres have been reported by different authors, depending on the type of polymer and the active component [4-6]. However, little information is till now available in the literature on the effect due to the use of such excipients on the biopharmaceutical performance of
-irradiation on polymer microspheres have been reported by different authors, depending on the type of polymer and the active component [4-6]. However, little information is till now available in the literature on the effect due to the use of such excipients on the biopharmaceutical performance of ![]() -irradiated microparticulate systems. The study of the effects of
-irradiated microparticulate systems. The study of the effects of ![]() -irradiation on new formulations such as microspheres intended for intravitreal administration becomes necessary also because local toxicity is related to particular properties that can be affected by sterilisation.
-irradiation on new formulations such as microspheres intended for intravitreal administration becomes necessary also because local toxicity is related to particular properties that can be affected by sterilisation.
In this work, we investigated the effect of the gamma radiation from ![]() on the mean value of the number of ethoxy groups in micellar solutions of the nonionic surfactant TRITON X-100, a very common and widely used tensoactive both in the cosmetics and pharmaceutical industries as solubilizer, emulsifier and detergent. In that way, the effect of the gamma radiation in the head of the surfactant is assessed.
]]>
on the mean value of the number of ethoxy groups in micellar solutions of the nonionic surfactant TRITON X-100, a very common and widely used tensoactive both in the cosmetics and pharmaceutical industries as solubilizer, emulsifier and detergent. In that way, the effect of the gamma radiation in the head of the surfactant is assessed.
]]>
MATERIALS AND METHODS
Reagents
The nonionic surfactant used was polyoxyethylene-toctylphenyl ether, with an average of 9.5 oxyethylene (EO) units per molecule (Triton X-100, figure 1), from Rohm & Haas Co. Surfactant concentration was 1% (wt), ca. 1.6 x ![]() mol
mol ![]() which is well above the CMC of Triton X-100 (2.4 x
which is well above the CMC of Triton X-100 (2.4 x ![]() mol
mol ![]() [7]); the percentage of surfactant is based on the amount of water present (in all the experiments distilled water was used). The surfactant was used as received.
[7]); the percentage of surfactant is based on the amount of water present (in all the experiments distilled water was used). The surfactant was used as received.
The mixtures were prepared 24 hours in advance to ensure full hydration of micelles. The cloud point under these conditions was 64.5oC.
Gamma irradiation of the samples
Mass spectrometry analysis
The mass spectrometry analysis was performed in the reflective mode of a BRUKER / BIFLEX III mass spectrometer, equipped with a 337 nm UV nitrogen laser (3 ns FWHM, 200 µJ mean energy per pulse) from Laser Science Inc. Samples were analyzed using Laser Desorption Ionization (LDI) and Matrix Assisted Laser Desorption Ionization (MALDI). The standard dried droplet method was used for the sample preparation and a TX-100 typical concentration of ![]() w/w was used. The TX-100 molecule ionization was ensured by applying small quantities of Na and K ions in the sample solution. For the LDI and MALDI analyses a laser intensity of 0.75 and 0.15 GW
w/w was used. The TX-100 molecule ionization was ensured by applying small quantities of Na and K ions in the sample solution. For the LDI and MALDI analyses a laser intensity of 0.75 and 0.15 GW![]() was used, respectively. The 4-Hydoxy-
was used, respectively. The 4-Hydoxy-![]() -cyanocinnamic acid (
-cyanocinnamic acid (![]() -CHCA) was used as a matrix for the MALDI analysis, with a concentration of analyte (TX-100) to matrix (
-CHCA) was used as a matrix for the MALDI analysis, with a concentration of analyte (TX-100) to matrix (![]() -CHCA) molecules of 1:10. All the mass spectra correspond to an average of 15 and 10 laser shots
-CHCA) molecules of 1:10. All the mass spectra correspond to an average of 15 and 10 laser shots
for LDI and MALDI analyses, respectively.
RESULTS AND DISCUSSION
Mass Spectrometry and structural changes Commercial polyethoxylated surfactants, such as Triton X-100, are obtained as a polymeric distribution having the same tail structure but different head, with a mean value of ethoxylated (EO) groups of 9.5. To investigate the effect of the dose in this mean value a mass spectrometry technique was used. A typical mass spectrum of the non irradiated surfactant solution at 1% wt shows a distribution of peaks between 449 and 861, which corresponds to a distribution range of EO units around 9 (figure 2).
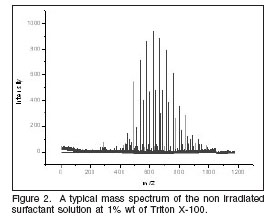

Small aliquots of Triton X-100 aqueous solutions (1% wt) were irradiated at doses between 0.1 and 100 kGy, and analysed using the mass spectrometry technique. The mean distribution values were calculated using the same method and compared to the non irradiated (figure 4).

The ![]() values for very low doses (less than 1kGy) show an average value of 9.25 ± 0.31, and a slight decrease is observed towards a dose value of 50 kGy. For larger doses, the change in the mean values relative to the non-irradiated sample is equivalent to losing one group in the EO polymeric chain. These changes as a function of the irradiation dose are probably a consequence of the indirect action of gamma radiation on surfactant molecules, i.e. the interaction with free radical products of the water radiolysis. The direct interaction is less efficient, considering that the irradiation was performed in a dilute aqueous solution.
values for very low doses (less than 1kGy) show an average value of 9.25 ± 0.31, and a slight decrease is observed towards a dose value of 50 kGy. For larger doses, the change in the mean values relative to the non-irradiated sample is equivalent to losing one group in the EO polymeric chain. These changes as a function of the irradiation dose are probably a consequence of the indirect action of gamma radiation on surfactant molecules, i.e. the interaction with free radical products of the water radiolysis. The direct interaction is less efficient, considering that the irradiation was performed in a dilute aqueous solution.
For the indirect interaction, there are three possible domains in the chemical structure of Triton X-100 (figure 1) that can be sensitive to these radical attacks: the hydrocarbon chain, the aromatic ring and the EO polymeric unit. However, considering that these solutions were well above CMC of surfactant, it is logical to assume that most of the primary degradation of the surfactant molecule would occur on the polyoxyethylene chain (EO), due to the shielding effect that the ethoxylated groups have over the tail [8]. A similar decrease was reported by Pellizeti et al. for the mean value of the EO groups, due to the action of hydroxyl (OH) radicals in an aqueous system of a nonionic ethoxylated surfactant (Igepal CO-720, 6.0 x ![]() mol
mol ![]() , [9]). In an early report on effects of gamma irradiation upon aqueous solutions of different kinds of surfactants, it was found that bond cleavage of oxyethylene in polyoxyethylene surfactant (POE) was the main chemical reaction occurring after radiolysis of water [10].
, [9]). In an early report on effects of gamma irradiation upon aqueous solutions of different kinds of surfactants, it was found that bond cleavage of oxyethylene in polyoxyethylene surfactant (POE) was the main chemical reaction occurring after radiolysis of water [10].
CONCLUSIONS
REFERENCES
1. DELGADO A, SORIANO I, SANCHEZ E, OLIVA M, EVORA C. Eur J Pharm Biopharm. 2000;( 50): 227.
2. Forsberg S, Johansson JM. Lindskoug B. Life Scien.1970; 14 ( 2): 795.
3. European Guideline 3AQ4a. The use of ionising radiation in the manufacture of medicinal products. Official Publications in the European Communities London, 1992.
4. MARTÍNEZ-SANCHO C, HERRERO-VANRELL R, NEGRO S. J Controlled Release. 2004;( 99): 41.
5. MONTANARI L, CILURZO F, VALVO L, FAUCITANO A, BUTTAFAVA A, GROPPO A, GENTA I, CONTI B. J Controlled Release. 2001;( 75): 317.
6. DORATI R, GENTA I, MONTANARI L, CILURZO F, BUTTAFAVA A, FAUCITANO A, CONTI B. J Controlled Release. 2005;( 107): 78.
7. ROSS J, OLIVIER JP. J Phys Chem. 1959;( 63): 1671.
8. Henglein A, PROSKE TH. Makromol Chem. 1978;( 179): 2279.
9. Pelizetti E, Minero C, MAURINO V, SCIAFANI A, HIDAKA H, SERPONE N. Environ Sci Technol. 1989;( 23): 1380-1.
10. PERKOWSKI J, MAYER J. J Radioanal Nuclear Chem Articles. 1992;( 157): 27.