
RESEARCH
Induction of chitinases and glucanases in Trichoderma spp. strains intended for biological control
Inducción de quitinasas y glucanasas en cepas de Trichoderma spp. promisorias como agentes para el control biológico
]]> Ivonne González1, Danay Infante1, Benedicto Martínez1, Yailén Arias1, Noyma González1, Ileana Miranda2, Belkis Peteira1
1Grupo de Fitopatología.
2Grupo de Plagas Agrícolas, Dirección Protección de Plantas, Centro Nacional de Sanidad Agropecuaria, CENSA. AP 10, San José de las Lajas, Mayabeque, Cuba.
ABSTRACT
Use of fungi of the Trichoderma genus for the biological control of pests and diseases is based, to a large extent, on their secretion of hydrolytic enzymes. The latter include chitinases and glucanases, which degrade the cell wall of phytopathogenic fungi as well as the cuticle of insects and nematodes. The aim of this study was to assess the induction dynamics of chitinases and glucanases in ten strains of Trichoderma spp. grown in liquid media with different inducers: basal medium, basal medium supplemented with 0.5% chitin and basal medium supplemented with 0.2% gelatin. Chitinase and glucanase activity were evaluated at the first, third, fifth and seventh day of culture. The highest values of chitinase activity were obtained in basal and basal media supplemented with chitin; β-1.3-glucanase, on the other hand, exhibited higher levels of activity in basal and basal media supplemented with gelatin.
Keywords: Trichoderma, chitinases, glucanases, biological control.
La secreción de enzimas hidrolíticas es uno de los mecanismos de los hongos del género Trichoderma, para ejercer el control biológico de plagas y enfermedades. Las quitinasas y glucanasas son dos de estas enzimas que degradan la pared celular de los hongos fitopatógenos y la cutícula de insectos y nematodos. Se evaluó la dinámica de inducción de esas enzimas en diez cepas de Trichoderma spp. en tres medios líquidos con inductores diferentes: medio basal, medio basal suplementado con quitina al 0.5% y medio basal suplementado con gelatina al 0.2%. Las actividades quitinasas y glucanasas se evaluaron al primer, tercer, quinto y séptimo día del cultivo. Los mayores valores de la actividad quitinasa se alcanzaron en los medios basal y basal suplementado con quitina, mientras que los medios basal y basal suplementado con gelatina resultaron mejores inductores de las β-1.3-glucanasas.
Palabras clave: Trichoderma, quitinasas, glucanasas, control biológico.
INTRODUCTION
Fungi from the genus Trichoderma have been widely used as biological control agents under both greenhouse and open fi eld conditions, due to their ability to prey on a diverse range of foliar and soil phytopathogens [1-3]. These fungi employ different strategies against their targets: competition for space and nutrients, mycoparasitism, production of inhibitory compounds, inactivation of enzymes from the pathogenic agent, and the induction of resistances [4-6].
Trichoderma parasitizes the hyphae of phytopathogenic fungi using prehensile coils and hooks that penetrate their cell walls, aided by the hydrolytic activity of chitinases and glucanases [4]. Cell walls of phytopathogenic fungi such as Sclerotium rolfsii, Rhizoctonia solani and Pythium spp. [7] are composed mainly of β-1.3-glucans and chitin, including also cellulose in some oomycetes, such as Pythium spp. [8].
Chitinases and glucanases can also degrade the cuticle of insects, composed mainly of chitin [9-12]. Chitin is also present at the cuticle of nematode eggs, whose viability decreases severely when treated with Trichoderma species [13, 14].
Different strains of Trichoderma usually exhibit different levels of expression of these hydrolytic enzymes, leading in turn to differences in performance when used as biological control agents. The objective of the present work, therefore, was to study the induction dynamics of chitinases and glucanases in a set of Trichoderma strains intended for biological control, employing three liquid media with different inductors: basal medium, basal medium supplemented with 0.5% chitin and basal medium supplemented with 0.2% gelatin.
]]> MATERIALS AND METHODS
Strains and induction media
This work employed strains 1, 13, 17, 25, 75, 78, 79, 85 and 90 of Trichoderma sp. from the strain collection of the Plant Mycology laboratory of the National Center for Agricultural Health (CENSA), classified through molecular biology methods as belonging to the species Trichoderma asperellum Samuels [15], and strain TS3, from the Institute for Plant Health Research, INISAV; all selected based on their capacity for antagonizing with known phytopathogens [16]. The strains were stored in Malt-Agar medium (National Center for Biopreparations, Biocen) at 4 °C. In order to perform the dynamics experiment, the strains were subcultured in potato-dextrose-water medium (PDA, Biocen) using 9 mm Petri dishes and incubated statically for 3 days at 28 °C, after which the mycelia were harvested for further experimentation.
Protein induction in liquid media
The protein induction experiment used liquid media containing different compounds, selected on the basis of their ability to induce the synthesis of hydrolytic enzymes: liquid basal medium containing yeast extract at 1 g/L and peptone at 4 g/L, liquid basal medium supplemented with chitin at 5 g/L, and liquid basal medium supplemented with gelatin at 0.2% (w/v) [17]. The first two were sterilized at 120 °C for 20 min, whereas the medium containing gelatin was sterilized at 115 °C for 15 min.
The media were aliquoted in portions of 20 mL into 100 mL bottles, and were then inoculated with four mycelial disks with a diameter of 5 mm, taken from the periphery of a pure colony from each strain, and incubated statically at 28 °C, protected from light. Each treatment was repeated three times, using three replicates in each occasion. Bottles were withdrawn from the incubator at day 1, 3, 5 and 7 after inoculation. Culture supernatants were filtered on Whatman paper (3 mm CHR), and the fi nal extracts were stored at -20 °C until used in enzyme assays.
Protein quantification
Total protein concentration was determined with the method described by Bradford [18], reading the absorbance of the protein-Coomassie Blue G-250 complex at 595 nm in a spectrophotometer (Lasso Spec III, Lasso Biotech LTDA) and interpolating the obtained values into the absorbances of a standard curve prepared from a 1 mg/mL stock solution of Bovine Serum Albumin (BSA).
Enzyme assays
Chitinase activity was determined by mixing 0.5 mL of the culture supernatant to be assayed with 0.2 mL of colloidal chitin at 10 mg/mL, prepared according to Boller et al. [19], incubating the mixture at 37 °C for 1 h and then adding 0.1 mL of 0.8 M sodium tetraborate,pH 8.8. The resulting mixture was centrifuged at 6708 x g for 5 min, transferring then 500 μL of the supernatant to a glass tube and heating it to 100 °C for 3 min. Afterward, 1 mL of p-dimethyl amino benzaldehyde was added, incubating the solution at 38 °C for 20 min. A standard curve was prepared from an Nacetyl glucosamine stock at 1 mg/mL and processed as described above for the samples. Optical density was measured at 585 nm. Enzyme activity was calculated using the following expression:
]]>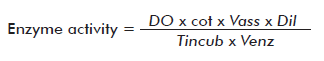
Where OD: optical density; cot: cotangent of the angle of the standard curve; Vass: assay volume; Dil: dilution; Tincub: incubation time and Venz: enzyme volume. Activity is expressed in μmol of formed product/min·mL of enzyme.
Determinations of enzyme activity for β-1.3-glucanases from culture supernatants used a method employing 96-well microtiter plates, developed by Zheng and Wozniak [20].
Specific activity was determined using the expression:

A one-way ANOVA was used to determine the existence of statistically significant differences between strains regarding the specific activity of chitinases and glucanases in each culture medium. Means were compared with Duncan’s multiple ranks test in cases where there were differences (p < 0.05). In order to identify culture media and culture times favoring the induction of chitinase and glucanase activities, a main components analysis was performed with the InfoStat version 2009 statistical software package [21].
RESULTS AND DISCUSSION
Species of the Trichoderma genus are frequently used as biological control agents due to their ability to parasitize a wide variety of phytopathogenic fungi [14]. Both chitinases and glucanases are involved in the development of this parasitic association [22]. Although T. asperellum is one of the less studied species of the genus [23], it has served however as the source from which a number of endochitinases and β-1.3-glucanases have been purifi ed; in addition, the patterns of expression of these enzymes, depending on the carbon source of the culture medium, have also been studied [23-25]. This research has demonstrated that enzyme expression is infl uenced by the type of strain, culture conditions and the substrate employed in the production of cell wall-degrading enzymes [26, 27].
The strains studied in the present work were assigned, based on the level of expression of their chitinases, to five different groups: I (75), II (78), III (1, 79, 85), IV (13, 17, 25, 90) and V (TS3) (Figure 1). Strain 75 exhibited the highest chitinase activity. Liquid media maximizing this expression were basal medium, at days 5 and 7 post-inoculation, and basal medium supplemented with chitin, at day 1 post-inoculation.
By day 7 post-inoculation, the basal medium supplemented with chitin induced high levels of chitinase in strain 78. Strain TS3 exhibited only low levels of chitinase activity, which peaked at day 1 post-inoculation in basal medium supplemented with gelatin. The strains from groups III and IV exhibited intermediate levels of chitinase activity. Chitinase activity was favored, in the fi rst group, by basal medium (at day 1) and basal medium supplemented with gelatin (at day 7). In the second group, the expression of this enzyme was favored by basal medium (at day 3) and basal medium supplemented either with chitin or gelatin (at day 5).
Based on observed glucanase levels, the analyzed strains cluster into four groups (Figure 2). In this occasion group I was comprised of strains 1, 13, 17 and 25; group II was comprised of strain 75; group III was comprised of strains 79, 85, 90 and TS3, and group IV included only strain 78.
Basal medium alone (at days 1 and 5) or supplemented with gelatin (at day 1) induced the highest levels of enzyme activity in group I, whereas group II was favored by basal medium (at day 7) and basal medium supplemented with gelatin (at day 3). The remaining strains exhibited low enzyme activities throughout the experimental period.
]]> The medium with gelatin induced the highest β-1.3-glucanase activity in strains 1, 13, 17, 25 and 75 of T. asperellum. Marcello et al. demonstrated that T. asperellum produces high levels of β-1.3-glucanase when grown in media containing starch and cell walls from R. solani [23].The characterization of both enzymes (chitinases and glucanases) is a useful tool for the selection, together with other parameters, of the best isolates for biological pest control purposes.
Strain 75 of T. asperellum exhibited the highest levels of chitinase and glucanase activity (Figure 3), followed by strain 78 and the group formed by strains 1, 13, 17 and 25. In all cases, strain TS3 had the lowest levels of enzyme activity.
Strains 17, 75 and 78 have previously been used with success for the biological control of S. rolfsii and R. solani [15]. This result is consistent with the high levels of chitinase and glucanase secretion exhibited by these strains during the present study.
The levels of chitinase and β-1.3-glucanase in culture supernatants of strain TS3, a strain used for the control of phytoparasitic nematodes of the Meloidogyne genus [28], were low under all culture conditions. It should be borne in mind, however, that mycoparasitism involves not only chitinases and β-1.3-glucanases, but also β-1.6-glucanases, α-1.3-glucanases and proteases, which have not been evaluated in the present study and might play an important role in the antagonism of this strain towards phytopathogens [5, 29]. An example of the latter possibility has been the development of Beauveria bassiana (Balsamo) Vuillemin transformants expressing a protease-chitinase fusion CDEP1:Bbchit1), which exhibit much faster penetration kinetics towards insect cuticles [30]. Strain TS3 might also be simply outcompeting pathogens for physical space and nutrients, secreting inhibitor compounds [4] or inducing resistance [6]; these are all well-documented mechanisms by which Trichoderma antagonizes phytopathogens. Dennis and Webster [31], as well as Elad and Henis [32], have demonstrated the production of antibiotic and lytic intracellular enzymes in a number of Trichoderma spp. strains, which were shown to participate and be responsible for the antagonizing activity of the fungus. Trichoderma harzianum antagonized with Meloidogyne incognita eggs through the production of anti-nematode compounds that limited the penetration of this pathogen through the roots, either affecting directly the nematodes, or simply making the roots less attractive.
The basal medium was a very effective inducer of the enzyme activities assayed in the present work. Strains 75 and 78 showed the greatest potential for future application as biofungicidal products.
REFERENCES
1. Jayalakshmi SK, Raju S, Usha Rani S, Benagi VI, Sreeramulu K. Trichoderma harzianum L. as a potential source for lytic enzymes and elicitor of defense responses in chickpea (Cicer arietinum L.) against wilt disease caused by Fusarium oxysporum f. sp. ciceri. Aust J Crop Sci. 2009;3(1):44-52.
]]>2. Tondje P, Roberts DP, Bon M, Widner T, Samuels GJ, Ismaiel AA, et al. Isolation and identification of mycoparasitic isolates of Trichoderma asperellum with potential for suppression of black pod disease of cacao in Cameroon. Biol Control. 2007;43:202-12.
3. Küçük Ç, Kıvanç M. Mycoparasitism in the biological control of Gibberella zeae and Aspergillus ustus by Trichoderma harzianum strains. J Agric Technol. 2008; 4(2):49-55.
4. Zeilinger S, Omann M. Trichoderma biocontrol: signal transduction pathways involved in host sensing and mycoparasitism. Gene Regul Syst Bio. 2007;1:227-34.
5. Verma M, Brar SK, Tyagi RD, Surampalli RY, Valéro JR. Antagonistic fungi, Trichoderma spp.: Panoply of biological control. Biochem Eng J. 2007;37(1):1-20.
6. Kapulnik Y, Chet I. Induction and accumulation of PR proteins activity during early stages of root colonization by the mycoparasite T. harzianum strain T-203. Plant Physiol Biochem. 2000;38:863-73.
7. Chet I. Trichoderma-application and mode of action, and potential as biocontrol agent of soilborne plant pathogenic fungi. In: Chet I, editor. Innovative approaches to plant disease control. New York: John Wiley; 1987. p. 137-60.
8. Bartnicki-Garcia S. Fundamental aspects of hyphal morphogenesis. In: Ashworth JM, Smith JE, editors. Microbial differentiation. Cambridge: Cambridge University Press; 1973. p. 245-68.
9. Sharma P, Pandey R. Biological control of root-knot nematode; Meloidogyne incognita in the medicinal plant; Withania somnífera and the effect of biocontrol agents on plant growth. Afr J Agric Res. 2009;4(6):564-7.
10. Shakeri J, Foster HA. Proteolytic activity and antibiotic production by Trichoderma harzianum in relation to pathogenicity to insects. Enzyme Microb Technol. 2007;40(4):961-8.
11. Goswami J, Pandey RK, Tewari JP, Goswami BK. Management of root knot nematode on tomato through application of fungal antagonists, Acremonium strictum and Trichoderma harzianum. J Environ Sci Health. Part B: Pestic, Food Contam, Agric Wastes. 2008;43(3):237-40.
12. Bokhari F. Efficacy of some Trichoderma species in the control of Rotylenchulus reniformis and Meloidogyne javanica. Arch Phytopathol Plant Prot. 2009;42(4): 361-9.
13. Haggag WM, Amin AW. Efficiency of Trichoderma species in control of Fusarium-rot, root knot and reniform nematodes disease complex on sunflower. Pakistan J Biol Sci. 2001;4(3):314-8.
14. Jin RD, Suh JW, Park RD, Kim YW, Krishnan HB, Kim KY. Effect of chitin compost and broth on biological control of Meloidogyne incognita on tomato (Lycopersicon esculentum Mill.). Nematology. 2005;7:125-32.
15. Martínez B, Infante D, Reyes Y. About to identification of some Trichoderma isolates reported in Revista de Protección Vegetal. Rev Protección Veg. 2010;25(2):135.
16. Martínez B, Reyes Y, Infante D, González E, Baños H, Cruz A. Selección de aislamientos de Trichoderma spp. candidatos a biofungicidas para el control de Rhizoctonia sp. en arroz. Rev Protección Veg. 2008;23(2):118-25.
17. Peteira B, Estévez I, Montes de Oca N, Hidalgo-Díaz L. Estabilidad de la cepa IMI SD 187 de Pochonia chlamydosporia var. catenulata en medio sólido. Rev Protección Veg. 2007;22(2):124-7.
18. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248-54.
19. Boller T, Gehri A, Mauch F, Vögeli U. Chitinase in bean leaves: induction by ethylene, purification, properties and possible function. Planta. 1983;157:22-31.
20. Zheng Y, Wozniak CA. Adaptation of a beta-1,3-glucanase assay to microplate format. Biotechniques. 1997;22(5):922-6.
21. Di Rienzo JA, Balzarini M, Gonzalez L, Casanoves F, Tablada M, Robledo C. InfoStat. Software estadístico [Internet]. Versión 2009. Córdoba: Universidad Nacional de Córdoba, Argentina; 2009.
22. Limon MC, Pintor-Toro JA, Benitez T. Increased antifungal activity of Trichoderma harzianum transformants that overexpress a 33-kDa chitinase. Phytopathology. 1999;89(3):254-61.
23. Marcello CM, Steindorff AS, da Silva SP, Silva Rdo N, Mendes Bataus LA, Ulhoa CJ. Expression analysis of the exo-beta-1,3-glucanase from the mycoparasitic fungus Trichoderma asperellum. Microbiol Res. 2010;165(1):75-81.
24. Viterbo A, Montero M, Ramot O, Friesem D, Monte E, Llobell A, et al. Expression regulation of the endochitinase chit36 from Trichoderma asperellum (T. harzianum T-203). Curr Genet. 2002;42(2):114-22.
25. Bara MT, Lima AL, Ulhoa CJ. Purification and characterization of an exo-beta-1,3-glucanase produced by Trichoderma asperellum. FEMS Microbiol Lett. 2003;219(1):81-5.
26. Howell CR. Mechanisms employed by Trichoderma species in the biological control of plant diseases: the history and evolution of current concepts. Plant Dis. 2003;87(1):4-10.
27. Harman GE, Howell CR, Viterbo A, Chet I, Lorito M. Trichoderma species--opportunistic, avirulent plant symbionts. Nat Rev Microbiol. 2004;2(1):43-56.
28. Muiño BL, Botta E, Pérez E, Moreno D, Fernández E. Uso de Trichoderma como alternativa al bromuro de metilo en los cultivos protegidos, flores y ornamentales en Cuba. Fitosanidad. 2006;10(2):179-80.
29. Radjacommare R, Venkatesan S, Samiyappan R. Biological control of phytopathogenic fungi of vanilla through lytic action of Trichoderma species and Pseudomonas fluorescens. Arch Phytopathol Plant Prot. 2010;43(1):1-17.
30. Fang W, Feng J, Fan Y, Zhang Y, Bidochka MJ, Leger RJ, et al. Expressing a fusion protein with protease and chitinase activities increases the virulence of the insect pathogen Beauveria bassiana. J Invertebr Pathol. 2009;102(2):155-9.
31. Dennis C, Webster J. Antagonistic properties of species groups of Trichoderma. II. Production of Volatile antibiotics. Trans Br Mycol Soc. 1971;57:41-8.
32. Elad Y, Chet I, Henis Y. Degradation of plant pathogenic fungi by Trichoderma harzianum. Can J Microbiol. 1982;28(7):719-25.
Received in June, 2011.
Accepted for publication in November, 2011.
Ivonne González. Dirección Protección de Plantas, Centro Nacional de Sanidad Agropecuaria, CENSA. AP 10, San José de las Lajas, Mayabeque, Cuba. E-mail: marquetti@censa.edu.cu.
]]>