 ]]>
]]>
Preguntas y respuestas sobre aspectos generales del tratamiento farmacológico en el paciente obeso
Questions and answers about general aspects of the pharmacotherapy for the obese patients
Dr. José Hernández Rodríguez, Dr. Manuel Emiliano Licea Puig, Dra. Lizet Castelo Elías-Calles
Centro de Atención al Diabético. Instituto Nacional de Endocrinología (INEN). La Habana, Cuba. ]]>
RESUMEN
Introducción: a través del uso de la farmacoterapia en el paciente obeso se ha intentado obtener pérdidas de peso, mayores y más rápidas que las reportadas con los métodos convencionales de tratamiento, lo que sin dudas facilita una disminución objetiva de la morbilidad y mortalidad observada en los pacientes con este problema de salud.
Objetivos: describir los aspectos generales del tratamiento farmacológico en el paciente obeso y mencionar algunos de los medicamentos usados con este fin.
Desarrollo: resulta importante conocer los aspectos generales del tratamiento farmacológico de la obesidad. Consideraciones sobre cómo hacer la selección del fármaco ideal, los principios generales para su uso, cuáles son los criterios de elección de los pacientes y qué fármacos deben ser evitados en el paciente con obesidad, son elementos útiles a conocer para garantizar una asistencia médica de calidad a las personas con obesidad. Productos conocidos e indicados como tratamiento farmacológico de la obesidad pasaron al desuso, por su pobre efectividad o por presentar toda una serie de efectos secundarios. El uso de orlistat o xenical debe de ser la alternativa más común en el tratamiento actual con fármacos para la obesidad. La reciente aprobación de nuevos medicamentos como la lorcaserina (belviq) y qnexa (o qsymia), debe representar una opción de utilidad.
Conclusiones: el conocimiento de los aspectos generales del tratamiento farmacológico de la obesidad es indispensable para su uso adecuado. La posibilidad de usar nuevos productos resulta una alternativa esperanzadora.
Palabras clave: obesidad, tratamiento farmacológico, pérdida de peso.
]]>
ABSTRACT
Introduction: through the use of pharnacotherapy in the obese patients, it has been intended to reach bigger and faster weight loses than those reported by the conventional therapeutic methods, all of which undoubtedly facilitates the objective reduction of morbidity and mortality observed in patients suffering this health problem.
Objectives: to describe the general aspects of the drug therapy in the obese patient and to mention some of the drugs used.
Development: it is important to know the general aspects of the pharmacotherapy for obesity. Considerations on how to select the optimal drug, the general principles of using it, the criteria for the selection of patients and the types of drugs to be avoided in the obese people are key elements to assure a quality medical care. Some well-known products that were prescribed as pharmaco treatment for obesity are not longer in use because of their ineffectiveness or adverse effects. The use of orlistat or xenical should be the most common alternative to treat this disease at present. The recent approval or new drugs such as Lorcaserina (belviq) and qnexa (qsymia) may represent useful options.
Conclusions: the knowledge about the general aspects of the pharmacotherapy aimed at obesity is indispensable. The possibility of using new drugs turns out to be a hopeful alternative.
Key words: obesity, drug treatment, weight loss.
]]>
INTRODUCCIÓNLa obesidad (OB) es una enfermedad que presenta características muy peculiares, entre las que podemos destacar las siguientes:1-14
En la práctica médica habitual existen dificultades para conseguir resultados significativos y duraderos en el tratamiento del paciente obeso. De ahí que estemos hablando del cumplimiento riguroso de medidas difíciles de seguir por la mayoría de los pacientes.2,13 A través del uso de la Ft en el paciente obeso, se ha intentado obtener pérdidas de peso mayores y más rápidas que las reportadas con los métodos convencionales de tratamiento, lo que, sin dudas, contribuye a una disminución objetiva de la morbilidad y mortalidad observada en los casos con este problema de salud. La efectividad de los métodos de reducción de peso es limitada y existe un elevado índice de fracasos, ya que en la mayoría de los pacientes las recaídas son frecuentes y se acompañan de la recuperación del peso previo.15,16
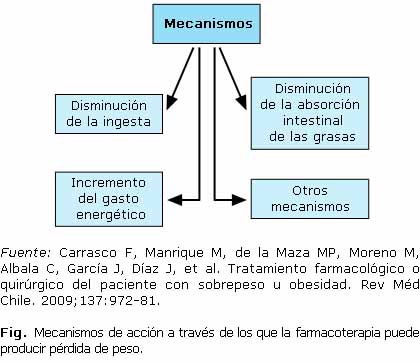
La Ft se basa en el uso de medicamentos capaces de producir una pérdida de peso y grasa corporal a través de diferentes mecanismos, que podemos observar en la figura.17 Su empleo, junto con el tratamiento higiénico dietético, así como de otras medidas, puede constituir una ayuda considerable para lograr este objetivo, y disminuir y/o controlar el impacto de las comorbilidades que casi siempre acompañan a la OB.
¿Cuál es la meta perseguida por la mayoría de las personas con OB?
Tener la posibilidad de disfrutar del placer de comer (incluso en grandes cantidades), y a pesar de ello evitar las consecuencias del sobrepeso, con la ayuda de un preparado cómodo en su administración y que evite cualquier problema secundario18 es la meta perseguida por la mayoría de las personas con OB, meta que, por el momento no es realista.
¿Idealmente, qué características debe tener una droga para bajar de peso?
Una droga para bajar de peso debe reunir las características siguientes:19
]]> • Reducción demostrada de peso y enfermedades asociadas.• Efectos secundarios tolerables o transitorios.
• Sin reacciones adversas mayores después de años de uso.
• Eficacia mantenida a largo plazo.
• Sin propiedades adictivas.
• Mecanismo(s) de acción conocido(s).
• Costo razonable.
Evidentemente, todavía no se cuenta con un medicamento que reúna estas bondades en su totalidad. La droga ideal para bajar de peso por el momento no existe. ]]>
Se describen a continuación los principios básicos para el uso racional de los fármacos antiobesidad:20
- Uso solo como coadyuvante del tratamiento dietético y los cambios en el estilo de vida. Por el momento, no existe ninguna terapia farmacológica que permita liberar el aporte calórico en el tratamiento de la OB.
- Los fármacos actuales no curan la OB, y su efectividad se prolonga únicamente durante su uso.
- La administración debe realizarse siempre bajo supervisión médica continuada.
- La elección del fármaco debe ser individualizada, una vez evaluados los riesgos asociados a su uso.
]]>
¿Cuáles son los criterios de selección para terapia con drogas anti-obesidad en pacientes obesos?
Los criterios de selección aparecen a continuación:21-23
- Pacientes con IMC ³ 30 kg/m2 o un IMC de ³ 27 kg/m2 con comorbilidades, como: diabetes mellitus tipo 2 (DM 2), hipertensión arterial (HTA), dislipidemia (DLP), historia familiar de padres obesos con muerte prematura por cardiopatía isquémica (CI) o DM 2 con complicaciones, entre otras, en los que la dieta y el ejercicio no han logrado la pérdida de peso esperada.
- Pacientes que físicamente tienen serias limitaciones por su peso corporal (obesidad mórbida), y que no estén aptos o no deseen CB.
¿Qué otros aspectos se deben conocer?
Es de interés conocer:24 ]]>
- En general, estos medicamentos pueden ser administrados más allá de 3 meses si se ha perdido al menos 5 % del peso corporal desde el inicio del tratamiento.
- Si se recupera el peso perdido inicialmente, a pesar de haber cumplido correctamente las indicaciones, el tratamiento debe ser interrumpido.
- La pérdida de peso durante el tratamiento es usualmente recobrada con el paso del tiempo después que cesa el tratamiento farmacológico, pero típicamente a un ritmo más lento que cuando se produjo la pérdida de peso.
- Un resultado razonable y médicamente significativo para muchas personas obesas, es la pérdida del 10 % de peso corporal en los primeros 6 meses de tratamiento farmacológico.
- Se insiste en que el uso de medicamentos como única opción es prácticamente inefectivo.
- Ante la presencia de obesidad visceral, si el medicamento disminuye la acumulación de tejido adiposo intrabdominal, debe de ser considerado efectivo y apropiado.
¿Qué fármacos deben ser evitados en un paciente con OB?
El uso de algunos medicamentos puede favorecer el incremento de peso, y deben ser evitados, siempre que sea posible, en un paciente con OB. En el cuadro se pueden observar los más representativos.24-26 ]]>
Existen diferentes productos comercializados para la pérdida de peso que contienen ingredientes no declarados, y que por tanto, su consumo resulta un gran riesgo.27 El consumo de las fórmulas anti-obesidad de moda en muchos países, suelen ser un auténtico cocktail explosivo compuesto por extractos tiroideos, derivados anfetamínicos, diuréticos y benzodiacepinas, entre otras. No se recomienda el uso de parches dérmicos o productos light sin evidencia científica, ya que podrían ocasionar riesgos irreparables a la salud de quienes los utilicen.24
¿Cuáles han sido los productos más conocidos e indicados como tratamiento farmacológico de la OB a lo largo de estos últimos años?
A continuación se expone una relación de los productos más conocidos e indicados como tratamiento farmacológico de la OB a lo largo de estos últimos 100 años, muchos de los cuales pasaron al desuso, por su pobre efectividad o por presentar una serie de efectos secundarios que obligaron a su retiro del mercado, así como otros recientemente aprobados. Se hace mención también, a ciertos medicamentos que se encuentran en estudio y que se proponen para ser usados como monoterapia, y en otros casos, como terapia combinada en el tratamiento de estos pacientes.
Como se puede ver, estos productos pueden actuar utilizando uno, o la combinación de diferentes mecanismos de acción: ]]>
- Productos farmacológicos que facilitan la disminución de la ingesta:• Anfetaminas (anfetamina, metanfetamina y fenmetracina).28
• Derivados de las anfetaminas (fentermina, dietilpropión, anfepromona, fendimetracina, benzfetamina, fenilpropanolamina, fenproporex y clobenzorex).26,28,29
• Actividad similar a las anfetaminas (mazindol).26,28
• Psicoestimulantes o estimulantes psicomotores: metilfenidato.26
• Inhibidores selectivos de la recaptación de serotonina (fenfluramina, dexfenfluramina, fluoxetina, paroxetina, sertralina, fluvoxamina, paroxetina, citalopram y sertralina).26
• Combinaciones de derivados de las anfetaminas con inhibidores selectivos de la recaptación de serotonina: fenfluramine-phentermine (aminorex).30
• Inhibidores selectivos de la recaptación de noradrenalina y serotonina: sibutramina o reductil o meridia.31-33
• Antagonistas del sistema endocanabinoide (bloqueadores selectivos de los receptores canabinoides de tipo 1): rimonabant o acomplia.17, 34-37 En estudio: taranabant, otenabant y SLV-319.
]]> • Hormonas capaces de producir saciedad en casos específicos de carencia de esta y utilizadas en el tratamiento de la OB (leptina).38• Inductores de la saciedad por acción directa sobre el aparato digestivo (fibra vegetal: Fibra Guar®, Fibra Leo® y Metamucil®, metilcelulosa y glucomanan).17
- Productos farmacológicos que facilitan un incremento del gasto energético:
• Desacoplador de la cadena respiratoria (dinitrofenol).39,40
• Anfetaminas.28
• Derivados de las anfetaminas.26,28,29
• Actividad similar a las anfetaminas.26,28
• Psicoestimulantes o estimulantes psicomotores: metilfenidato.26
• Combinaciones de derivados de las anfetaminas con inhibidores selectivos de la recaptación de serotonina: fenfluramine-phentermine (aminorex).30
]]> • Inhibidores selectivos de la recaptación de noradrenalina y serotonina.26• Modificaciones metabólicas específicas que facilitan la pérdida calórica: efedrina, cafeína.41
- Hormonas con propiedades de de tipo lipolíticas/antilipogénicas en el tratamiento de la OB:
• Hormonas tiroideas17 (útil solo en caso de hipotiroidismo y OB).
• Gonadotropina coriónica humana (útil solo en caso de hipogonadismo y OB).
• Hormona de crecimiento (útil solo en caso de déficit de dicha hormona y OB).
- Productos farmacológicos inhibidores de la amilasa pancreática: faseolamina.42,43 ]]>
- Productos farmacológicos que disminuyen la absorción intestinal de las grasas: orlistat o xenical.44-47
- Productos farmacológicos sustitutos de las grasas: olestra.48
- Productos farmacológicos que producen pérdida de peso por deshidratación:17 diuréticos (tiazidas: furosemida, entre otras) y laxantes.
- Productos alternativos no farmacológicos, que supuestamente actúan a través de diferentes mecanismos:49
]]>• Chitosán, picolinato de cromo, ephedra sinica o ma huang (contiene efedrina), garcinia cambogia, glucomanano, hidroximetilbutirato, plantago psyllium, piruvato, yerba mate y yohimbina, spirulina, vinagre de manzana, ácido linoleico y centella asiática, entre otras.
- Fármacos que pueden favorecer la pérdida de peso en personas con prediabetes y/o DM fundamentalmente. Entre ellos se han usado:
- Drogas antiobesidad, su futuro inmediato: los medicamentos que se señalan a continuación han sido estudiados para ser usados en el tratamiento de la OB, en monoterapia o terapia combinada. Los mecanismos de acción de estos fármacos pueden ser, en muchos casos, complementarios, por lo que muchos abogan por los efectos sinérgicos de las combinaciones entre medicamentos que operan a través de sistemas distintos, que generalmente producen principalmente una disminución del apetito y/o aumentan la pérdida calórica. En el caso del cetilistate actúa a través de la disminución de la absorción intestinal de las grasas. Entre ellos se citan:
· Tesofensina79-81
· Liraglutide81,82 (ya citado)***
· Cetilistate81,83,84
· Bupropiona + naltrexona (contrave)75,82,85-88
· Bupropiona + zonisamida (empatic)75,82
· Fentermina + topiramato (qnexa o qsymia)75,82,89-91**
· Pramlintide + metreleptina82,92,93
Lo expuesto permite afirmar que la OB es una enfermedad que requiere de un tratamiento integral e individualizado. Se conoce que puede ser de utilidad el empleo de medidas farmacológicas en un gran número de pacientes para obtener la pérdida de peso esperada, y de esta forma, disminuir sus consecuencias, de ahí el gran valor de conocer los aspectos generales de este tipo de tratamiento. Además, se puede decir lo limitado que resultan los tratamientos farmacológicos para la obesidad, el uso de orlistat o xenical debe de ser la alternativa más común en el tratamiento actual con fármacos para la obesidad.
La prescripción racional de los análogos de acción prolongada de la GLP-1 (exenatide o byetta y el liraglutide o victosa),57-63 de los inhibidores de la DPP-4 (sitagliptina y la vildagliptina),65-67 o del análogo sintético de la hormona amilina (pramlintide), para uso fundamentalmente en pacientes con DM 2 con sobrepeso corporal u OB, ha significado un paso de avance extraordinario. La reciente aprobación del uso de la lorcaserina (belviq) y de qnexa (o qsymia), para el tratamiento farmacológico de la OB, es, sin dudas, una opción esperanzadora.
_________________________
* Medicamentos aprobados en el tratamiento farmacológico del paciente con DM 2 y OB.
** Fármacos recientemente aprobados para uso en el tratamiento farmacológico de la OB.
*** Fármacos recientemente aprobados para uso en el tratamiento farmacológico de la OB y en el paciente obeso con DM 2.
]]>
REFERENCIAS BIBLIOGRÁFICAS
1. Pi-Sunyer FX. The obesity epidemic: pathophysiology and consequences of obesity. Obes Res. 2002;10:97S-104S.
2. Martínez Olmos, MÁ, Casanueva Freijo F. Avances en el tratamiento de la obesidad. Aplicaciones a la práctica clínica. Medicine. 2009;10(46):3083-90.
3. Plaza J, Ciurana JM, Vergara LL, Mateos F, Romero MI. Prevalencia de Obesidad en escolares. Rev Clin Med Fam. 2008;2(3):106-10. ]]>
4. Martos GÁ, Barrios V, Argente J. Fundamentos clínicos y diagnóstico-terapéuticos de la obesidad en la infancia. Rev Esp Ped. 2009;65:408-22.
5. Burgos P, Rescalvo F, Ruiz T, Velez M. Estudio de obesidad en el medio sanitario. Med segur trab. 2008;54(213):75-80.
6. Cardone A, Borracci RA, Milin E. Estimación a largo plazo de la prevalencia de obesidad en la Argentina. Rev Argent Cardiol. 2010;78(1):23-9.
7. Hetherington MM, Cecil JE. Gene-environment interactions in obesity. Forum Nutr. 2010;63:195-203.
8. Baudrand R, Arteaga U, Moreno M. Adipose tissue as an endocrine modulator: hormonal changes associated with obesity. Rev méd Chile. 2010;138(10):1294-301. ]]>
9. Ruano M, Silvestre V, Aguirregoicoa E, Criado L, Duque Y, García G. Nutrición, síndrome metabólico y obesidad mórbida. Nutr. Hosp. 2011;26(4):759-64.
10. Weaver JU. Classical endocrine diseases causing obesity. Front Horm Res. 2008;36:212-28.
11. Frelut ML. Obesidad en el adolescente. EMC-Tratado de Medicina. 2011;15(2):1-9.
12. Martos GA, Argente J. Obesidades pediátricas: de la lactancia a la adolescencia. An Pediatr (Barc). 2011;75(1):63.e1-63.e23.
13. Salas J, Rubio MA, Barbany M, Moreno B y Grupo Colaborativo de la SEEDO. Consenso SEEDO 2007 para la evaluación del sobrepeso y la obesidad y el establecimiento de criterios de intervención terapéutica. Med Clin (Barc). 2007;128:184-96. ]]>
14. OMS. Obesidad y sobrepeso [homepage en Internet] Ginebra, Suiza. Nota descriptiva N° 311. Marzo de 2011; 2011 [citado 10 de abril de 2012]. Disponible en: http://www.who.int/mediacentre/factsheets/fs311/es/o
15. Romero H, Martinez MA, J.L. Pereira JL, García PP. Tratamiento farmacológico de la obesidad. Rev Esp Obes. 2005;3(1):13-25.
16. Curioni CC, Lourenço PM. Long-term weight loss after diet and exercise-a systematic review. International Journal of Obesity and related metabolic disorders. 2005;29:1168-174.
17. Carrasco F, Manrique M, de la Maza MP, Moreno M, Albala C, García J, Díaz J, et al. Tratamiento farmacológico o quirúrgico del paciente con sobrepeso u obesidad. Rev Méd Chile. 2009;137:972-81.
18. Martul P, Rica I, Vela A, Grau G. Tratamiento de la obesidad infanto-juvenil. An Esp Pediatr. 2002;56[Supl 4]:17-27.
19. WHO. Obesity. Preventing and management the global epidemic. Report of a WHO Consultation on Obesity. Geneva: WHO; 1997.
20. Halpern A, Mancini MC. Treatment of obesity: an update on anti-obesity Medications. Obesity reviews. 2003;4:25-42.
21. Zanella MT, Ribeiro FF. Emerging drugs for obesity therapy. Arq Bras Endocrinol Metab. 2009;53(2):271-80.
22. A report of the Nutrition Committee of the Royal College of Physicians of Lond. Anti-obesity drugs: Guidance on appropriate prescribing and management [homepage en Internet]; 2003 [citado 23 de julio de 2009]. Disponible en: http://www.rcplondon.ac.uk/pubs/contents/adbc956d-da14-436d-bdd1-c87cd15b2440.pdf
23. Expert Panel on the Identification, Evaluation and Treatment of Overweight and Obesity in Adults. Executive summary of the clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. Archives of Internal Medicine. 1998;158:1855-67.
24. Bastarrachea RA ¿Cuál es el papel actual del tratamiento farmacológico de la obesidad en personas adultas? Rev Endocrinol Nutr. 2004;12(Supl. 3):5130-5. ]]>
25. Cuevas A, Reyes MS. Lo último en diagnóstico y tratamiento de la obesidad ¿Hay lugar aún para la terapia conservadora? Rev méd Chile. 2005;133(6):713-22.
26. Quiñones G. Efecto de los medicamentos en el apetito. Trastornos de la Conducta Alimentaria. 2011;13:1437-60.
27. FDA News release. FDA Expands Warning to Consumers About Tainted Weight Loss Pills [homepage en Internet]. 2009 [citado 23 de julio de 2009]. Disponible en: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2008/ucm116998.htm
28. Glazer G. Long-term pharmacotherapy of obesity 2000: a review of efficacy and safety. Arch Intern Med. 2001;161:1814-24.
29. Kernan WN, Viscoli CM, Brass LM, Broderick JP, Brott T, Feldmann E, et al. Phenylpropanolamine and the risk of hemorrhagic stroke. N Engl J Med. 2000;343:1826-32.
30. Gurtner HP. Aminorex pulmonary hypertension. Pennsylvania: University of Pennsylvania Press; 1990. p. 397-411.
31. Pinto ME, Manrique HA. Retiro de sibutramina por riesgo de enfermedad cardiovascular. Rev perú med exp salúd pública. 2010;27(3):489-90.
32. Lavalle A M, Costabel JP, Alves de Lima A, Trivi M, Giniger A. Taquicardia ventricular asociada con la utilización de sibutramina. Rev argent cardiol. 2011;79(6):541-3.
33. Gómez-Barrado JJ, Turégano S, Garcipérez de Vargas FJ, Porras Y. Acute Coronary Syndrome in a Young Woman Treated With Sibutramine. Rev Esp Cardiol. 2010;63(2):240-52.
34. Després JP, Golay A, Sjöström L. Rimonabant in Obesity-Lipids Study Group. Effects on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med. 2005;353:2121-34.
35. Pi-Sunyer FX, Aronne LJ, Heshmati HM, Devin J, Rosenstock J and RIO North America Study Group. Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients. RIO-North America: a randomized controlled trial. JAMA. 2006;295:761-75.
36. Van Gaal LF, Pi-Sunyer X, Despre's JP, Mc Carthy C, Scheen AJ. Efficacy and safety of rimonabant for improvement of multiple cardiometabolic risk factors in overweight/obese patients: pooled 1-year data from the RIO program. Diabetes Care. 2008;31 (Suppl 2):S229-40.
37. Jones D. End of the line for cannabinoid receptor 1 as an anti-obesity target? Nat Rev Drug Discov. 2008;7:961-2.
38. Licinio J, Caglayan S, Ozata M, Yildiz B, de Miranda P, O'Kirwan F, et al. Phenotypic effects of leptin replacement on morbid obesity, diabetes mellitus, hypogonadism, and behavior in leptin-deficient adults. Proc Natl Acad Sci USA. 2004;101:4531-6.
39. Tainter ML, Stockton AB, Cutting WC. Use of dinitrophenol in obesity and related conditions. JAMA. 1933;101:1472-5.
40. Council on Pharmacy and hemistry. Dinitrophenol not acceptable for N.N.R. JAMA. 1935;105:31-3.
41. Dulloo AG. Herbal simulation of ephedrine and caffeine in treatment of obesity. Int J Obes. 2002;26:590-2.
42. Díaz E, Aguirre C, Gotteland M. Efecto de un inhibidor de a-amilasa sobre la reducción de peso de mujeres obesas. Rev Chil Nutr. 2004;31:306-17.
43. Tucci SA, Boyland EJ, Halford JCG. The role of lipid and carbohydrate digestive enzyme inhibitors in the management of obesity: a review of current and emerging therapeutic agents. Diabetes Metab Syndr Obes. 2010;3:125-43.
44. Idelevich E, Kirch W, Schindler C. Current pharmacotherapeutic concepts for the treatment of obesity in adults. Ther Adv Cardiovasc Dis. February 2009;3(1):75-90.
45. Velázquez M, Sanín LH, Ávila JA, Díaz CV, López SJ. Orlistat en sujetos mexicanos. Ensayo clínico comparativo de efectividad terapéutica. Revista Salud Pública y Nutrición [serie en Internet]. Julio-Septiembre 2011 [citado 31 de diciembre de 2012];12(3). Disponible en: http://www.medigraphic.com/pdfs/revsalpubnut/spn-2011/spn113e.pdf
46. Yeste D, Carrascosa A. El manejo de la obesidad en la infancia y adolescencia: de la dieta a la cirugía. Endocrinol Nutr [serie en Internet]. 2012 [citado 31 de diciembre de 2012];380. Disponible en: http://www.elsevier.es/sites/default/files/elsevier/eop/S1575-0922%2812%2900168-4.pdf
47. Cruz CM, Céspedes MD, Delgadillo JP, Torres G. Pancreatitis Aguda como consecuencia del uso de Orlistat. Rev Cient Cienc Méd. dic. 2011;14(2):35-8.
48. Caixas A. Tratamiento farmacológico de la obesidad. Endocrinol Nutr. 2000;47(1):16-20.
49. Mhurchu CN, Poppitt SD, Mcgill AT, Leahy FE, Bennett DA, Lin RB, et al. The effect of dietary supplement, Chitosan, on body weight: a randomized controlled trial in 250 overweight and obese adults. Int J Obes. 2004;28:1149-56.
50. Ong CR, Molyneaux LM, Constantino MI, Twigg SM, Yue DK. Long-term efficacy of metformin therapy in nonobese individuals with type 2 diabetes. Diabetes Care. 2006;29:2361-4.
51. Park MH, Kinra S, Ward KJ, White B, Viner RM. Metformin for obesity in children and adolescents: a systematic review. Diabetes Care. 2009;32:1743-5.
52. Quinn SM, Baur LA, Garnett SP, Cowell CT. Treatment of clinical insulin resistance in children: a systematic review. Obes Rev. 2010;11:722-30.
53. Crespo S. Metformina, a 50 años de su uso: eficacia, tolerancia, seguridad y nuevas indicaciones. Rev Cient Cienc Med. 2009;12(2):23-5.
54. Hernández A, Torres O, Carrasco B, Nasiff A, Castelo L, Pérez L, et al. Tratamiento farmacológico de la prediabetes. Rev Cubana Endocrinol. 2011;22(1):36-45.
55. Díaz E, Aguirre C, Gotteland M. Efecto de un inhibidor de a-amilasa sobre la reducción de peso de mujeres obesas. Rev Chil Nutr. 2004;31:306-17.
56. Tucci SA, Boyland EJ, Halford JCG. The role of lipid and carbohydrate digestive enzyme inhibitors in the management of obesity: a review of current and emerging therapeutic agents. Diabetes Metab Syndr Obes. 2010;3:125-43.
57. Folli F, Guardado Mendoza R. Potential use of exenatide for the treatment of obesity. Expert Opinion on Investigational Drugs. 2011;20:1717-22.
58. Goodall G, Costi M, Timlin L, Reviriego J, Sacristán JA, Smith-Palmer J, et al. Coste-efectividad de exenatida en comparación con insulina glargina en pacientes con obesidad y diabetes mellitus tipo 2 en España. Endocrinol Nutr. 2011;58(7):331-40.
59. Verge D, López X. Impact of GLP-1 and GLP-1 receptor agonists on cardiovascular risk factors in type 2 diabetes. Curr Diabetes Rev. 2010;6:191-200.
60. Garber AJ. Long-acting glucagon-like peptide 1 receptor agonists: a review of their efficacy and tolerability. Diabetes Care. 2011;34(Suppl 2):S279-S284.
61. Buse JB, Rosenstock J, Sesti G, Schmidt WE, Montanya E, Brett JH, et al. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: a 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet. 2009;374:39-47.
62. Plutzky J. The Incretin Axis in Cardiovascular Disease. Circulation. 2011;124:2285-9.
63. Blonde L, Montanya E. Comparison of liraglutide versus other incretinrelated anti-hyperglycaemic agents. Diabetes Obes Metab. 2012;14(Suppl 2):20-32.
64. Quevedo J, Tur J, Serra G, Burguera B. Utilidad de los fármacos antiobesidad en la diabetes mellitus tipo 2. Av Diabetol. 2010;26:161-6.
65. Di Girolamo G, Tamez AL, Tamez HE. Inhibidores de la dipeptidil peptidasa-4: farmacodinamia, farmacocinética y seguridad. Med Int Mex. 2008;24(2):142-7.
66. González M, Pereg V, Burgera YB. Novedades en terapia hipoglucemiante. Fármacos con acción incretina. Endocrinol Nutr. 2008;55(Supl 2):73-7.
67. Davidson JA, Parente EB, Gross JL. Incretin mimetics and dipeptidyl peptidase-4 inhibitors: innovative treatment therapies for type 2 diabetes. Arq Bras Endocrinol Metabol. 2008;52:1039-49.
68. Lee NJ, Norris SL, Thakurta S. Eficacy and Harms of the Hypoglycemic Agent Pramlintide in Diabetes Mellitus. Ann Fam Med. 2010;8:542-9.
69. Meneghini LF, Orozco-Beltran D, Khunti K, Caputo S, Damçi T, Liebl A, et al. Weight Beneficial Treatments for Type 2 Diabetes. J Clin Endocrinol Metab. 2011;96(11):3337-53.
70. Aronne L, Fujioka K, Aroda V, Chen K, Halseth A, Kesty NC, et al. Progressive reduction in body weight after treatment with the amylin analog pramlintide in obese subjects: a phase 2, randomized, placebo-controlled, dose-escalation study. J Clin Endocrinol Metab. 2007;92:2977-83.
71. Riddle M, Pencek R, Charenkavanich S, Lutz K, Wilhelm K, Porter L. Randomized comparison of pramlintide or mealtime insulin added to basal insulin treatment for patients with type 2 diabetes. Diabetes Care. 2009;32:1577-82.
72. Artola S. Panorama de los tratamientos actuales disponibles para los pacientes con diabetes tipo 2. Algoritmo terapéutico de la ADA/EASD. Seguridad y tolerabilidad. Aten Primaria. 2010;42(1):24-32.
73. Smith SR, Prosser WA, Donahue DJ, Morgan ME, Anderson CM, Shanahan WR, et al. Lorcaserin (APD356), a selective 5HT (2C) agonist reduces body weight in obese men and women. Obesity (Silver Spring). 2009;17:494-503.
74. Bays HE. Lorcaserin and adiposopathy: 5-HT2c agonism as a treatment for sick 'fat' and metabolic disease. Expert Rev Cardiovasc Ther. 2009;7:1429-45.
75. Jones D. Novel pharmacotherapies for obesity poised to enter marke. News & analysis. 2009;8:833-4.
76. Muñoz-Rivas N. Ensayo randomizado, multicéntrico de lorcaserina frente a placebo en el manejo del sobrepeso y obesidad. Rev Clin Esp. 2011;211(1):52-3.
77. Smith SR, Weissman NJ, Anderson ChM, Sanchez M, Chuang E, Stubbe S, et al. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med. 2010;363:245-56.
78. Astrup A. Drug Management of Obesity-Efficacy versus Safety. N Engl J Med. 2010;363(3):288-90.
79. Astrup A, Meier DH, Mikkelsen BO, Villumsen JS, Larsen TM. Weight loss Produced by tesofensine in patients with Parkinson's or Alzheimer's disease. Obesity (Silver Spring). 2008;16:1363-9.
80. Astrup A, Madsbad S, Breum L, Jensen TJ, Kroustrup JP, Larsen TM. Effect of tesofensine on bodyweight loss, body composition and quality of life in obese patients: a randomized, double-blind, placebo-controlled trial. The Lancet. 2008;372(9653):1906-13.
81. Faria AM, Mancini MC, de Melo ME, Cercato C, Halpern A. Progressos recentes e novas perspectivas em farmacoterapia da obesidade. Arq Bras Endocrinol Metab. 2010;54:516-29.
82. Jones D. Suspense builds on anti-obesity rollercoaster ride. News & analysis. 2011;10:5-6.
83. Padwal R. Cetilistat, a new lipase inhibitor for the treatment of obesity. Curr Opin Invest Drugs. 2008;9:414-21.
84. Bryson A, De la Motte S, Dunk C. Reduction of dietary fat absorption by the novel Gastrointestinal lipase inhibitor cetilistat in healthy volunteers. Br J Clin Pharmacol. 2009;67:309-15.
85. Lee MW, Fujioka K. Naltrexone for the treatment of obesity: re-view and update. Expert Opin Pharmacother. 2009;10:1841-5.
86. Simpson KA, Martin NM, Bloom SR. Hypothalamic regulation of food intake and clinical therapeutic applications. Arq Bras Endocrinol Metabol. 2009;53:120-8.
87. Greenway FL, Whitehouse MJ, Guttadauria M, Anderson JW, Atkinson RL, Fujioka K, et al. Rational design of a combination medication for the treatment of obesity. Obesity (Silver Spring). 2009;17:30-9.
88. Padwal R. Contrave, a bupropion and naltrexone combination therapy for the potential treatment of obesity. Curr Opin Invest Drugs. 2009;10:1117-125.
89. Gadde KM, Allison DB, Ryan DH, Peterson CA, Troupin B, Schwiers ML, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. The Lancet. 2011;377(9774):1341-52.
90. Rubino DM, Gadde KM. A review of topiramate and phentermine: a combined therapeutic approach for obesity. Clinical Lipidology. 2012;7(1):13-25.
91. National Institutes of Health (NHI). FDA Approves 2nd New Weight-Loss Drug. U.S. National Library of Medicine [homepage en la Internet]. 2012 [citado 10 de septiembre de 2012]. Disponible en: http://www.nlm.nih.gov/medlineplus/news/fullstory_127324.html
92. Trevaskis JL, Lei C, Koda JE, Weyer C, Parkes DG, Roth JD. Interaction of leptin and amylin in the long-term maintenance of weight loss in diet-induced obese rats. Obesity (Silver Spring). 2010;18:21-6.
93. Ravussin E, Smith SR, Mitchell JA, Shringarpure R, Shan K, Maier H, et al. Enhanced weight loss with pramlintide/metreleptin: an integrated neurohormonal approach to obesity pharmacotherapy. Obesity (Silver Spring). 2009;17:1736-43.
Recibido: 30 de noviembre de 2012.
Aprobado: 18 de enero 2013. ]]>
José Hernández Rodríguez. Centro de Atención al Diabético del INEN. Ave 17, esquina a D, Vedado, municipio Plaza de la Revolución. La Habana, Cuba. Correo electrónico: pepehdez@infomed.sld.cu ]]>