ORIGINAL ARTICLE
Drug interactions in children with respiratory diseases in the pediatric unit of a teaching hospital in Brazil
Interacciones medicamentosas observadas en niños con enfermedades respiratorias en la unidad pediátrica de un hospital docente de Brasil
Márcia Yuriko Masukawa,I Glenda Batalho Veríssimo,I Maria Helena Vianello Richtzenhain,II Alessandra LinardiII
IBSN. Department of Physiological Sciences. Santa Casa de São Paulo. School of Nursing. SP, Brazil.
IIPhD, Associate Professor. Department of Physiological Sciences. Santa Casa de São Paulo School of Medical Sciences. SP, Brazil.
ABSTRACT
Introduction: the pharmacokinetic and pharmacodynamic responses to drugs can change during childhood, with children being more susceptible to adverse drug reactions than adults. The frequency of adverse reactions and length of hospitalization generally increase as the number of drugs administered increases.
Objective: evaluating the drug associations used in children hospitalized with respiratory disorders in the pediatric unit of a teaching hospital in Brazil.
Methods: this retrospective study was based on the quantitative analysis of data collected from medical records of hospitalized children aged 7-12 years with diagnoses for respiratory diseases and cystic fibrosis with pulmonary exacerbation. The descriptive analyses of data were done using SPSS® v.13.0 software. The Micromedex database was used to identify drug interactions and to determine the classification based on severity.
Results: there were 111 possible drug interactions for respiratory diseases (25 major, 75 moderate, 10 minor and one contraindicated drug association) in 49 medical records. For cystic fibrosis, there were five possible drug interactions (four major and one minor) in 18 medical records.
Conclusions: these findings indicate that pediatric prescription should be confined to essential drugs. The prescription should contain the smallest possible number of medications to prevent possible drug interactions. The availability of a multidisciplinary team combined with a program of active pharmacovigilance can help to prevent the occurrence of drug. interactions.
Key words: cystic fibrosis; drug interaction; pediatric; respiratory tract diseases.
RESUMEN
]]> Introducción: las respuestas farmacocinéticas y farmacodinámicas al consumo de medicamentos pueden cambiar en la niñez al encontrarse niños que son más propensos a desarrollar reacciones adversas a los medicamentos que los adultos. Por lo general, la frecuencia de las reacciones adversas y el tiempo de hospitalización aumentan a medida que se incrementa el número de fármacos suministrados.Palabras clave: fibrosis quística; interacción medicamentosa; pediátrico; enfermedades del tracto respiratorio.
INTRODUCTION
In clinical practice, the rational prescribing of drugs should consider the dose capable of generating a therapeutic effect while causing minimal adverse reactions.1 In certain conditions, a combination of two or more drugs can enhance the overall therapeutic effect. However, as the number of drugs administered increases, the occurrence of drug interactions becomes significant and can lead to adverse reactions, toxicity and prolonged hospitalization.2-4 An adverse drug reaction is defined by the World Health Organization (WHO) as any noxious, unintended or undesired effect of a drug that occurs at doses used in humans for prophylaxis, diagnosis or therapy. Multiple factors can contribute to adverse drug reactions, including multiple drug therapy, disease severity and age.2-5
]]> In particular, the pharmacokinetic and pharmacodynamic characteristics of a drug may change during childhood development, with children being more susceptible to adverse drug reactions than adults.6,7 Accordingly, effective and safe drug therapy in children requires an understanding of organ maturation and of drug absorption, distribution, metabolism and excretion, as well as drug action.8,9 For example, some drug-metabolizing enzymes do not become active until a certain age is reached and this may lead to the accumulation of a drug in the body; in contrast, other enzymes may show a higher metabolic capacity compared to adults, resulting in the increased production of toxic metabolites.6,7In addition, mature renal function is achieved at one year of age, whereas the participation of transporter proteins in the active renal excretion and reabsorption of many drugs during childhood development is unclear.10,11 The reduction in total plasma proteins (including albumin) in neonates and young infants increases the free fraction of some drugs, thereby influencing their bioavailability and distribution to tissues.6,12,13 Most drugs are administered orally to children and gastric pH increases in neonates and infants to reach adult values at ~2 years of age. In addition, gastrointestinal motility decreases in neonates and reaches adult levels in older infants and children.6,13 With regard to pharmacodynamics, there is little information on the interaction between drugs and receptors and the consequence(s) of these interactions during childhood development.6 Unfortunately, many drugs are given to children in an “off label” manner that requires more significant therapeutic monitoring.14 The number of prescribed drugs and posology constitute risk factors for adverse reactions and toxicity; monitoring these factors requires a constant review of clinical protocols and a large therapeutic arsenal.4 In this study, we evaluated the drug associations used in children hospitalized with respiratory disorders in the pediatric unit of a teaching hospital in Brazil.
METHODS
The study consisted of a retrospective and descriptive quantitative analysis of the prescription patterns for pediatric patients with respiratory problems hospitalized in the Irmandade da Santa Casa de Misericórdia de São Paulo (ISCMSP) in São Paulo city from January to December, 2011. This study was approved by the Ethics Committee of the ISCMSP (protocol no. 346/11).
The subject sample was limited to patients with diagnoses defined by ICD-10, Chapter X, J00 to J99, and Chapter IV, E84.0, corresponding to respiratory diseases and cystic fibrosis with respiratory disorders, respectively.15 Patients aged 7-12 years were included in the study.
A form containing two parts was used to compile data from the medical records:
- Part 1: contained information on patient identification such as registration number, date of admission, name (initials), birth date, age, sex, weight, height, skin color (as defined by the Brazilian Institute of Geography and Statistics - IBGE), hospital discharge and duration of hospitalization.
- Part 2: contained information relating to the patient´s admission: diagnosis (ICD-10 criterion/definition), prescription date, drug, dose, route and interval of administration.
]]> The Thomson Micromedex database was used to identify drug interactions that were then classified according to severity. When there was a health hazard that required immediate medical intervention the drug interaction was classified as major. The interaction was considered moderate when it exacerbated the patient´s disease or adverse effects and required drug substitution. A minor interaction caused clinical effects with an adverse reaction but did not require drug replacement. When the concomitant use of drugs was not recommended the interactions were classified as contraindicated. Since dipyrone and fenoterol hydrobromide are not commercialized in the USA, we reviewed the literature data indicative of possible drug interactions involving these compounds. The data were compiled using the program Microsoft Office Excel®. The descriptive analyses and assessment of the correlation between variables were done using Statistical Package for the Social Sciences (SPSS®) v.13.0 software.
RESULTS
RESPIRATORY DISEASES
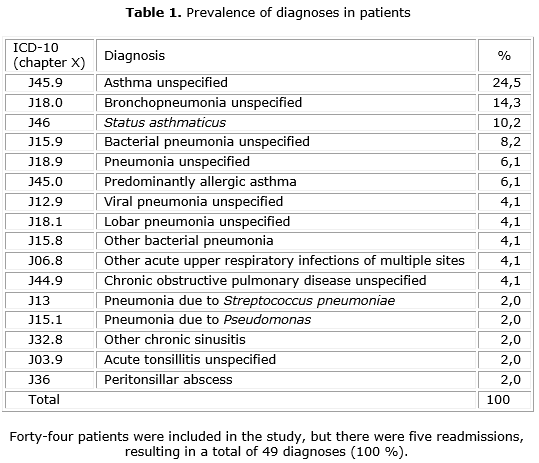
One hundred and twenty-six medical records were evaluated, of which 82 were excluded because they involved patients outside the age limit (exclusion criterion). Of the remaining 44 records, 24 (54.5 %) were males and 20 (45.5 %) were females. There were five re-admissions totaling 49 medical records for the study. The length of hospitalization ranged from 1 to 45 days. There were 16 different diagnoses (table 1), the most prevalent being unspecified asthma detected in 12 medical records (24 %), unspecified bronchopneumonia present in seven medical records (14 %) and status asthmaticus in five medical records (10 %). The total number of prescribed drugs was 90 and there were 1 to 21 prescribed drugs per medical record (mean ± SD: 8.0 ± 5.4); one patient received no medication during hospitalization. There were 391 (100 %) administrations of medication, with the intravenous route being the most frequently used (37.3 %), followed by oral administration (29.7 %), inhalation (20.2 %), gastrostomy (7.4 %) and the nasoenteral route (5.4 %).
Table 2 shows the prescribed drugs recorded in the medical records. Corticosteroids, nonsteroidal anti-inflammatory drugs, betalactam antibiotics, beta-adrenergic bronchodilators and anti-cholinergic bronchodilators were the most prescribed drugs.
There was a significant prevalence of major and moderate possible interactions between drugs administered concomitantly by the intravenous route (32 % and 18.7 %, respectively) and between drugs administered concomitantly by the oral and intravenous routes (moderate= 16 % and major= 14.7 %). Among the major interactions, the most prevalent involved chloral hydrate, phenobarbital or fentanyl with midazolam (28 %) that can induce respiratory depression. The most prevalent moderate interactions involved clarithromycin and corticosteroids (10.7 %; induced toxic effects of corticosteroids), phenobarbital and corticosteroids (6.7 %; reduction in therapeutic efficacy), acetyl salicylic acid and corticosteroids (5.3 %; increased risk of ulcers), midazolam and ranitidine (5.3 %; increase in midazolam bioavailability), atracurium besylate and corticosteroids (5.3 %; reduction in atracurium besylate efficacy, muscle weakness and myopathy), diazepam and phenytoin (4 %; increased toxic effects of phenytoin), carbamazepine and phenytoin (4 %; alteration in the plasma concentrations of both drugs), hydrochlorothiazide and corticosteroids (4 %; hypokalemia and subsequent cardiac arrhythmias), phenytoin and corticosteroids (4 %; reduction in corticosteroid efficacy), ciprofloxacin and corticosteroids (4 %; increased risk of tendon rupture) and carbamazepine and corticosteroids (4 %; reduction in corticosteroid efficacy). For minor interactions, those involving phenytoin and ranitidine (40 %; increase in phenytoin concentrations) and carbamazepine and clobazam (20 %; decrease in carbamazepine and metabolite concentrations) were the most significant. Only one contraindicated association (fluconazole and fluticasone plus salmeterol; increased risk of QT prolongation and arrhythmias) was detected.
CYSTIC FIBROSIS
Eleven patients (3 males and 8 females) diagnosed with cystic fibrosis and pulmonary exacerbations were included in this study and there were seven re-admissions, totaling 18 medical records. The length of hospitalization ranged from 14 to 23 days and the total number of prescription drugs was 27. The number of prescribed drugs ranged from 6 to 12 per medical record (7.9 ± 4.3). There were 159 (100 %) administrations of medications and the oral route was used most (40 %), followed by inhalation (34 %) and intravenous administration (21 %). The most prevalent bacteria described in the medical records were Pseudomonas aeruginosa (20 %), followed by Staphylococcus aureus (10 %), Streptococcus viridans (10 %) and Bulkholderia cepacia, Klebsiella pneumoniae and Neisseria spp. (8.8 % each).
Figure 2 shows that betalactam antibiotics were the most prescribed medications (13 %), followed by mucolytic dornase alfa (10.1 %), pancreatic enzymes (8.8 %), nutritional support (8.8 %) and the antibiotic amikacin (8.8 %). The Micromedex data base identified five medical records with possible drug interactions. Four medical records had four major interactions (vancomycin plus amikacin) and only one had a minor drug interaction (amikacin plus piperacillin and tazobactam).
]]>DISCUSSION
RESPIRATORY DISEASES
Intravenous injection was the major route of administration (37.3 %) used in respiratory diseases. In addition, the most prevalent major and moderate interactions were observed with drugs administered concomitantly by the intravenous route. The intravenous administration of drugs in children has great clinical relevance. The importance of this route in daily nursing practice includes different methods and periods of administration, a range of agents used for dilution, and drug incompatibility resulting from the combination of drugs in solution.16 In addition, the drug is 100 % bioavailable when administered intravenously and may therefore show greater distribution throughout the organism; this can lead to adverse drug reactions and interactions.
There was significant prescription of dipyrone (metamizole) for intravenous and oral administration, and this apparently reflected the importance of preventing and treatment symptoms such as pain and fever. However, since there is no description of dipyrone in Micromedex, the general literature was used to determine possible drug interactions. Dipyrone has an antipyretic and analgesic action mediated by the inhibition of cyclooxygenase (responsible for the synthesis of arachidonic acid metabolites, principally prostaglandins) and activation of the opioid and cannabinoid systems (prostaglandin-independent pathway).17 In the medical records evaluated there were dipyrone associations with hydrochlorothiazide, furosemide, captopril, amlodipine and captopril. Dipyrone can reduce the diuretic effects of furosemide and hydrochlorothiazide and decrease the efficacy of antihypertensives such as captopril (angiotensin-converting enzyme inhibitor) and amlodipine (calcium channel blocker). The mechanism of these interactions seems to involve a reduction in prostaglandins levels.18,19 In addition, dipyrone can induce cytochrome P450 enzymes (CYP3A4 and CYP2B6) and UDP-glucoronosyltransferase (a conjugation enzyme) that have a very significant role in metabolizing drugs. Thus, dipyrone may trigger various drug interactions by modulating the activity of cytochrome P450 or the conjugation pathway.20-22
Corticosteroids were the most prescribed drugs and therefore more prone to cause drug interactions. Based on the Micromedex database, the drugs that presented the highest number of possible drug interactions were phenytoin (23), clarithromycin (18), prednisone (16), midazolam (15), carbamazepine (13), methylprednisolone (11) and ranitidine (10). Three medical records reported the prescription of 21 drugs; two of these cases had six possible drug interactions while the third had 18 possible drug interactions. The latter medical record included a prescription for prednisone and midazolam, two drugs with higher frequencies of interactions because both are metabolized by cytochrome P450 enzymes.23-25 In addition, this same record had a prescription for deferasirox, a CYP3A4/5 inducer and moderate inhibitor of CYP2C8,26 and ranitidine, a weak inhibitor of CYP.27 The combination of these drugs explains the greater probability of drug interactions in this case when compared to the other two medical records with the same number of prescribed drugs. We also observed that in one medical record with 15 drugs prescribed there were only three possible drugs interactions while in another record that also had 15 drugs there were 16 possible drug interactions. In the latter medical record there was prescription of phenytoin (a CYP2C/3A inducer), clarithromycin (a CYP3A4 inhibitor), prednisone, midazolam, carbamazepine (a CYP2C9/3A inducer) and methylprednisolone.28,29 Furthermore, the five medical records mentioned above also included a prescription for dipyrone, a CYP inducer (see Discussion in previous paragraph). Therefore, drugs with a low therapeutic index (e.g., phenytoin, carbamazepine, corticosteroids) and that require careful dose control are the most associated with adverse effects related to drug interactions. Most of these drugs are used in prolonged treatment and many also are biotransformed by hepatic enzymes that can induce additional drug interactions in child.1,30
These findings indicate that pediatric prescription should be confined to essential drugs. The prescription should contain the smallest possible number of medications to prevent possible drug interactions. In addition, detailed knowledge of the drugs prescribed to hospitalized children with different illnesses, sex and age will be useful in understanding drug interactions and in avoiding adverse reactions. In this regard, nurses have a key role in ensuring quality care, in participating in drug therapy and in identifying adverse events and drug interactions.
CYSTIC FIBROSIS
There was no correlation between hospitalization time and the number of drug interactions in the data analyzed (SPSS® v.13.0 software). In addition, there was no correlation between the number of drugs prescribed per patient and the occurrence of drug interactions, despite the elevated number of drugs per admission (range: 6 to 12). These findings probably reflect the fact that the hospital where this study was done is a referral center for the treatment of cystic fibrosis. A search of Micromedex identified the association of amikacin (an aminoglycoside) and vancomycin (a glycopeptide) as a major interaction that may increase the risk of ototoxicity and nephrotoxicity. The association of vancomycin and amikacin results in combined toxicity that may injure the target organ, e.g., kidneys.34-36 In the present study, amikacin was administered once a day while vancomycin and piperacillin plus tazobactam were administered once every 6 h, with the duration of therapy ranging from 14 to 21 days. The administration of aminoglycosides in a single daily dose reduces the nephrotoxicity, ototoxicity and vestibular toxicity caused by these antibiotics.37,38 One of the medical records revealed a minor interaction due to the association of amikacin and piperacillin/tazobactam (betalactam) that may have attenuated the efficacy of the aminoglycoside if both antibiotics were administered intravenously in the same solution or via the same venous access.
Aminoglycosides are used because of their effectiveness against gram-negative bacilli and their action extends to gram-positive bacteria when associated with betalactam or glycopeptide antibiotics.38,39 The acute and chronic pulmonary infections induced by the presence of mucus and an inflammatory response can determine the morbidity and mortality of cystic fibrosis patients. The treatment of cystic fibrosis with acute pulmonary exacerbations often involves the intravenous administration of aminoglycosides in combination with other classes of antibiotics.39,40 These antibiotic associations are very important for the eradication of resistant P. aeruginosa. Polymicrobial infections are common in cystic fibrosis and the most prevalent bacteria reported in the medical records of our patients were P. aeruginosa, S. aureus and S. viridans.Pseudomonas aeruginosa is the most prevalent bacteria in cystic fibrosis and usually appears in children of school age or in adolescents.38,39,41 This species is the principal cause of the respiratory exacerbations associated with respiratory infections in cystic fibrosis. Correct identification of pathogens is a major challenge and choice of the correct treatment is fundamental for preventing microbial resistance to these antibiotics.42,43 Since P. aeruginosa is the most prevalent pathogen in cystic fibrosis, treatment with oral fluoroquinolones is a therapeutic option for mild exacerbations, whereas antipseudomonal betalactams associated with an aminoglycoside, administered intravenously, are used for severe exacerbations.41 The use of combination antibiotic therapy, the dosing frequency, duration of therapy and parenteral administration for the treatment of respiratory tract infections in cystic fibrosis may induce significant drug interactions. However, in reference centers, the efficacy of treatment with aminoglycosides and the occurrence of adverse reactions and toxicity are monitored periodically. In the reference center where this study was done, the dosage schedules were chosen in an attempt to prevent drug interactions. In this situation, the availability of a multidisciplinary team combined with a program of active pharmacovigilance can help prevent the occurrence of drug interactions.
The inappropriate use of drugs is observed throughout the world in health care system and the consequences are enormous for patients and communities. Drugs are inappropriately prescribed, dispensed or sold, while many patients fail to take them correctly. In contrast, according to WHO, the rational use of medicines is defined as “Patients receive medications appropriate to their clinical needs, in doses that meet their own individual requirements, for an adequate period of time, and at the lowest cost to them and their community.”44 Additionally, monitoring studies aims to focus on drugs prescription, dispense and administration and may promote the appropriate use and the reduction of abuse or misuse of drugs.45 These studies could collaborate to develop a guide to prescribers, dispensers and the general public. According to this, the rational use of medicines involves all healthcare professionals, directly or indirectly. Previous reports described nurses are able to detect and identify adverse reactions.46-48 However, according to recent studies, contribution of nurses fall shorts than physicians and pharmacists.49,50
According to nurses self-reports, the lack of pharmacological background, mainly regarding relation between theory and practice aspects (i.e. mechanism of drug action and their interactions) is the main factor responsible for this insufficient participation.51 Therefore, educational programs, discussion groups and pharmacovigilance training could be encouraged to improve nursing involvement in detecting and reporting drug-related problems. These strategies will enable nurses to play prominent role in pharmacovigilance practices and could reduce significantly drug interactions and adverse reactions.
Acknowledgments
G.B.V. was supported by a PIBIC studentship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
REFERENCES
]]> 1. Therapeutic National Formulary. Ministério da Saúde, Secretaria de Ciência, Tecnologia e Insumos Estratégicos, Departamento de Assistência Farmacêutica e Insumos Estratégicos. 2th ed. Brasília (DF) [Internet]. 2010 [cited 2014 April 14]. Available from: http://bvsms.saude.gov.br/bvs/publicacoes/formulario_terapeutico_nacional_2010.pdf2. Cruciol-Souza JM, Thomson JC. A pharmacoepidemiologic study of drug interactions in a Brazilian teaching hospital. Clinics. 2006;61:515-20.
3. Carleton BC, Smith MA, Gelin MN, Heathcote SC. Paediatric adverse drug reaction reporting: understanding and future directions. Can J Clin Pharmacol. 2007;14(1):e45-57.
4. Lobo MG, Pinheiro SM, Castro JG, Momenté VG, Pranchevicius MC. Adverse drug reaction monitoring: support for pharmacovigilance at a tertiary care hospital in northern Brazil. BMC Pharmacol Toxicol. 2013;14(5):1-7.
5. Hartshorn EA. Evolution of drug-drug interactions: a personal viewpoint. Ann Pharmacother. 2006;40:112-3.
6. Kearns GL, Abdel-Rahman SM, Blander SW, Blowey DL, Leeder JS, Kauffman RE. Developmental pharmacology – drug disposition, action, and therapy in infants and children. N Engl J Med. 2003;349(12):1157-67.
7. Barterlink IH, Rademaker CMA, Schobben AF, van den Anker JN. Guidelines on pediatric dosing on the basis of developmental physiology and pharmacokinetic considerations. Clin Pharmacokinet. 2006;45(11):1077-97.
8. Morselli PL, Franco-Morselli R, Bossi L. Clinical pharmacokinetics in newborns and infants. Age-related differences and therapeutic implications. Clin Pharmacokinet. 1980;5(6):485-527.
9. Johnson TN. The development of drug metabolising enzymes and their influence on the susceptibility to adverse drug reactions in children. Toxicology. 2003;192(1):37-48.
10. Hayton WL. Maturation and growth of renal function: dosing renally cleared drugs in children. AAPS Pharm Sci. 2000;2(1):22-8.
11. Sweet DH, Bush KT, Nigam SK. The organic anion transporter family: from physiology to ontogeny and the clinic. American Journal of Physiology. Renal Physiology. 2001;281:F197-F205.
12. Strolin BM, Baltes EL. Drug metabolism and disposition in children. Fundam Clin Pharmacol. 2003;17(3):281-99.
13. Anderson GD. Developmental pharmacokinetics. Semin Pediatr Neurol. 2010;17(4):208-13.
14. Turner MA, Catapano M, Hirschfeld S, Giaquinto C. Paediatric drug development: the impact of evolving regulations. Adv Drug Deliv Rev. 2014;73C,2-13.
15. International Classification of Diseases (ICD-10) [Internet]. 2015 [cited 2015 May 14]. Available from: http://apps.who.int/classifications/icd10/browse/2015/en
16. Peterlini MA, Chaud MN, Pedreira, ML. Drug therapy orphans: the administration of intravenous drugs in hospitalized children. Rev Lat Am Enfermagem. 2003;11(1):88-95.
]]>17. Jasiecka A, Máslanka T, Jaroszewski JJ. Pharmacological characteristics of metamizole. Pol J Vet Sci. 2014;17(1):207-14.
18. Rosenkranz, B, Lehr KH, Mackert G, Seyberth HW. Metamizole-furosemide interaction study in healthy volunteers. Eur J Clin Pharmacol. 1992;42(6):593-8.
19. Hinz B, Cheremina O, Bachmakov J, Renner B, Zolk O, Fromm MF, et al. Dipyrone elicits substantial inhibition of peripheral cyclooxygenases in humans: new insights into the pharmacology of an old analgesic. FASEB J. 2007;21(10):2343-51.
20. Kraul H, Pasanen M, Saguache H, Stenbäck F, Park SS, Gelboin HV, et al. Immunohistochemical properties of dipyrone-induced cytochromes P450 in rats. Hum Exp Toxicol. 1996;15(1):45-50.
21. Saussele T, Burk O, Blievernicht JK, Klein K, Nussler A, Nussler, et al. Selective induction of human hepatic cytochromes P450 2B6 and 3A4 by metamizole. Clin Pharmacol Ther. 2007;82(3):265-74.
]]>22. Qin WJ, Zhang W, Liu ZQ, Chen XP, Tan ZR, Hu DL, et al. Rapid clinical induction of bupropion hydroxylation by metamizole in healthy Chinese men. Br J Clin Pharmacol. 2012;74(6):999-1004.
23. Von Moltke LL, Greenblatt DJ, Schmider J, Harmatz JS, Shader RI. Metabolism of drugs by P450, 3A isoforms. Implications for drug interactions in psychopharmacology. Clin Pharmacokinet. 1995;29(1):33-44.
24. Czock D, Keller F, Rasche FM, Häussler U. Pharmacokinetics and pharmacodynamics of systemically administered glucocorticoids. Clinical Pharmacokinetics Journal. 2005;44(1):61-98.
25. Bergmann TK, Barraclough KA, Lee KJ, Staatz CE. Clinical pharmacokinetics and pharmacodynamics of prednisolone and prednisone in solid organ transplantation. Clin Pharmacokinet. 2012;51(11):711-41.
26. Skerjanec A, Wang J, Maren K, Rojkjaer L. Investigation of the pharmacokinetic interactions of deferasirox, a once-daily oral iron chelator, with midazolam, rifampin, and repaglinide in healthy volunteers. J Clin Pharmacol. 2010;50(2):205-13.
]]> 27. Rendić S. Drug interactions of H2-receptor antagonists involving cytochrome P450 (CYPs) enzymes: from the laboratory to the clinic. Croat Med J. 1999;40(3):357-67.28. Westphal JF. Macrolide-induced clinically relevant drug interactions with cytochrome P-450A (CYP) 3A4: an update focused on clarithromycin, azithromycin and dirithromycin. Br J Clin Pharmacol. 2000;50(4):285-95.
29. Perucca E. Clinically relevant drug interactions with antiepileptic drugs. Br J Clin Pharmacol. 2006;61(3):246-55.
30. Gonzalez FJ, Coughtrie M, Tukey RH. Metabolismo dos fármacos. In: Brunton LL, Chabner BA, Knollmann BC, eds. As bases farmacológicas da terapêutica de Goodman & Gilman. 12 ed. Porto Alegre: AMGH Publisher Ltda./MacGraw-Hill Education; 2012. p. 123-43.
31. Borowitz D, Robinson KA, Rosenfeld M, Davis SD, Sabadosa KA, Spear SL, et al. Cystic Fibrosis Foundation evidence-based guidelines for management of infants with cystic fibrosis. Journal of Pediatrics. 2009;155(6):S73-93.
32. Filho LVRFS, Damaceno N, Reis FJC, Hira AY. Registro brasileiro de fibrose cística [Internet]. 2010 [cited 2014 March 22]. Avaible from: http://www.cysticfibrosisdata.org/LiteratureRetrieve.aspx?ID=135160
33. Adde FV, Marostica PJC, Ribeiro MAGO, Santos CIS, Sol D, Vieira SE. Fibrose cística: diagnóstico e tratamento [Internet]. 2014 [cited 2014 April 20] Avaible from: http://www.projetodiretrizes.org.br/ans/diretrizes/fibrose_cistica-diagnostico_e_tratamento.pdf
34. Prayle A, Smyth AR. Aminoglycoside use in cystic fibrosis: therapeutic strategies and toxicity. Curr Opin Pulm Med. 2010;16(6):604-10.
35. Liangos O. Drugs and AKI. Minerva Urol Nefrol. 2012;64(1):51-62.
36. Khalili H, Bairam S, Kargar M. Antibiotics induced acute kidney injury: incidence, risk factors, onset time and outcome. Acta Med Iran. 2013;51(12):871-8.
37. Barza M, Loannidis JPA, Capelleri JC, Lau J. Single or multiple daily doses of aminoglycosides: a meta-analysis. Br Med J. 1996;312(7027):338-45.
38. Flume PA, Mogayzel PJJr, Robinson KA, Goss, CH, Rosenblatt, RL, Kuhn RJ, et al. Clinical Practice Guidelines for Pulmonary Therapies committee. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am J Respir Crit Care Med. 2009;180(9):802-8.
39. Smyth AR, Bhatt J. Once-daily versus multiple-daily dosing with intravenous aminoglycosides for cystic fibrosis. Cochrane Database Syst Rev. 2014;2:CD002009.
40. Bhatt JM. Treatment of pulmonary exacerbations in cystic fibrosis. Eur Respir Rev. 2013;22:205-16.
41. Castro MCS, Firmida MC. O tratamento na fibrose cística e suas complicações. HUPE. 2011;10:82-108.
42. Geller DE. Aerosol antibiotics in cystic fibrosis. Respir Care. 2009;54(5):658-70.
43. Levy CE. Microbiologia no trato respiratório na fibrose cística. In: Paschoal IA, Pereira MC, eds. Fibrose Cística. São Paulo: Yends; 2010. p. 201-12.
44. Promoting Rational Use of Medicines: Core Components-WHO Policy perspectives on medicine, No. 005, September 2002. Essential medicines and Health Products Information Portal. A World Health Organization Resource [internet]. 2015 [cited 2015 October 23]. Available from: http://www.who.int/medicines/publications/policyperspectives/ppm05en.pdf
45. Jain S, Upadhyaya P, Goyal J, Kumar A, Jain P, Seth V, et al. A systematic review of prescription pattern monitoring studies and their effectiveness in promoting rational use of medicines. Perspect Clin Res. 2015;6(2):86-90.
46. Bergqvist M, Ulfvarson J, Andersen Karlsson E, von Bahr C. A nurse-led intervention for identification of drug-related problems. Eur J Clin Pharmacol. 2008;64:451–56.
47. Morrison-Griffiths S, Walley TJ, Park BK, Breckenridge AM, Pirmohamed M. Reporting of adverse drug reactions by nurses. Lancet. 2003;361(9366):1347-8.
48. Ulfvarson J, Mejyr S, Bergman U. Nurses are increasingly involved in pharmacovigilance in Sweden. Pharmacoepidemiol Drug Safety. 2007;16(5):532-7.
]]>49. Johansson-Pajala RM, Martin L, Fastbom J, Jorsäter Blomgren K. Nurses' self-reported medication competence in relation to their pharmacovigilant activities in clinical practice. J Eval Clin Pract. 2015;21(1):145-52.
50. Mendes D, Alves C, Batel Marques F. Nurses’ spontaneous reporting of adverse drug reactions: expert review of routine reports. J Nurs Man. 2014;22:322-30.
51. King RL. Nurses’ perceptions of their pharmacology educational needs. J Adv Nurs.2004;45(4):392-400.
Recibido: 26 de agosto de 2015.
Aprobado: 3 de noviembre de 2015.
]]> Márcia Yuriko Masukawa. Department of Physiological Sciences. Santa Casa de São Paulo. School of Nursing. SP, Brazil. Correo electrónico: marciaymxix@gmail.com ]]>