A POLYCLONAL -ANTIBODY-IMMUNOPEROXIDASE-CONJUGATE FOR THE SPECIFIC DETECTION OF PORCINE CIRCOVIRUS TYPE 2
ANTICUERPO POLICLONAL CONJUGADO CON PEROXIDASA PARA LA DETECCIÓN ESPECÍFICA DE CIRCOVIRUS PORCINO TIPO 2
L.J. Pérez, Heidy Díaz de Arce, Maritza Barrera, Sara Castell, María T. Frías
Departamento Virología, Centro Nacional de Sanidad Agropecuaria (CENSA), Apartado 10, San José de las Lajas, La Habana, Cuba. Correo electrónico: lesterjosue@censa.edu.cu ]]>
ABSTRACT
Porcine circovirus 2 is nowadays accepted as the essential infectious agent of postweaning multisystemic wasting syndrome which causes severe economic losses in porcine production worldwide. The diagnosis of PMWS is a difficult task and must follow three criteria: (i) the presence of compatible clinical signs, (ii) the presence of characteristic microscopic histopathological lesions, and (iii) the presence of PCV2 within these lesions in moderate or high. The presence of PCV2 in lymphoid tissues must be demonstrated by in situ hybridization or immunohistochemical methods. The in situ hybridization is a more complex and expense compared to other diagnostic tools, on the other hand, one problem concerning the immunohistochemical methods for PMWS diagnostic is the lack of a commercial anti-PCV2 peroxidase conjugate; therefore, the aim of this work was to obtain a polyclonal-antibody-immunoperoxidase-conjugate for the PCV2 specific detection. An anti-PCV2-peroxidase conjugated for the PCV2 specific detection was obtained based on the use of the available commercial vaccine against PCV2 as immunogenic inoculation for producing a polyclonal antibody in rabbits. The conjugate obtained was able to discriminate between PCV2 and PCV1 infections and a high sensitivity and specificity of the conjugate were observed.
Key words: porcine circovirus 2; immunohistochemical; conjugate; polyclonal antibody
RESUMEN
El circovirus porcino 2 (PCV2) se considera actualmente como el agente infeccioso esencial del síndrome del desmedro post-destete (PMWS), el cual causa grandes pérdidas económicas en la producción porcina de todo el mundo. El diagnóstico del PMWS es difícil y debe seguir tres criterios: (i) la presencia de signos clínicos compatibles, (ii) la presencia de lesiones histopatológicas microscópicas características y (iii) la presencia de cantidades moderadas o altas del PCV2 dentro de las lesiones. La presencia de PCV2 en los tejidos linfoides se debe demostrar por hibridación in situ o métodos inmunohistoquímicos. La hibridación in situ es un método más complejo y costoso comparado con otras herramientas diagnósticas, por otra parte, un problema con relación a los métodos inmunohistoquímicos para el diagnóstico de PMWS es la carencia de un conjugado con peroxidasa comercial específico contra PCV2, por lo tanto el objetivo de este trabajo fue obtener un anticuerpo policlonal conjugado con peroxidasa par la detección específica de PCV2. Se obtuvo un conjugado con peroxidasa para la detección específica de PCV2 con el uso de una vacuna contra PCV2 disponible comercialmente como inmunogéno en conejos con el fin de obtener el anticuerpo policlonal contra PCV2. El conjugado obtenido fue capaz de discriminar la infección por PCV2 de la infección por PCV1, y mostró elevadas sensibilidad y especificidad.
Palabras clave: cirovirus porcino 2; inmunohistoquímica; conjugado; anticuerpo policlonal
]]>
INTRODUCTIONPorcine circoviruses are classified into two species included into Circoviridae family (1). PCV1 was discovered as a noncytopathic contaminant of the continuous porcine kidney cell line PK-15 (ATCC CCL-33 and is not regarded as a pathogen for pigs (2,3). PCV2 is nowadays accepted as the essential infectious agent of postweaning multisystemic wasting syndrome (PMWS) (4) porcine dermatitis and nephropathy syndrome (PDNS), (5,6), and has been also associated to reproductive failure (7,8,9). All these syndrome were collectively grouped as porcine circovirus diseases (PCVD) (10).
The PMWS is the most important disease within PCVD due to this syndrome disease causes severe economic losses in consequences of an increased mortality rates and reduced feed conversion efficiency in weaning and fattening pigs at the worldwide (10). In addition, PMWS is considered an immunosu-ppressant disease, thence facilitates the infection with opportunist pathogens affecting the swine herd health status (11).
The diagnosis of PMWS is difficult and must follow three criteria: (i) the presence of compatible clinical signs, (ii) the presence of characteristic microscopic histopathological lesions, and (iii) the presence of PCV2 within these lesions in moderate or high amounts (10,12). The presence of PCV2 DNA or antigen in lymphoid tissues must be demonstrated by in situ hybridization and immunohistochemical methods (10,12,13).
In spite of that in situ hybridization has recently become increasingly important in diagnostic procedures (14,15,16), the use of this technique remains restricted to a few diagnostic laboratories because of its greater technical complexity and expense compared to other diagnostic tools. In the case of the immunohistochemical although a certain expertise is required, this is a cost effective and easy method that could be implemented in several laboratories for the PMWS diagnostic. Nevertheless, one problem concerning the immunohistochemistry methods for the PMWS diagnostic is the lack of a commercial anti-PCV2 peroxidase conjugate, for this reason, several laboratories has developed its own anti-PCV2 conjugate for this purpose (17,18). Therefore, the aim of this work was to obtain a polyclonal-antibody-immunoperoxidase-conjugate for the PCV2 specific detection.
MATERIAL AND METHOD
Virus and cell lines
The Cuban PCV2 field isolate identified as PNE1 (genome sequence deposited in GenBank under the accession number FM999737) from CENSA viral collection (19) was propagated as follows: the PK15A cell line monolayer was trypsinised and resuspended to contain 5x104 cells/mL in growth medium (Dulbecco's modified Eagle's medium, DMEM) supplemented with 10% foetal bovine serum (Gibco, free from virus) and 100 µg/ml gentamicin. Three-millilitres of the virus propagated was added to 20 mL of this cell suspension and seeded in 10 mL volumes into two 25 cm2 cell culture flasks. After 18 h incubation at 37ºC the resulting semi-confluent monolayers were treated with 300mMD-glucosamine in Hanks basal salt solution for 30 min and the virus/cells were passaged.
Nucleic acid isolation and polymerase chain reaction ]]>
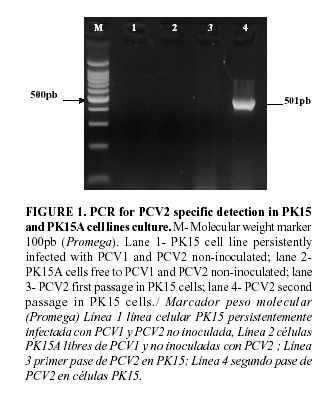
Total DNA was extracted from 100 µl of each sample (10% tissue homogenate), with Wizard® Genomic DNA Purification Kit, (Promega, Madison, WI, USA) following manufacturer instructions. DNA was diluted in 10 µl of nuclease free water (Promega, Madison, WI, USA). To detect PCV2 DNA in order to evaluate the status of PCV2 infections in pigs, a polymerase chain reaction (PCR) assay was carry out as described by Sandvik et al., (2001) (20), using the primers PCV2-2A: 5'-CACCTTCGGATATACTGTCAA-3' and PCV2-2B:5'-TACATGGTTACACGGATA TTGTA-3'. The primer pair targeted an amplicon of 501 bp of the ORF2. Briefly, the amplification reaction was carried out in a volume of 50 µl comprised of 2 µl of DNA sample (extracted as described above), 1x GoTaq® Flexi DNA Polymerase (Promega, Madison, WI, USA) [200uM of each dNTP, 1.5mM MgCl2 (pH 8.5)] and 1 µM of each primer. The PCR reaction was done under the conditions described by Sandvik et al. (20) in a thermal cycler (Eppendorf Mastercycler). The amplicons (10 µl) were visualized by electrophoresis on 2.0% agarose gel in TBE buffer (90mM Tris-borate, 2 mM EDTA) ethidium bromide stained.Polyclonal antiserum
A rabbit polyclonal antiserum was produced at the Institute, briefly: two Chinchilla rabbits were intradermic immunizated with a total 8ml of a killed PCV1-2 chimeric vaccine (Suvaxyn® PCV2 One DoseTM; Fort Dodge Animal Health, Inc.,Fort Dodge, IA, USA; Lot number 1861133A) delivered in 10 doses on the back in multiple sites close to the neck and groin. A booster inoculation was carried out 3 weeks later at same inoculum and via. In order to improve affinity of the antiserum a third inoculation at 4 weeks after a second inoculation was performed. The animals were monitored; blood samples were taken before each immunization and evaluated on PK15 monolayer infected with PCV2 by an immunoperoxidase monolayer assay described by Fort et al. (21). Rabbits were euthanized, and serum was harvested 2 weeks after the final immunization and anti-PCV2 antiserum was assessed.
IgG purification and conjugated
The immunoglobulin using a protein A-Sepharose (Amersham, Pharmacia) column chromatography were purified following standard protocols for this method (22). The concentration of the immunoglobulin purified was calculated using the expression [1]. A total of 10 mg/mL of immunoglobulin purified was conjugated with peroxidase (Peroxidase from horseradish Type XII, Sigma) using the oxidation with periodate method (23) and the Conjugation ratio (IgG/peroxidase) was calculated.
Conc(mg/mL)=O.D280*dilution factor*extinction coef IgG(rabbit) expression [1]
Specificity of the anti-PCV2 conjugate
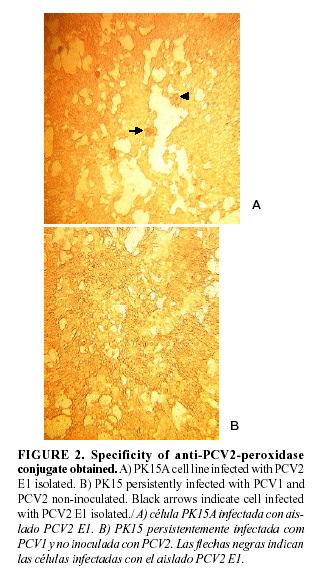
The specificity of the anti-PCV2 conjugated was evaluated using PK15A cell line infected with the PCV2 isolated E1 and the PK15 cell line persistently infected with PCV1. A immunoperoxidase monolayer assay as described by Fort et al., 2007 (21) was carry out and the presences of PCV2 and PCV1 in PK15A and PK15 cell lines was checked thorough a PCR assay described by Sandvik et al. (20) and Fenaux et al. (24) respectively.
RESULTS ]]>
Obtaining the anti-PCV2-peroxidase conjugateTwo fraction of IgG total corresponding to a concentration of 24mg/mL each were collected. The anti-PCV2-peroxidase conjugated was obtained and the IgG/peroxidase ratio showed a value of 0.4. The obtained conjugated was mixed with 1% of bovine serum albumin (BSA) and a final mix (v/v) glycerol/ anti-PCV2-peroxidase conjugated was conserved at -20ºC.
Test of specificity of the anti-PCV2 conjugate
The presence of PCV2 E1 isolated in PK15A infected cell lines was verified. Likewise the absence of PCV2 in PK15 persistently infected with PCV1 was tested, (figure1). (In this same sense the presence of PCV1 in persistently infected with PCV1 was make sure (data not shown)). The anti-PCV2-peroxidase conjugate obtained when a dilution 1/40 was used revealed a specific reaction in PCV2 infected cell and a lack of color in PK15 PCV1 infected cell was observed, figure 2. Hence, the anti-PCV2-peroxidase conjugate obtained showed to be specific in the PCV2 detection.

]]>
DISCUSSION
In this work an anti-PCV2-peroxidase conjugated for the PCV2 specific detection was obtained.
The PCV2 is considered an emerging pathogen, causing economically important diseases worldwide (25,26). This virus reported by Meehan et al., 1998 as a novel circovirus (27) has nucleotide identity of 66% concerning to the proteins encoded by ORF2 to be compared with PCV1 (28), thence these two viral agent can be differentiated using polyclonal antibodies against the capside protein (18). ]]>
The fact that for the PMWS diagnosis the presence of the PCV2 within these lesions in moderate or high amounts of the virus must be demonstrated by immunohistochemical methods (10,12) as well as the lack of commercial anti-PCV2-peroxidase conjugate imply the requirement by the laboratories involved in the PMWS diagnosis to develop their own anti-PCV2-peroxidase conjugate.The use of the killed PCV1-2 chimeric vaccine for obtaining polyclonal antibodies against PCV2 protein capside described here is a useful strategy that could be assume for those laboratories with problems in cell cultures technologies or because of the complicated PCV2 isolation and multiplication, hence to obtain enough viral amount to be used as immunogenic inoculation will be a very difficult task.
The PCV2 infection and replication in cell culture occur to very low titer of the virus (29), thus with the purpose of increasing the viral amount different strategies has been described (29,30). Nevertheless, the fact that the PCV2 multiplication in cell culture is carried out to very low amount of the virus suggests that the anti-PCV2-peroxidase conjugated obtained has a high sensitivity and could be able to distinguish between subclinical PCV2 infection and PMWS condition.
The high sensitivity and specificity of the anti-PCV2-peroxidase conjugate obtained in this work will allow perform the PMWS diagnosis in the future. Nevertheless a further evaluation of this conjugate in tissues samples will be needed to carry out.
In summary, in this paper we described the anti-PCV2-peroxidase conjugate obtainment based on the use of the available commercial vaccine against PCV2 as immunogenic inoculation to produce a polyclonal antibody in rabbits. The obtained conjugate was able to discriminate between PCV2 and PCV1 infections and a high sensitivity of the conjugate was observed.
REFERENCES
1. Todd D, McNulty MS, Mankertz A, Lukert PD, Randles JW, Dale JL. Circoviridae. In: van Regenmortel MHV, Fauquet CM, Bishop DHL, Carstens EB, Estes MK, Lemon SM, Malinoff J, Mayo MA, McGeoch DJ, Pringle CR, Wickner RB. (Eds.), Virus Taxonomy. Seventh report of the ICTV. Academic Press, San Diego. 2000.
2. Allan GM, McNeilly F, Cassidy JP, Reilly GA, Adair B, Ellis WA, et al. Pathogenesis of porcine circovirus; experimental infections of colostrum deprived piglets and examination of pig foetal material. Vet Microbiol. 1995;44:49-64.
3. Tischer I, Mields W, Wolff D, Vagt M, Griem W. Studies on epidemiology and pathogenicity of porcine circovirus. Arch Virol. 1986;91:271-276.
4. Harding J, Clark E. Recognizing and diagnosing postweaning multisystemic wasting syndrome (PMWS). Swine Health Prod. 1998;5:201-203.
5. Smith WJ, Thompson JR, Done S. Dermatitis/ nephropathy syndrome of pigs. Vet Rec.1993;132:47.
6. Rosell C, Segalés J, Ramos-Vara JA, Folch JM, Rodriguez-Arrioja GM, Duran CO, et al. Identification of porcine circovirus in tissues of pigs with porcine dermatitis and nephropathysyndrome. Vet Rec. 2000;146:40-43.
7. West KW, Bystrom J, Wojnarowicz C. Myocarditis and abortion associated with intrauterine infection of sows with porcine circovirus-2. J Vet Diagn Invest.1999;11(6):530-532.
8. O'Connor B, Gauvreau H, West K, Bogdan J, Ayroud M, Clark E, et al. Multiple porcine circovirus 2 associated abortion and reproductive failure in a multiple-site swine production unit. Can Vet J. 2001;42:551-553.
9. Ohlinger VF, Schmidt U, Pesch S. Studies on pathogenic aspects of the post-weaning multisystemic wasting syndrome (PMWS). 2000. In: Proceedings of the 16th International Pig Veterinary Society Congress.
10.Segalés J, Allan GM, Domingo M. Porcine circovirus diseases. Anim Health Res Rev.2005;6:119-142.
11.Darwich L, Segalés J, Mateu E. Pathogenesis of postweaning multisystemic wasting syndrome caused by Porcine circovirus 2: an immune riddle. A review. Arch Virol. 2004;149:857-74.
12.Chae C. Postweaning multisystemic wasting syndrome: a review of aetiology, diagnosis and pathology. Vet J. 2004;168:41-49.
13.Segalés J, Domingo M. Post-weaning multisystemic wasting syndrome (PMWS) in pigs: a review. Vet Quart. 2002;24:109-24.
14.Dupont K, Nielsen EO, Bækbo P, Larsen LE. Genomic analysis of PCV2 isolates from Danish archives and a currentPMWS case-control study supports a shift in genotypes with time. Vet. Microbiol. 2007.doi: 10.1016/j.vetmic.2007.09.016.
15.Grau-Roma L, Crisci E, Sibila M, López-Soria S, Nofrarias M, Cortey M, et al. A proposal on porcine circovirus type 2 (PCV2) genotype definitions and their relation with postweaning multisystemic wasting syndrome (PMWS) occurrence. Vet Microbiol. 2008;128:23-35.
16.Rosell C, Segales J, Plana-Duran J, Balasch M, Rodriguez-Arrioja GM, Kennedy S, et al. Pathological, immunohistochemical, and in situ hybridization studies of natural cases of postweaning multisystemic wasting syndrome (PMWS) in pigs. J Comp Pathol. 1999;120:59-78.
17.McNeilly F, McNair I, Mackie DP, Meehan BM, Kennedy S, Moffett D, et al. Production, characterisation and applications of monoclonal antibodies to porcine circovirus 2. Arch Virol. 2001;146:909-922.
18.Sorden SD, Harms PA, Nawagitgul P, Cavanaugh D, Paul PS. Development of a polyclonal-antibody-based immunohistochemical method for the detection of type 2 porcine circovirus in formalin-fixed, paraffin-embedded tissue. J Vet Diagn Invest. 1999;11:528-530.
19.Pérez LJ, Díaz de Arce H, Percedo MI, Domínguez P, Frías MT. First report of porcine circovirus type 2 infections in Cuba. Res Vet Sci. Submitted for publication.
20.Sandvik T, Grierson S, King DP, Spencer Y, Banks M, Drew T. Detection and genetic typing of porcine circovirus DNA isolated from archived paraffin embedded pig tissues. Comp. Virol. Proceedings - ss DNA Viruses of Plants, Birds, Pigs and Primates, Saint-Malo, 24-27 September, 2001.
21.Fort M, Olvera A, Sibila M, Segalés J, Mateu E. Detection of neutralizing antibodies in postweaning multisystemic wasting syndrome (PMWS)-affected and non-PMWS-affected pigs. Vet Microbiol. 2007;125:244-55.
22.Kruger NJ, Hammond JBW. Purification of Immunoglobulins Using Protein A-Sepharose. Methods in Molecular Biology. 2008 chapter, 26, pp363-371. DOI:10.1385/0-89603-126-8:363.
23.Wilson MB, Nakane PK. Recent developments in the periodate method of conjugating horseradish peroxidase (HRPO) to antibodies. En: Knapp W, Hollubat K, Wick G, eds. Immunofluorescence and related staining techniques. New York: Academic, 1978; 215.
24.Fenaux M, Halbur PG, Gill M, Toth TE, Meng XJ. Genetic characterization of type 2 porcine circovirus (PCV-2) from pigs with postweaning multisystemic wasting syndrome in different geographic regions of North America and
development of a differential PCR-restriction fragment length polymorphism assay to detect and differentiate between infections with PCV-1 and PCV-2. J Clin Microbiol. 2000;38:2494-503.
25.Hesse R, Kerrigan M, Rowland R. Evidence for recombination between PCV2a and PCV2b in the field. Virus Res. 2008;132:201-07.
26.Ostanello F, Caprioli A, Di Francesco A, Battilani M, Sala G, Sarli G, et. al. Experimental infection of 3-week-old conventional colostrum-fed pigs with porcine circovirus type 2 and porcine parvovirus. Vet. Microbiol. 2005;108:179-86.
27.Meehan BM, McNeilly F, Todd D, Kennedy S, Jewhurst VA, Ellis JA, et. al. Characterization of novel circovirus DNAs associated with wasting syndromes in pigs. J Gen Virol. 1998;79:2171-79.
28.Hamel AL, Lin LL, Nayar GP. Nucleotide sequence of porcine circovirus associated with postweaning multisystemic wasting syndrome in pigs. J Virol. 1998;72:5262-5267.
29.Allan GM, McNeilly F, Kennedy S, Draft B, Clarke EG, Ellis JA, et al. Isolation of porcine circovirus-like virus from pigs with a wasting disease in the USA and Europe. J Vet Diagn Invest. 1998;10(1):3-10.
30.Misinzo G, Delputte PL, Lefebvre DJ, Nauwynck HJ. Porcine circovirus 2 infection of epithelial cells is clathrin-, caveolae- and dynamin-independent, actin and Rho-GTPase-mediated, and enhanced by cholesterol depletion. Virus Res. 2009;139:1-9.
]]>
(Recibido 23-8-2009; Aprobado 3-9-2009) ]]>