REPORT
The inhibition of pathogen-associated molecular patterns confers high protection against fungi and oomycetes in plants
Inhibición de patrones moleculares asociados confiere una alta protección frente a hongos y oomicetos en las plantas
]]>
Ingrid Hernandez-Estévez1, Roxana Portieles-Alvarez1, Yussuan Silva2, Merardo Pujol-Ferrer1, Osvaldo Oliva-Borbón1, Orlando Borrás-Hidalgo1
1 División de Plantas. Centro de Ingeniería Genética y Biotecnología, CIGB. Ave. 31 e/ 158 y 190, Cubanacán, Playa, CP 11600, La Habana, Cuba.
2 Instituto de Investigaciones del Tabaco, IIT. Carretera Tumbadero km 8 ½, San Antonio de los Baños, Mayabeque, CP 3500, La Habana, Cuba.
ABSTRACT
Crops of agricultural interest are highly affected by fungi- and oomycetes-caused diseases in Cuba and worldwide. The search for alternatives for its control continues, as a major challenge with the use of biotechnological techniques. In nature, plants are exposed to biotic stress and develop resistance against pathogenic infection through the fast activation of the innate immune system. Such an effective resistance response requires the detection and fast inhibition of the evolutionary conserved pathogen-associated molecular patterns (PAMPs). These PAMPs comprise, among others, proteases and polygalacturonases, which mediate the initial pathogenicity mechanisms during infection that counteract the initial plant defensive responses. In this work, inhibitors of pathogen’s proteases and polygalacturonases were developed to generate plant resistance against a wide spectrum of fungi- and oomycetes-caused diseases. Tobacco plants expressing a polygalacturonase inhibitor conferred, for the first time, high levels of resistance against this type of pathogens under field conditions. Additionally, a novel protease inhibitor effective against pathogens’ proteases was identified and characterized, which also provided resistance against pathogenic oomycetes in plants. This research granted the 2013 Award of the Cuban National Academy of Sciences.
Keywords: disease resistance, polygaracturonase inhibitor, protease inhibitor, plant protection, fungi, oomycetes.
RESUMEN
Las enfermedades provocadas por hongos y oomicetos en cultivos son las mayores limitantes en la producción agrícola a escala mundial. Buscar alternativas biotecnológicas para el control de estas enfermedades es el mayor reto de los investigadores de este tema. Las plantas están expuestas a estrés biótico, y resisten a la infección de agentes patógenos por medio de la activación del sistema de inmunidad innato. Esta activación depende de la rápida detección o inhibición de patrones moleculares conservados en muchos agentes patógenos. Los patrones moleculares asociados con agentes patógenos, como las proteasas y las poligalacturonasas, son mecanismos de patogenicidad utilizados por los agentes patógenos en el inicio de la infección, para vencer las defensas de la planta. Este artículo describe el uso de inhibidores de proteasas y poligalacturonasas de agentes patógenos para la resistencia de un amplio espectro de enfermedades provocadas por hongos y oomicetos en plantas. La expresión de un inhibidor de poligalacturonasas en plantas de tabaco permitió obtener, por primera vez, niveles elevados de resistencia a agentes patógenos en condiciones de campo. Además se identificó, caracterizó y analizó la función de un nuevo inhibidor de proteasa de agentes patógenos. Este inhibidor confirió altos niveles de resistencia a oomicetos patógenos de plantas. El trabajo mereció el Premio Anual de la Academia de Ciencias de Cuba, 2013.
Palabras clave: resistencia a enfermedades, inhibidor de poligalacturonasa, inhibidor de proteasa, protección de plantas, hongos, oomicetos.
INTRODUCTION
Throughout the evolution, plants have developed strategies to recognize pathogens and to generate an effective protective response. Likewise, pathogens have evolved mechanisms to evade, suppress or both the plant defensive responses. Plants are resistant to microbial infection through its basal defensive mechanism of the innate immune system. It becomes activated by the recognition of evolutionary-conserved pathogens’ molecules which were denominated pathogen-associated molecular patterns (PMAPs), which include proteins, enzymes, peptides, carbohydrates and lipids.
In general, there are two PAMPs activation pathways in plants [1]. The first one is mediated by PMAPs receptors or inhibitors. The second one is mainly intracellular and acts through polymorphic proteins bearing nucleotide binding sites and leucine repeats (NBS-LRR), most of them encoded by resistance genes (R). In fact, some authors have proposed a so-called ‘Zig-zag’ model for the functioning of the immune system in plants [1]. On its first phase, PAMPs are detected by its receptors or neutralized by host inhibitors, as part of the PAMPs-induced immune activation, which could halt plant colonization by the pathogen. On the second phase, a given effector is recognized by one of the NBS-LRR proteins what triggers an activation pathway (the effector activated immunity pathway).
]]> In these processes, the cell wall is the primary line of defense against pathogenic microorganisms. Most of them produce cell wall lytic enzymes, particularly relevant for its specialized penetration structures. Among them, polygalacturonases (PGs) play a significant role at the initial infection stages. In fact, a set of proteins known as polygalacturonases-inhibitory proteins (PGIPs), which recognize PGs and interferes plant cell wall degradation [2].PGIPs bear leucine-rich repeats, as most PAMPs receptors [3], and are able to recognize PGs from microorganisms and insects. They not only bind to PGs and delay pectin hydrolysis, but also favor the accumulation of oligogalacturonides (OG), a type of damage-associated molecular patterns (DAMPs) which, like PAMPs, activate the innate immune response in plants [4]. For example, there was well established how relevant PGIPs are for plant resistance against the infection of the necrophytic fungus Botrytis cinerea. Transgenic tomato and grape plants expressing a pear PGIP, and transgenic tobacco and Arabidopsis plants expressing a PGIP from or Arabidopsis, respectively, shown improved resistance against Botrytis sp. infection in green house experiments [5-8]. Monocotyledonous plants have been also protected by the transgenic expression of a bean PGIP against the infection by Fusarium graminearum and Bipolaris sorokiniana fungi, in spite of showing low cell wall pectin content [9, 10].
For the same purposes, protein inhibitors have been also considered, being among the main sets of proteins induced by the plant-pathogen interaction. Plant protease inhibitors are normally expressed in seeds and tubers and become induced in the plant’s vegetative organs as in leaves and roots. They display two main functions: 1) to regulate the plant’s endogenous proteases, and 2) to inhibit the exogenous proteases of plant pathogens.
The use of protease inhibitors to protect plants from fungi and bacterial infection has been reported [11-14]. Increased levels of chemotrypsin and trypsin inhibitors have been correlated with plant resistance against different pathogens [12, 15]. This process was first identified in tomato plants infected with Phytophthora infestans [16]. Previous studies showed that potato tubers accumulate serin-proteinase inhibitors in response to the attack by P. infestans [17, 18]. Noteworthy, PAMPs-mediated resistance shows a wider spectrum and last longer than that mediated by protease inhibitors.
Hence, in this work we report the results obtained on using both mechanisms of PAMPs inhibitors-mediated resistance for plant protection against fungi and oomycetes.
RESULTS AND DISCUSSION
Polygalacturonase inhibitory protein (PvPGIP2)
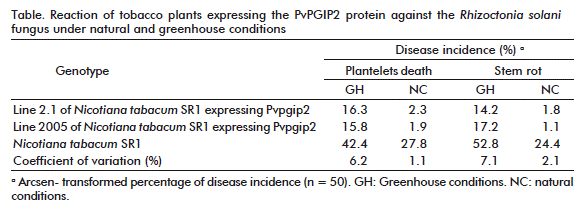
The effect of the polygalacturonase inhibitory protein (PvPGIP2) of Phaseolus vulgaris protects tobacco plants against the infection of relevant pathogens such as the Rhizoctonia solani fungus and Phytophthora parasitica var. nicotianae and Peronospora hyoscyami f. sp. tabacina oomycetes. Hence, the use of PvPGIP2 as a powerful, wide spectrum genetic engineering tool was proposed to confer disease resistance.
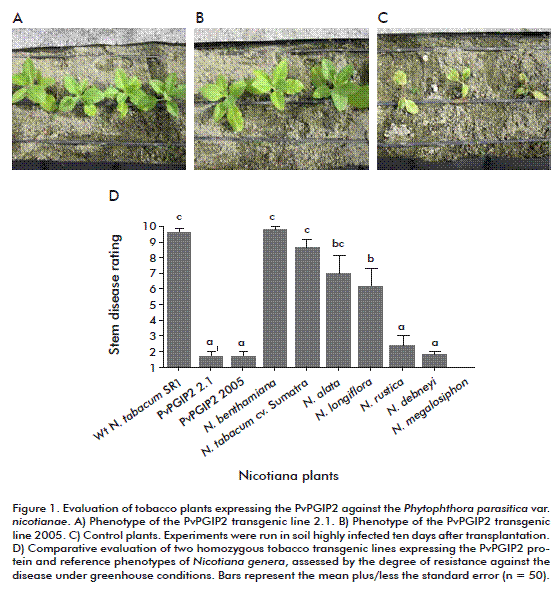
Under greenhouse conditions, the main symptoms of R. solani, control plants developed small stem lesions, which progressively spread through the stem, turning it brown and causing its death. By the contrary, transgenic tobacco lines expressing PvPGIP2 protein developed sparse and very limited disease symptoms (Table). In fact, symptoms coincided with an increase in fungus biomass in colonized control roots while there was no significant increase in transgenic lines. Moreover, under greenhouse conditions, both transgenic tobacco lines expressing PvPGIP2 were extraordinarily resistant to P. parasitica var. nicotianae (Figure 1).
]]>
Two weeks after inoculation, mild symptoms appeared in control plants, while there were no detectable symptoms in transgenic plants. Nevertheless, severe disease effects were evident in control plants after 5 weeks (withered leaves and stem rot) but, remarkably, transgenic plants remained healthy with a resistance similar to the natural high resistance shown by Nicotiana species.
Experiments were also run under field conditions during the winter, when the cold and wet climate promotes the incidence of blue mold disease in tobacco, which is caused by P. hyoscyami f. sp. tabacina in Cuba. Once again, transgenic plants developed high levels of resistance comparable to that of naturally-resistant Nicotiana species, demonstrating that the expression of the PvPGIP2 gene, which encodes a PG-inhibitory protein, is a feasible way to confer high resistance against fungi and oomycetes under greenhouse or field conditions. This is a good strategy to confer resistance in economically relevant crops against oomycetes, a high-incidence group of microorganisms which causes great economic losses and significant environmental damage on natural ecosystems.
Pathogen protease-inhibitory protein NmIMSP
Gene regulation during the N. megalosiphon - P. parasitica var. Nicotianae interaction was characterized with the aid of SuperSAGE technology, particularly targeting induced microbial protease inhibitor expression. A cDNA coding for a protease inhibitor named NmIMSP was identified as overexpressed and associated to the defensive response of N. megalosiphon. The highest expression levels were detected in leaves, which remained constant over the test period.
Conversely, the functional silencing of NmIMSP expression compromised the N. megalosiphon resistance to the infection. Stem damage one week post-inoculation in unsilenced control plants was established as the unit of disease-related damage (n = 15; mean ± standard deviation), with a 1-to-10 evaluation scale from high resistance to susceptibility. Transgenic plants remained resistant when unsilenced or by silencing an unrelated gene, while NmIMSP-silenced transgenic plants showed a 3.2 ± 0.1 damage degree. Highly susceptible N. tabacum cv. ‘Sumatra’ plants were used as control (damage degree 9.8 ± 0.1). These results corroborated the role of this gene on the observed plant defensive response.
Further evidences were obtained in N. benthamiana plants, where the expression of the NmIMSP gene at high levels made plants highly resistant to P. parasitica var. nicotianae and P. hyoscyami f. sp. tabacina infection, under greenhouse conditions (Figure 2). Phylogenetic studies in a set of 25 proteins revealed that NmIMSP belongs to a subgroup of Nicotiana IMSPs. Specifically, NmIMSP was highly homologous to N. tabacum IMSP members, including one induced during the interaction of the tobacco mosaic virus with N. tabacum cv. Samsun NN. Noteworthy, N. tabacum cv. Samsun NN and N. benthamiana are highly susceptible to P. parasitica var. nicotianae and P. hyoscyami f. sp. tabacina.
Unlikely, the few differences in the ISMP aminoacidic sequences among Nicotiana members would not be responsible for the increased resistance provided by ISMP in the N. benthamiana susceptible specie against these oomycetes. Probably, the baseline or delayed expression of ISMP in N. tabacum cv. Samsun NN and N. benthamiana plants would be insufficient to stop these pathogens’ infection.
]]> Nevertheless, the overexpression of the ISMP gene in the species tested conferred high protection against the infection by P. parasitica var. nicotianae and P. hyoscyami f. sp. tabacina, respectively. Undoubtedly, it was evidenced the relevance of this gene for the plants’ defensive response against oomycetes.
MAIN PRACTICAL RELEVANCE OF THE STUDY
The major contribution of the study was to increase plant resistance against high incidence pathogens of economically relevant plants, through the use of genes encoding the protease inhibitor and the polygalacturonidase, as part of genetic improvement programs. By these means, a wide-spectrum resistance can be developed in crops, by inhibiting PAMPs.
REFERENCES
1. Jones JD, Dangl JL. The plant immune system. Nature. 2006;444(7117):323-9.
2. De Lorenzo G, D'Ovidio R, Cervone F. The role of polygalacturonase-inhibiting proteins (PGIPs) in defense against pathogenic fungi. Annu Rev Phytopathol. 2001;39:313-35.
3. Casasoli M, Federici L, Spinelli F, Di Matteo A, Vella N, Scaloni F, et al. Integration of evolutionary and desolvation energy analysis identifies functional sites in a plant immunity protein. Proc Natl Acad Sci USA. 2009;106(18):7666-71.
4. Brutus A, Sicilia F, Macone A, Cervone F, De Lorenzo G. A domain swap approach reveals a role of the plant wall-associated kinase 1 (WAK1) as a receptor of oligogalacturonides. Proc Natl Acad Sci USA. 2010;107(20):9452-7.
5. Powell AL, van Kan J, ten Have A, Visser J, Greve LC, Bennett AB, et al. Transgenic expression of pear PGIP in tomato limits fungal colonization. Mol Plant-Microbe Interact. 2000;13(9):942-50.
6. Ferrari S, Vairo D, Ausubel FM, Cervone F, De Lorenzo G. Tandemly duplicated Arabidopsis genes that encode polygalacturonase-inhibiting proteins are regulated coordinately by different signal transduction pathways in response to fungal infection. Plant Cell. 2003;15(1):93-106.
7. Aguero CB, Uratsu SL, Greve C, Powell AL, Labavitch JM, Meredith CP, et al. Evaluation of tolerance to Pierce's disease and Botrytis in transgenic plants of Vitis vinifera L. expressing the pear PGIP gene. Mol Plant Pathol. 2005;6(1):43-51.
8. Manfredini C, Sicilia F, Ferrari S, Pontiggia D, Salvi G, Caprari C, et al. Polygalacturonase-inhibiting protein 2 of Phaseolus vulgaris inhibits BcPG1, a polygalacturonase of Botrytis cinerea important for pathogenicity, and protects transgenic plants from infection. Physiol Mol Plant Pathol. 2005;67(2):108-15.
9. Janni M, Sella L, Favaron F, Blechl AE, De Lorenzo G, D'Ovidio R. The expression of a bean PGIP in transgenic wheat confers increased resistance to the fungal pathogen Bipolaris sorokiniana. Mol Plant Microbe Interact. 2008;21(2):171-7.
10. Ferrari S, Sella L, Janni M, De Lorenzo G, Favaron F, D'Ovidio R. Transgenic expression of polygalacturonase-inhibiting proteins in Arabidopsis and wheat increases resistance to the flower pathogen Fusarium graminearum. Plant Biol. 2012;14 Suppl 1:31-8.
11. Haq SK, Atif SM, Khan RH. Protein proteinase inhibitor genes in combat against insects, pests, and pathogens: natural and engineered phytoprotection. Arch Biochem Biophys. 2004;431(1):145-59.
12. Kim JY, Park SC, Hwang I, Cheong H, Nah JW, Hahm KS, et al. Protease inhibitors from plants with antimicrobial activity. Int J Mol Sci. 2009;10(6):2860-72.
]]>13. Lorito M, Broadway RM, Hayes CK, Woo SL, Noviello C, Williams DL et al. Proteinase inhibitors from plants as a novel class of fungicides. Mol Plant-Microbe Interact. 1994;7(4):525-7.
14. Mosolov VV, Loginova MD, Fedurkina NV, Benken II. The biological significance of proteinase inhibitors in plants. Plant Sci Lett. 1976;7(2):77-80.
15. Ryan CA. Protease inhibitors in plants: genes for improving defenses against insects and pathogens. Annu Rev Phytopathol. 1990;28:425-49.
16. Woloshuk CP, Meulenhoff JS, Sela-Buurlage M, van den Elzen PJ, Cornelissen BJ. Pathogen-induced proteins with inhibitory activity toward Phytophthora infestans. Plant Cell. 1991;3(6):619-28.
17. Valueva TA, Revina TA, Gvozdeva EL, Gerasimova NG, Ozeretskovskaia OL. [Role of proteinase inhibitors in potato protection]. Bioorg Khim. 2003;29(5):499-504.
18. Valueva TA, Revina TA, Kladnitskaya GV, Mosolov VV. Kunitz-type proteinase inhibitors from intact and Phytophthora-infected potato tubers. FEBS Lett. 1998;426(1):131-4.
Received in July, 2014.
Accepted in December, 2014.
]]> Ingrid Hernandez-Estévez. División de Plantas. Centro de Ingeniería Genética y Biotecnología, CIGB. Ave. 31 e/ 158 y 190, Cubanacán, Playa, CP 11600, La Habana, Cuba. E-mail: ingrid.hernandez@cigb.edu.cu. ]]>