
ARTÍCULO ORIGINAL
Obtention of dry extract from aqueous extracts of Justicia pectoralis Jacq. (tilo)
Obtención de extractos secos a partir de extractos acuosos de Justicia pectoralis (tilo)
]]>
PhD. Jorge Enrique Rodríguez Chanfrau, PhD. Orestes Dario López Hernández, MSc. Yanier Núñez Figueredo, Tech. Carlos Rodríguez Ferrada, Tech. Antonio Nogueira Mendoza
Center for Drug Research and Development. Havana, Cuba.
Introduction: Justicia pectoralis Jacq. (Acanthaceae), is a medicinal species native to the American tropics. In Cuba it is used in folk medicine in the treatment of nervous disorders. The Cuban Pharmaceutical Industry is working on the development of technological processes for the obtention of pharmaceutical quality raw materials from natural products.
Objectives: develop a technological process to obtain dry extract from aqueous extracts of J. pectoralis.
Methods: a process to obtain dry extract by spray drying from aqueous extract of J. pectoralis was developed. The influence of solid-liquid r ratio and extraction time on coumarin content was evaluated by applying a response surface methodology (RSM). The drying process and later pharmacological evaluation of the dry powder were studied.
Results: results show that solid-liquid ratio was the statistically significant main effect, having a strong positive influence on coumarin extraction in our working conditions. Extraction time and interaction between both factors were not significant for regression models. On the other hand, evaluation of the influence of inert additives on drying s showed that the best results were obtained with the use of 30 % soluble starch, which did not affect sedative activity. ]]>
Conclusions: a solid-liquid ratio of 1:20 and an extraction time of 15 minutes were suggested as optimum parameters for coumarin extraction.
Key words: aqueous extract, coumarin, Justicia pectoralis Jacq., response surface methodology, pharmacological evaluation, sedative activity, spray drying.
Introducción: Justicia pectoralis Jacq. (Acanthaceae) es una especie medicinal nativa de la América tropical. En Cuba es usada en la medicina tradicional en el tratamiento de afecciones nerviosas. La industria farmacéutica cubana trabaja en el desarrollo de procesos tecnológicos para la obtención de materias primas de calidad farmacéutica a partir de productos naturales.
Objetivo: desarrollar un proceso tecnológico para la obtención de extractos secos a partir de extractos acuosos de Justicia pectoralis.
Métodos: se desarrolló un proceso para la obtención de extracto seco por spray drying a partir de extracto acuoso de Justicia pectoralis. La influencia que sobre el contenido de cumarina tenían la relación sólido-líquido y el tiempo de extracción fue estudiada aplicando un diseño superficie respuesta. Se estudió el proceso de secado y la posterior evaluación farmacológica del polvo seco.
Resultados: estos mostraron que la relación sólido-líquido tenía una fuerte influencia estadísticamente significativa sobre la extracción de la cumarina en las condiciones de trabajo experimentadas. El tiempo de extracción y la interacción entre ambos factores no fueron significativos para el modelo de regresión estudiado. Por otro lado, la evaluación de la influencia de aditivos inertes en el proceso de secado mostró que los mejores rendimientos se alcanzaban cuando se empleaba almidón soluble 30 %, y no se afectaba la actividad sedante del producto.
Conclusiones: una relación sólido-líquido de 1:20 y un tiempo de extracción de 15 min resultaron las condiciones óptimas para la extracción de cumarina.
]]>
Palabras clave: extracto acuoso, cumarina, Justicia pectoralis Jacq., metodología superficie respuesta, evaluación farmacológica, actividad sedante, spray drying.
INTRODUCTION
Illnesses related to the Central Nervous System are a health problem in the world. Natural products have been used for the treatment and control of illnesses related to this pathology.
Justicia pectoralis Jacq. (Acanthaceae), known as Tilo, is a medicinal species native to the American tropics. It is used in folk medicine for its curative properties. In Cuba it is used in the treatment of alterations of the nervous system.1 Various studies have revealed the presence of betaine, coumarin, 7 hydroxycoumarin, flavonoids (apigenin, swertisin and swertiajaponin), lignans and more than 15 amino acids.2-4
Coumarins belong to a group of compounds known as benzopyrones, which is the main constituent responsible for the aromatic fragrance of dried leaves of Justicia pectoralis.5 Pharmacological activities of coumarin, for example anti-inflammatory, antioxidant, antiallergic, hepatoprotective, antithrombotic, antiviral, sedative and anticarcinogenic activities have been described.4,6,7
Nowadays, the Cuban Pharmaceutical Industry is working on the development of technological processes for the obtention of pharmaceutical quality raw materials from natural products. Spray drying is a simple and effective method to obtain dry powder with attractive physical characteristics, excellent flow and packing properties that greatly facilitate handling and transport.8,9 ]]>
In Cuba, the established process for Justicia pectoralis extract obtention is hydroalcoholic repercolation.10 This procedure requires a lengthy extraction time (4 days) and a long ethanol evaporation time. On the other hand, drying studies carried out by Rodriguez et al, have demonstrated loss by adhesion of the product to the walls of the chamber during the drying process, resulting in poor product recovery.11A process to obtain aqueous extract of Justicia pectoralis was developed. Extraction temperature was 100 ºC. The influence of solid-liquid ratio and extraction time on coumarin content was evaluated by applying response surface methodology (RSM). The drying process and later pharmacological evaluation of the dry powder was studied.
METHODS
Plant material
The sample of Justicia pectoralis Jacq. (var. pectoralis) was collected at Dr. Juan Tomás Roig Medicinal Plants Experimental Station in Artemisa, Cuba. Herbarium specimens, voucher ROIG 4636, have been deposited in herbaria at the Experimental Station. ]]>
Establishment of the technological process
Experimental design
Response Surface Methodology (RSM) with a 32 central composed experimental design (Statgraphics plus 5.1, USA) was applied to evaluate the effects of extraction time (ET) and solid liquid ratio (SLR) on coumarin content (%). The coded independent variables used are listed in Table 1. The levels of independent parameters were based on preliminary experimental results.
Drying process
]]>
The preliminary study showed loss by adhesion of the product to the walls of the chamber during the drying process, making it necessary to employ inert additives. The inert additives used in our study were 10 DE Maltodextrin (Arancia-cpc, Mexico) and Soluble Starch (Riedel de Haën, Germany) at concentrations between 10 and 30 % versus the dry weight of liquid extract content.The carried solution was prepared by dissolving the additive in 1000 mL of aqueous extract under constant stirring before spray drying. The aqueous extracts were spray dried in a Büchi B 191 model spray dryer (Switzerland). Inlet air temperature (140 °C) and outlet air temperature (80 °C) were carried out according to the method described by Rodríguez et al.11 The product was fed into the spray dryer at room temperature 600 L/h: the rate was varied to regulate exit air temperature at the desired value. After cooling to room temperature, the powder was placed in plastic bags and stored until analysis.
Drying efficiency was assessed by determining the total powder content obtained in the process and comparing it with the theoretical powder content that can be obtained. Three batches (5 liter scale) with the best inert additive were prepared and drying efficiency was determined.
Aqueous extract and drying powder analysis
Coumarin content in the aqueous extract was determined by HPLC using an Aluspher® 100 (RP-select B 5 mm) pre-column and a Lichrospher® 100, RP 18 column (250 x 4 mm, 5 µm). The mobile phase used was methanol-water (60:40 v/v). Chromatography conditions were mobile phase flow 1 ml/min, UV detector l= 274 nm, and injection volume 20 µL.
Coumarin content in the drying powder was determined according to the method described by Rodríguez et al.13
Dry matter content, pH and organoleptic characteristics in the aqueous extract were determined according to NRSP with some modifications.14
]]>
Pharmacological studies
Our interest was to know whether the technological process developed affected the sedative activity of the product. A pharmacological evaluation (Open field activity method) was conducted of samples obtained from the inert additives study. Later, a pharmacological evaluation was carried out of samples obtained in 5 liter scale.
Animals
Male albino mice (Swiss, 1822 g) in thiopental-induced sleep, open field activity and aggressive behavior test. All animals were housed in groups of five under standard laboratory conditions of temperature, humidity and lighting (12:12-h light/dark). Animals had free access to food and water, except during the experiment. They were deprived of food but not of water 6 h before drug administration. Each group consisted of ten animals. All experiments were carried out between 8:00 am to 11:00 am in accordance with the Institutional Animal Ethical Committee which approved the study. Animal care was in conformity with Canadian Council for Animal Care guidelines.
Evaluation of the influence on pharmacological activity ]]>
In the inert additives study for the evaluation of their influence on pharmacological activity, animals were divided into four groups: Distilled water- treated group (NC), Spray dried hydroalcoholic extract (20 mg/kg)-treated group (PC) obtained according to Rodriguez et al.,11 Spray dried aqueous extract without inert additives (20 mg/kg)-treated group (TG), and Spray dried aqueous extract with inert additives (20 mg/kg)-treated group (TGIA).
Pharmacological evaluation of samples obtained to the scale of 5 liters
Animals were divided into five groups: Distilled water treated group (NC), Diazepam (1 mg/kg)-treated group (PC), and Spray dried aqueous extract with inert additives (5, 20 and 80 mg/kg)-treated groups.
Open field activity
Open field activity was carried out according to a previously reported method with some modifications.15,16 Thirty minutes after administration of vehicle or test compound a mouse was placed in the centre of a round open field 30 cm in diameter and 25 cm in height, and open field activity was measured during 6 minutes, recording how many times the animal stayed in the centre of the cage and the number of risings. ]]>
Aggressive behavior
This study was conducted according to the method described by Pinna et al.17 A group of animals were isolated in individual cages during six weeks, while another group remained together. Thirty minutes after administration of vehicle or test compound, aggressive behavior was evaluated by the introduction of an intruder mouse into the isolated mice's home cage. Aggressive activity (biting attacks and wrestling) by isolated mice was measured as total fighting time during a 20 min period.
Thiopental-induced sleep
The Thiopental-induced sleep study was conducted according to the method described by Fernandez et al.18 Thiopental Sodium (50 mg/kg) was injected intraperitoneally 30 min after administration of distilled water or test compound. An animal was placed on its back on a warmed (35 ºC) pad. The number of sleeping animals and the duration of loss of righting reflex were recorded. The time elapsed from loss of righting reflex until a mouse regained its righting reflex was measured.
Statistical analysis ]]>
Experimental design, data analysis and optimization procedure were performed using Statgraphic plus 5.1. All pharmacologically obtained results were expressed as mean ± standard deviation (SD) and assessed by analysis of variance (ANOVA) followed by Duncan's test. Results were considered significant when p< 0.05.
RESULTS
Technological studies
The dried plant material complies with the quality specifications established by the method of analysis.12
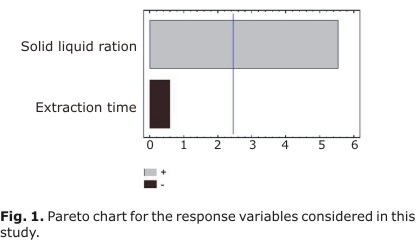
Maximum coumarin content (4.3 %) was recorded under the following experimental conditions: solid-liquid ratio 1:20 and extraction time 15 min. Analysis of variance (ANOVA) was used to evaluate the significance of coefficients in the models. Table 2 shows the regression coefficient values of equation and statistical parameters obtained from ANOVA. The p values were used to evaluate the importance of each coefficient and the interactions among the variables. To obtain a simple and yet realistic model, insignificant terms (p > 0.05) are eliminated from model through a backward elimination process. The statistical parameters obtained from the ANOVA for the reduced models are given in Table 2. Figure 1 shows the Pareto chart for the response variable.
]]>
A transparent liquid of brownish gray color and characteristic fragrance was obtained. Dry matter content in the extract was 3.27 ± 1.11 and pH was 5.41 ± 1.14.
Figure 2a shows the results of the influence of both inert additives on the drying of the aqueous extract of Justicia pectoralis. The best results were obtained with the use of the 30 % soluble starch (drying efficiency above 65 % considered appropriate for the working scale). On the other hand, no loss by adhesion of the product to the walls of the chamber during the drying process was observed, which favors product recovery.
A fine and amorphous powder of brown color was obtained. Coumarin content was more than 7.0 mg/g of dried powder. These results are similar to those reported by Rodriguez et al.18 in studies of drying of hydroalcoholic extracts of Justicia pectoralis by spray drying. Results of analysis of batches with 30 % soluble starch and an exit air temperature of 80 ºC were that those conditions were optimal to obtain dry powder from aqueous extract of Justicia pectoralis with a recovery greater than 70 %, considered appropriate for the working scale.
Pharmacological studies ]]>
Evaluation of the influence of inert additives on pharmacological activity (Open field activity method) showed no significant differences (p< 0.05) between the spray dried hydroalcoholic extract treated group (PC), the spray dried aqueous extract without inert additives treated group, and the spray dried aqueous extract with 30 % soluble starch treated group (Fig. 2b).In the pharmacological evaluation of samples obtained to the scale of 5 liters, the open field activity study (Fig. 3a) found that dried extract (5, 20 and 80 mg/kg, po.) reduced spontaneous locomotors activity and rearing in a dose-dependent manner during the observation period. Figure 3b shows the effects of test compound on aggressive behavior in isolated mice. Test compound reduces aggressive behavior in a dose-dependent manner.
The time of appearance of induced sleep in animals treated with the different doses of dry extract was significantly shorter (p< 0.05) than that obtained f in the negative control group (Fig. 3c). All test compound doses increased the number of sleeping animals compared with the control. Doses of 20 and 80 mg/kg caused sleep in all animals. Test compound 80 mg/kg and diazepam 1 mg/kg prolonged thiopental-induced sleep in a similar manner (Fig. 3d).
DISCUSSION
]]>
Experimental design
The process of optimization by RSM is advantageous because this method saves time, space and raw materials. Application of this method allowed us to study the main and possible effects of extraction time and solid-liquid ratio on coumarin content.
Results show that the different indicators of the process did not exhibit the same pattern. Solid-liquid ratio showed the statistically significant main effect, having a strong positive influence on coumarin extraction in our working conditions. Extraction time and the interaction between the two factors were not significant for regression models.
With respect to numerical optimization, a solid-liquid ratio of 1:20 and an extraction time of 15 minutes were suggested as optimum parameters for coumarin extraction from J. pectoralis. Suitability of the model equation for predicting optimum response values was tested under theses conditions and was validated experimentally. A mean value of 4.32 % of coumarin, obtained from real experiments (4.35 % value predicted), demonstrated the validity of the RSM model, indicating that the model was adequate for the extraction process.
Evaluation of the influence of inert additives on drying
The presence of water in solid-state systems has a significant impact on the stability of drugs not only by causing hydrolysis, but also because it affects other properties such as plasticity and ductility, film forming ability, and stickiness.19 ]]>
Spray drying is a simple and effective method to obtain dry powder. The presence of some amorphous compoundsThe inert additives used in our studies have individual indexes of drying greater than 1 and a high Tg value (Tg: 160 and IID: 1.6 for 10 DE Maltodextrin, Tg: 243 and IID: 2.4 for Soluble Starch), appropriate to improve the drying process. However, the glass transition temperature (Tg) of the aqueous extract of Justicia pectoralis was 59.6 ºC and the sticking temperature (Ta) was 79.7 ºC. Additionally, it was estimated that the glass transition temperature and the sticking temperature of the aqueous extract of Justicia pectoralis with soluble starch inert additive were 72.1 ºC and 92.1 ºC, respectively.21 This result is important because the fixed exit air temperature (80 °C) in the technological process, which is smaller than the sticking temperature of the aqueous extract/inert additive mixture (92.1 ºC), prevents sticking during the drying process, increasing product recovery. Results show that the technological process developed was suitable to obtain a pharmaceutical quality raw material.
Pharmacological studies
Results on the influence of inert additives on pharmacological activity suggest that the addition of 30 % soluble starch does not affect the sedative activity of J. pectoralis.
Pharmacological evaluation of samples obtained to the scale of 5 liters
The open field activity study of samples obtained to the scale of 5 L showed that dried extract reduced spontaneous locomotors activity and rearing in a dose-dependent manner during the observation period. Rodents usually show exploratory behavior when they are placed in a new environment. However, if the animals are pre-treated with depressant drugs, locomotive activity is decreased. Doses of 80 mg/kg of test compound showed similar behavior to diazepam 1 mg/kg (standard anxiolytic drugs). This result is typical of sedative drugs. ]]>
Dried extract obtained from aqueous extract of J. pectoralis as well as diazepam, a standard reference drug, increased the number of sleeping animals and prolonged thiopental-induced sleep time in mice.
Social isolation induces aggressive behavior in several strains of mice. Isolation-induced aggression is considered to be useful as an animal model for assessing inhibitory activity of the central nervous system. Different neurotransmitters such as serotonin, noradrenalin, dopamine and gamma aminobutyric acid (GABA) are considered to be involved in mediating aggressive behavior, with conflicting results on brain neurotransmitter metabolism.23,24 In a recent study (not published) we were unable to demonstrate anxiolytic-like activity in the test compound using the elevated plus-maze test in mice. Therefore, the sedative effect of the powder extract obtained from aqueous extracts of Justicia pectoralis Jacq. is not mediated via the GABA or Glycine systems.
Our results show anti-aggressive behavior in powder extract-treated mice. This result can be mediated by inhibitory effects on brain biogenic amines action or excitatory neurotransmitter release, and suggests an inhibitory effect on the central nervous system. This suggestion was strengthened by the increase in sleeping time in the test-compound treated groups over that in the control group in the thiopental-induced sleep model.
In summary, it was found that solid-liquid ratio was the most significant factor affecting coumarin extraction. On the other hand, evaluation of the influence of inert additives on drying shows that the best results were obtained with the use of 30 % soluble starch, not affecting the sedative activity of J. pectoralis. Results show that the technological process developed was suitable to obtain a pharmaceutical quality raw material from aqueous extract of J. pectoralis by spray drying.
REFERENCES
]]>
1. Rivero Martinez R, Rodríguez Chanfrau J. E. Justicia pectoralis Jacq. Some aspects about the chemical composition, pharmacology and toxicology (Review article). Rev Mex Ciencias Farmacéuticas. 2000;30(2):15-9
2. Méndez Arteaga J, Murillo Perea E, Yara Varón E, Suescún Ospina W, Niño Osorio J, Mosquera Martínez O. Climate influence on chemical composition and antioxidant activity of Justicia pectoralis Jacq. Rev Cubana Farm. 2011;45(1):88-100
3. La Serna B, Amor A, Martínez M. Preliminary chemical characterization of Justicia pectoralis. Rev Cubana Farm. 1989;23(3):256-64
4. Lino C, Traveira M, Viana G, Matos J. Analgesic and antiinflammatory activities of Justicia pectoralis Jacq. and its main constituents: coumarin and umbelliferone. Phytoter Res. 1997;11(3):211-5
5. O'Kennedy R, Douglas R. Coumarins. Biology, applications and mode of action. England: John Wiley and Sons Ltd.; 1997. p. 22-66.
6. Ritschel W, Hardt T. Pharmacokinetics of coumarin, 7 -hydroxycoumarin and -hydroxycoumarin glucuronide in the blood and brain of gerbils following intraperitoneal administration of coumarin. Drug Res. 1983;33:1254-8
7. Mac Rae W, Towers G. Justicia pectoralis: a study of the basis for its use as a hallucinogenic snuff ingredient. J Ethnopharmacol. 1984;12:93-111
8. Hickey A, Gunderton D. Pharmaceutical Process Engineering. New York: Marcel Dekker Inc.; 2001. p. 86-109. ]]>
9. Swami Handa S, Singh Khanuja S, Longo G, Rakesh D. Extraction technologies for medicinal and aromatic plants. Trieste: International Centre for Science and High Technology; 2008. p. 30-1.
10. NRSP 311. Fluid extracts. Technological processes. La Habana: Ministry of Health Publishers; 1992.
11. Rodríguez Chanfrau JE, López Hernández OD, Núñez Y, Rodríguez C, Carrillo C, Gil Apan J, et al. Obtention of pharmaceutical quality raw material from Justicia pectoralis Jacq. extracts by spray drying. Technology Development from a 30% hydroalcoholic extract. Latin American J Pharmacy. 2008;27(3):333-8.
12. NRSP 309. Vegetable Material. Assays methods. La Habana: Ministry of Health Publishers; 1992.
13. Rodríguez Chanfrau JE, López Hernández OD, Gil Apan JM. Método para la cuantificación de cumarina en extracto seco a partir de extractos de Justicia pectoralis Jacq. Rev Cubana Plant Med [revista en la Internet]. 2008 Sep [citado Abr 26 2011]; 13(3). Disponible en: http://scielo.sld.cu/scielo.php?script=sci_arttext&pid=S1028- 47962008000300004&lng=es
14. NRSP 315. Medications of Vegetable Origin. Fluid extracts of Justicia pectoralis Jacq. La Habana: Ministry of Health Publishers; 1992.
15. Archer J. Sex differences in the emotional behavior of laboratory mice. Br J Psychol. 1977;68(1):125-31.
16. Walsh RN, Cummins RA. The Open-Field Test: a critical review. Psychol Bull. 1976;83(3):482-504.
17. Pinna G, Agis-Balboa R, Pibiri F, Nelson M, Guidotti A, Costa E. Neurosteroid biosynthesis regulates sexually dimorphic fear and aggressive behavior in mice. Neurochem Res. 2008;33(10):1990-2007.
18. Fernández S, Wasowski C, Paladini A, Marder M. Synergistic interaction between hesperidin, a natural flavonoid, and diazepam. Eur J Pharmacol. 2005;512(2-3):189-98.
19. Brady J, Dürig T, Shang S. Developing solid oral dosage forms: pharmaceutical theory and practice. New York: Elservier Inc.; 2009. p. 187-217.
20. Roos Y, Karel M. Water and molecular weight effects on glass transitions in amorphous carbohydrates and carbohydrate solutions. J Food Science. 1991;56(1):1676-81.
21. López Hernández OD, Martinez Álvarez L. Design tools for vegetable extract spray drying processes for pharmaceutical use. Rev Cubana Plant Med. 2010;15(3):152-6.
22. Valzelli L. The "isolation syndrome" in mice. Psychopharmacology. 1973;31:305-20.
23. Matsuda T, Sakaue M, Ago Y, Sakamoto Y, Koyama Y, Baba A. Functional alteration of brain dopaminergic system in isolated aggressive mice. Japan J Neuropsychopharmacol. 2001;21:71-6.
24. Sakaue M, Ago Y, Murakami C, Sowa C, Sakamoto Y, Koyama Y, et al. Involvement of benzodiazepine binding sites in an antiaggressive effect by 5-HT1A receptor activation in isolated mice. European J Pharmacology. 2001;432:163-6.
Recibido: 20 febrero de 2013.
Aprobado: 30 de junio de 2013.
Jorge Enrique Rodríguez Chanfrau. Centro de Investigaciones y Desarrollo de Medicamentos. Ave 26 No. 1605. Nuevo Vedado. La Habana. CP 10600. Cuba. E-mail: jorge.rodriguez@infomed.sld.cu ]]>