My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Revista Cubana de Medicina Tropical
On-line version ISSN 1561-3054
Rev Cubana Med Trop vol.70 no.1 Ciudad de la Habana Jan.-Apr. 2018
ARTÍCULO ORIGINAL
In vitro inactivation of pathogenic bacteria by the use of ozone in different exposure times
Inactivación in vitro de bacterias patógenas mediante el uso de ozono en diferentes tiempos de exposición
Dora Inés Kozusny-Andreani,I Giovanna Andreani,I Luiz Fernando Avezum do Prado,I Amanda Oliva Spaziani,I Rodolpho César Oliveira Mellem Kairala,I Felipe Seixas Da Silva,I Renato Amaro ZangaroII
I Universidade Brasil. Fernandópolis - São Paulo, Brazil.
II Instituto de Engenharia Biomédica. São José dos Campos, São Paulo, Brazil.
ABSTRACT
Intrdoduction: in the area of health, ozone has many therapeutic properties. Several pathologies can be treated with ozone therapy, such as infectious, acute and chronic diseases caused by viruses, bacteria, fungi and parasites, autoimmune diseases, diseases with chronic ischemia, lung diseases, neuropathies, dermatological diseases, dental caries, among others.
Objective: to evaluate the effect of ozone applied in vitro in the following strains: Escherichia coli CCCD E003, Salmonella enterica subsp. enterica serovar Typhi CCCD S009, Staphylococcus aureus CCCD S003, Pseudomonas aeruginosa CCCD P013, Streptococcus mutans ATCC 25175 and Enterococcus faecalis ATCC 18211. For this purpose use was made of different cell concentrations and different times of exposure to ozone.
Methods: we used concentrations of 1 x 102, 1 x 103, 1 x 10 4, 1 x 105, 1 x 106, 1 x 107, 1 x 108 and 1 x 109 CFU/mL of NaCl (0.5 % w/v) exposed to ozone for different time intervals (30, 60, 90, 120, 150, 180, 210, 240, 270, 300, 330, 360, 390, 420, 450, 480, 510 and 540 s). Bacterial viability was determined by CFU and the colorimetric method with 2,3,5-Triphenyltetrazolium Chloride.
Results: it was found that the species S. aureus, E. coli, S. typhi, S. mutans and E. faecalis were sensitive to ozone, showing a decrease of 45-80 % of viable cells after 30 s of ozone exposure relative to the initial population, whereas P. aeruginosa was reduced 25 % compared to the initial population. The viability of bacteria exposed to ozone was dependent on the cell concentration and time exposure.
Conclusions: ozone had a bactericidal effect on the bacteria used in this study and that this effect was proportional to the concentration of bacterial cells and the time of exposure to O3. The results show significant efficacy of ozone to control populations of pathogenic bacteria, providing relevant information for its use in different areas, but always taking into account the microorganism involved.
Keywords: ozone; bactericidal activity; cell viability; in vitro.
RESUMEN
Introducción: el ozono tiene muchas aplicaciones terapéuticas en la esfera de la salud. Algunas patologías pueden tratarse con ozonoterapia, entre ellas enfermedades infecciosas, agudas y crónicas causadas por virus, bacterias, hongos o parásitos, enfermedades autoinmunitarias, enfermedades con isquemia crónica, enfermedades pulmonares, neuropatías, enfermedades dermatológicas y caries dentales, entre otras.
Objetivo: evaluar el efecto del ozono aplicado in vitro sobre las siguientes cepas: Escherichia coli CCCD E003, Salmonella enterica subesp. enterica serovar Typhi CCCD S009, Staphylococcus aureus CCCD S003, Pseudomonas aeruginosa CCCD P013, Streptococcus mutans ATCC 25175 y Enterococcus faecalis ATCC 18211. Con este propósito se utilizaron diferentes concentraciones celulares y diferentes tiempos de exposición al ozono.
Métodos: utilizamos concentraciones de 1 x 102, 1 x 103, 1 x 104, 1 x 105, 1 x 106, 1 x 107, 1 x 108 y 1 x 109 UFC/mL de NaCl (0,5 % m/v) expuestas a ozono durante diferentes intervalos de tiempo (30, 60, 90, 120, 150, 180, 210, 240, 270, 300, 330, 360, 390, 420, 450, 480, 510 y 540 s). La viabilidad bacteriana se determinó mediante UFC y el método colorimétrico con cloruro de 2,3,5-trifeniltetrazolio.
Resultados: se observó que las especies S. aureus, E. coli, S. typhi, S. mutans y E. faecalis eran sensibles al ozono, mostrando una disminución de 45-80 % de las células viables luego de una exposición de 30 s al ozono en comparación con la población inicial, mientras que la especie P. aeruginosa se redujo en un 25 % en comparación con la población inicial. La viabilidad de las bacterias expuestas al ozono dependió tanto de la concentración celular como del tiempo de exposición.
Conclusiones: el ozono mostró tener un efecto bactericida sobre las bacterias utilizadas en el estudio, y ese efecto fue proporcional tanto a la concentración de las células bacterianas como al tiempo de exposición al O3. Los resultados demuestran la significativa eficacia del ozono para controlar poblaciones de bacterias patógenas, y ofrecen información relevante con vistas a su uso en diferentes áreas, pero siempre teniendo en cuenta el microorganismo en cuestión.
Palabras clave: ozono; actividad bactericida; viabilidad celular; in vitro.
INTRODUCTIÓN
Ozone is a powerful oxidant much used in Europe and the United States, but little used in Brazil.1 He became notorious in recent decades mainly due to its highly oxidative activity that characterizes it as a potentially biocidal agent to act on bacteria, fungi, viruses and helminthes.2-5 The primary action of ozone on microorganisms occurs on the cell wall, resulting from oxidation of glycopeptides, glycoproteins and amino acids, changing the permeability and causing its rapid lysis.5-7 By penetrating inside the cell, ozone recombines with elements promoting the oxidation of cytoplasmic amino acids and nucleic acids, resulting in cleavage with consequent cell death.6,8-13
The oxidation and inactivation of bacteria by ozone are very fast, reaching different cellular constituents of non-specific manner. It can act on the purine and pyrimidine bases of nucleic acids of Escherichia coli.14 However, the sensitivity of bacteria to ozone is dependent on certain factors such as stage of growth and cell concentration, types of microorganisms, culture media, temperature, exposure time and ozone concentration, among others.5,10
Ozone has been used effectively in the food industry,12-14 water treatment,11,15 sewage treatment,16,17 persistent drugs,18,19 environment deodorization,20 machine hemodialysis disinfection,21 dental caries,22 and in several health human and animal treatment.23-26
The use of ozone in the elimination of pathogenic microorganisms in different fields requires the development and implementation of controlled studies to determine specific dosages aiming to improve the techniques employed and the results obtained. The aim of this study was to evaluate the effectiveness of ozone in the inactivation of Escherichia coli, Salmonella typhi, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus mutans and Enterocococcus faecalis, using in vitro model. For this purpose were used different cell concentrations and different times of exposure to ozone.
METHODS
BACTERIAL STRAINS AND CULTURE MEDIA
Was used standard strains of Escherichia coli CCCD E003, Salmonella enterica subsp. enterica serovar Typhi CCCD S009, Staphylococcus aureus CCCD S003, Pseudomonas aeruginosa CCCD P013 (CCCD-Collection of cultures CEFAR Diagnostic, Brazil), Streptococcus mutans ATCC 25175 and Enterococcus faecalis ATCC 18211 (American Type Culture Collection, Bioscan). The culture media used were Tryptic Soy Agar (TSA, Oxoid®, Cambridge, CB5 8BZ, UK) and tryptic soy broth (TSB, Oxoid®, Cambridge, CB5 8BZ, UK)
OZONE GENERATION
Ozone was produced by a reactor operating with corona effect (Ozone & Life), fed by pure oxygen. Ozone was generated with the constant flow concentration 4 mg/L. Ozonization of bacterial suspensions (1 000 mL) was performed in controlled temperature of 25 °C, using a gas diffuser connected to the reactor via silicon tube.27
BACTERIAL VIABILITY
The bacterial strains were grown in tryptic soy agar (TSA, Oxoid®) incubated at 37 °C for 24 h. A colony of each species was inoculated into 1000 mL of tryptic soy broth (TSB, Oxoid®) and incubated at 37 °C for 24 h. The initial bacterial density was determined by absorbance at 550 nm using McFarland standard (BioMerieux, Marcy-l'Etiole, France) which corresponds to a concentration of 1.0 x 108 CFU / mL. For the ozone treatment, the cell density of cultures were adjusted to concentrations of 1 x 10 2, 1 x1 03, 1 x 104, 1 x 105, 1 x 106, 1 x 107, 1 x 108 and 1 x 109 CFU/mL solution of NaCl (0.5 % w/v). As a control (no ozone treatment) was used a sample of each bacterial species at a concentration of 1 x 106 CFU/mL. The samples of 0.1 mL, untreated and treated with ozone were collected at different periods of time (0, 30, 60, 90, 120, 150, 180, 210, 240, 270, 300, 330, 360, 390, 420, 450, 480, 510 and 540 s),28 and inoculated into tryptic soy agar and incubated at 37 °C for 24-48 h and then colonies were counted. In those same periods of time, in order to verify the inhibitory efficiency of ozone were withdrawn 1mL from the sample, aiming to confirm the presence of viable microorganisms. Each sample was added to 50 mL of the dye (2.3,5 - Triphenyltetrazolium Chloride (TTC , Merck KGaA , Darmstadt , Germany), to show the activity of the enzyme dehydrogenase, involved in the breathing process. Hydrogenation of TTC in living cells produces the triphenyl formazan, which is a red substance, stable and non- diffusible. It became possible to distinguish the live samples stained in red, from the inactive samples that remains in the same color.29
The experiment was repeated four times and then obtained the average CFU/mL and percentage of cell viability for each time interval.
STATISTICAL ANALYSIS
The results were expressed as the means ± SD of three independent measurements for each experiment. The statistical evaluations were performed using the statistic- al software SPSS ver. 10. Significance was defined as a P value < 0.05.
RESULTS
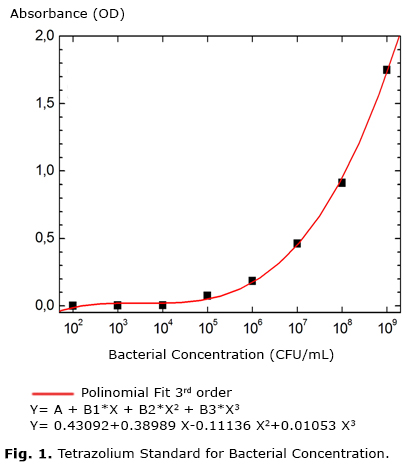
The bacterial viability was assessed by the methods of counting colonies on plates and color changing using the dye 2.3.5 triphenyl tetrazolium chloride (TTC). It was found that both methods were effective in determining the effect of ozone in bacteries, differing between them the period of time to obtain the results. Using the TTC we can get results in 10 min to Staphylococcus aureus CCCD S003, Streptococcus mutans ATCC 25175 and Enterococcus faecalis ATCC 18211. The reaction occurred in 20 min when evaluatedEscherichia coli CCCD E003, Salmonella typhi CCCD S009 and Pseudomonas aeruginosa CCCD P013. The CFU counts occurred 24 h after inoculation on tryptic soy agar and incubated at 37 °C. The results obtained from the same cell bacterial dilution and periode of ozonization were analyzed by the two methodologies and proved be consistent among them, indicating that the no bacteries growth in Petri dishes was accompanied by no color change of the medium contained in tubes. The standard optical density for the TTC was obtained from different concentrations of bacteria (Fig. 1), showing an exponential increase in optical density as a function of increasing concentration of bacteria, fitted by a third order polynomial. It was observed that different bacteria showed no significant change in this pattern. TTC dye has determined qualitatively (living cells/dead cells) cell viability after treatment with ozone in a short period of time.In 10 m was possible to obtain effective results of ozonation on E. coli, S. aureus, S. mutans, P. aeruginosa and in 20 min on S. typhi and E. faecalis.
The counts of CFU provided quantitative data on the viable cells relative to the initial number of each dilution, but was required 24 h to obtain the results. Both methods are effective for evaluation of the ozone effect on bacteria and the use of each one depends on the purpose of the study.
Inactivation of different bacterial species after action of ozone showed dependence on cell concentration and exposure time as shown in figure 2. The results showed that the strains of Staphylococcus aureus CCCD S003, Escherichia coli E003 CCCD, Streptococcus mutans ATCC 25175 and Enterococcus faecalis ATCC 18211 were sensitive to the effect of ozone like shows the figures 2A, 2B, 2E e 2F, indicating that inactivation cell varied according to the initial bacterial concentration. It took 60 s exposure to ozone at a concentration of 10 2 CFU/mL for S. aureus and E. faecalis were completely inactivated. For the same concentration, E. coli and S. mutans demanded 90 and 120 s respectively for total inactivation. With increasing bacteria concentration of the samples, ozone exposure time required for complete inactivation was also increased. Using 109 CFU/mL, 390 s were required for complete inactivation of S. mutans and S. aureus, and 450 s for the complete inactivation of E. coli and E. faecalis.
The species Salmonella typhi CCCD S009 (Fig. 2, C) and Pseudomonas aeruginosa CCCD P013 (Fig. 2, D), showed a relatively lower response to ozone, requiring longer periods of exposure to ozone to inactivate completely their species. Was required 510 s to completely inactivate the S. typhi concentration of 108 UFC/mL, whereas concentration of 109 CFU/mL remained still 15 % viable bacteria as shown in figure 2, C.
Inactivation of 102 CFU/mL of P. aeruginosa occurred after 270 s of exposure to ozone, and demanded 510s to inactivate completely 106 CFU/mL. After exposing to ozone concentrations of 107, 108 and 109 for 540 s respectively remain viable, 6, 20 and 30 % of cells (Fig.2, D)
For bacterial concentrations ranging from 102-109 CFU/mL and exposure to ozone by 510 s at 4 mg/L, allows completely inactivate species (2A) Staphylococcus aureus CCCD S003, (2B) Escherichia coli CCCD E003, (2E) Sptreptococcus mutans ATCC 25175 and (2F) Enterococcus faecalis ATCC 18211.
DISCUSSIÓN
The bacteria used in this study are generally micro-organisms involved in infectious diseases or as contaminants of medical equipment and hospital environments. In addition to the involvement of these microorganisms in infectious diseases in recent years been isolated multiresistant bacteria to different antibiotics, making complicated the treatment of infected persons.30,31 These problems led to numerous studies in the search for alternative treatments for the control of bacterial infections. This study evaluated the effect of ozonization on S. aureus, E. coli, S. typhi, P. aeruginosa, S. mutans and E. faecalis, checking cell viability at different periods of time, using the colorimetric tetrazolium (TTC) and CFU counts on plates. The results obtained by the colorimetric tetrazolium to verify the viability of bacterial species evaluated were confirmed by the counts, showing the efficiency of the method. The tetrazolium colorimetric test as described by Sylvester29 is an established method for determining the number of viable cells in proliferation and cytotoxicity studies. The assay provides a rapid, convenient, economical and thus has become a very popular technique for qualify viable cells in culture, providing reliable data.
Ozone is a powerful biocidal agent, capable of inactivating innumerable micro-organisms including Gram-negative and Gram-positive sporulated forms vegetative cells, spores, fungal or viral capsids, severely reducing the populations in a short exposure time.2-6 The reduction or inactivation of microbial population depends on ozone concentration, time of application and the micro-organisms involved.3,6,21
The results show that ozone inactivation of the bacterial species studied depends on the species and their concentration and exposure time to the gas (Fig. 2, A, B, C, D, E, F), showing to be effective for both, Gram-positive and Gram-negative species. there were significant differences between the bacterial strains and exposure times (p< 0.05). Similar results were obtained by Thanomsub28 evaluating the Gram-negative (E. coli and Salmonella) and gram positive bacteria (S. aureus and Bacillus subtilis). These authors found that the effectiveness of ozone depends on the bacterial concentration and exposure time, and when evaluated by scanning electron microscopy observed damage and deformity in the wall of the bacteria treated with ozone, and no change was observed in the structures of control samples without exposure to the gas. Prabakaran32 also show that ozone was effective for inactivating E. coli, S. typhi, and Pseudomonas fluorescens.Cells of S. aureus, E. coli, S. typhi, S. mutans and E. faecalis at a concentration of 1x10 2 UFC/ml were highly sensitive to ozone, showing a decrease of viable cells ranging from 45 to 80 % (Fig. 2, A, B, C, D, E, F) for the first 30 min of exposure to the gas, whereas P. aeruginosa in the same condition decreased by only 25 % of the initial population (Fig. 2, D). The results obtained for P. aeruginosa is probably related to the characteristics of bacterial species that has the ability to survive in poor nutrient and grow in water with low solids and dissolved organic compounds.33
Studies have shown variation in the sensitivity of the different microorganisms when exposed to ozone, and the temperature variation can be responsible for this effect, because the increase of temperature reduces the stability and solubility of the gas.8,30,34 In order to ensure reproducibility of the results, this study was conducted in an environment with controlled temperature at 25 °C. The culture medium was standardized for all bacteria, which were subjected to the same exposure time and concentration of ozone.
The results showed that ozone was effective in reducing the population of Staphylococcus aureus CCCD S003, Escherichia coli CCCD E003, Salmonella typhi CCCD S009, Pseudomonas aeruginosa CCCD P013, Sptreptococcus mutans ATCC 25175, Enterococcus faecalis ATCC 18211, leading to the conclusion that ozone had a bactericidal effect on the bacteria and that this effect was proportional to the concentration of bacterial cells and the time of exposure to O3.
BIBLIOGRAPHIC REFERENCES
1. Souza JB, Daniel LA. Inactivation of indicator microorganisms Escherichia coli, Clostridium perfringens and coliphages using ozone. Ambiência - Rev Setor Ciên Agr Ambient. 2008 [citado 2 Feb 2016];4:265-73. Disponible en: http://www.revistas.unicentro.br/index.php/ambiencia/issue/archive
2. Oliveira AF, Mendes HJ. Clinical applications of ozone in dentistry. Rev Saúde Com. 2009 [citado 12 Feb 2016];5:128-40. Disponible en: http://www.uesb.br/revista/rsc/v5/v5n2a06.pdf
3. Martins CC, Kozusny-Andreani DI, Mendes ECB. Ozônio no controle de micro-organismos em resíduos de serviços de saúde. Rev Baiana Enferm. 2015;29(4):318-29. doi.org/10.18471/rbe.v29i4.13678
4. Bocci V, Zanardi I, Travagli V. Ozonization of human HIV-infected plasmas for producing a global vaccine: How HIV-patients may help fight the HIV pandemia. Virulence. 2010;1:215-7. doi.org/10.4161/viru.1.3.11830
5. Chuajedton A, Aoyagi H, Uthaibutra J, Pengphol S, Whangchai K. Inactivation of Escherichia coli O157:H7 by treatment with different temperatures of micro-bubbles ozone containing water. Int Food Res J. 2017 [citado 2 Feb 2016];24(3):1006-10. Disponible en: http://www.ifrj.upm.edu.my
6. Zhang YQ, Wu QP, Zhang JM, Yang XH. Effects of ozone on membrane permeability and ultrastructure in Pseudomonas aeruginosa. J Appl Microbiol. 2011;111:1006-15. doi: 10.1111/j.1365-2672.2011.05113.x
7. Silva SB, Luvielmo MM, Geyer MC, Prá I. Potential use of ozone in the food processing. Semina: Ciênc Agr. 2011;32:659-82. doi.org/10.54333/1679-0359.2011v32n2p659
8. Kumar A, Bhagawati S, Tyagi P, Kumar P. Current interpretations and rationale of the ozone usage in dentistry: a systemic review of literature. Eur J Gen Dent. 2014;3(3):175-80. doi: 10.4103/2278-9626.141658
9. Alwi NA, Ali A. In vitro and microscopic analysis of ozone fumigation effects on Salmonella typhimurium and Listeria monocytogenes. Acta Hortic. 2013;1012:1247-54. doi: 10.17660/ActaHortic.2013.1012.168
10. Tordiglione P, Araimo Morselli FSM, Scarpa I, Puggioni G, Mancini I, Rosa G, et al. In Vitro Evaluation of Ozone Activity on Recent Clinically Isolated Bacterial Strains. Adv Microbiol. 2014;4:106-15. doi:10.4236/aim.2014.42015
11. Lapolli FR, Santos LF, Hassemer MEN, Aisse MM, Pivelli RP. Wastewater disinfection by ozonation. In: Gonçalves RF, ed. Disinfection of wastewater, removal of harmful organisms and pathogens: application for productive purposes such as agriculture, aquaculture and hydroponics. 2003. Vitória: PROSAB; p. 169-208.
12. Christ D, Savi GD, Scussel VM. Effectiveness of Ozone Gas Application Methods against Combined Multi-Contaminants in Food. Food Public Health. 2017;7(3):51-58. doi: 10.5923/j.fph.20170703.01
13. Trindade MA, Kushida MM, Montes Villanueva ND, dos Santos Pereira DU, de Oliveira AE. Comparison of ozone and chlorine in low concentrations as sanitizing agents of chicken carcasses in the water immersion chiller. J Food Prot. 2012;75:1139-43. doi: 10.4315/0362-028X.JFP-11-288
14. Brodowska AJ, Śmigielski K, Nowak A, Czyżowska A, Otlewska A. The Impact of Ozone Treatment in Dynamic Bed Parameters on Changes in Biologically Active Substances of Juniper Berries. PLoS ONE. 2015;10(12):e0144855. doi:10.1371/journal.pone.0144855
15. Pisarenko AN, Stanford BDD, Gerrity D, Snyder SA. Effects of ozone and ozone/peroxide on trace organic contaminants and NDMA in drinking water and water reuse applications. Water Res. 2012;46:316-26. doi: 10.1016/j.watres.2011.10.021
16. Rosenblum J, Ge C, Bohrerova Z, Yousef A, Lee J. Ozonation as a clean technology for fresh produce industry and environment: sanitizer efficiency and wastewater quality. J Appl Microbiol. 2012;113:837-45. doi: 10.1111/j.1365-2672.2012.05393.x
17. Wu D, Yang Z, Wang W, Tian G, Xu S, Sims A. Ozonation as an advanced oxidant in treatment of bamboo industry wastewater. Chemosphere. 2012;88:1108-13. doi: 10.1016/j.chemosphere.2012.05.011
18. Almeida E, Assalin MR, Rosa MA, Durán N. Tratamento de efluentes industriais por processos oxidativos na presença de ozônio. Quím. Nova. 2004;27:818-24. doi.org/10.1590/S0100-40422004000500023
19. Fedorova G, Grabic R, Nyhlen J, Järhult JD, Söderström H. Fate of three anti-influenza drugs during ozonation of wastewater effluents - degradation and formation of transformation products. Chemosphere. 2016;150:723-30. doi: 10.1016/j.chemosphere.2015.12.051.
20. Domeño C, Rodríguez-Lafuente A, Martos JM, Bilbao R, Nerín C. VOC removal and deodorization of effluent gases from na industrial plant by photo-oxidation, chemical oxidation and ozonization. Environ Sci Technol. 2010;1(44):2585-91. doi: 10.1021/es902735g
21. Canada MLM, Abelan US, Zangaro RA, Kozusny-Andreani DI, Yamazaki RFC. Effectiveness of ozonated water in the reprocessing of blood dialyzers. Braz J Biom Eng. 2014;30(3):215-19. doi: doi.org/10.1590/rbeb.2014.021
22. Burke FJ. Ozone and caries: a review of the literature. Dent Update. 2012 [citado 22 Abr 2016];39:271-78. Disponible en: https://www.ncbi.nlm.nih.gov/pubmed/22774691
23. Calunga JL, Paz Y, Menéndez S, Martínez A, Hernández A. Rectal ozone therapy for patients with pulmonary emphysema. Rev Med Chil. 2011; 139:439-47. doi: /S0034-98872011000400004
24. Wainstein J, Feldbrin Z, Boaz M, Harman-Boehm I. Efficacy of ozone-oxygen therapy for the treatment of diabetic foot ulcers. Diabet Technol therapeut, 2011;13:1255-60. doi: 10.1089/dia.2011.0018
25. Martínez-Sanches G, Delgado-Roche L, Díaz-Batista A, Pérez-Davison G, Re L. Effects of ozone therapy on haemostatic and oxidative stress index in coronary artery disease. Eur J Pharmacol. 2012;691:165-72. doi: 10.1016/j.ejphar.2012.07.010
26. Tiwari S, Avinash A, Katiyar S, Aarthi Iyer A, Jain S. Dental applications of ozone therapy: A review of literature. Saudi J Dent Res. 2017;8(1-2):105-11. http://dx.doi.org/10.1016/j.sjdr.2016.06.005
27. Tormin SC, Navarini A, Almeida JCOF, Travassos LHR, Negri MVG, Silva RA. Análise do efeito bactericida do ozônio sobre bactérias multirresistentes. Arq Med Hosp Fac Cienc Med Santa Casa São Paulo, 2016 [citado 2 Feb 2016];61:138-4. Disponible en: http://www.fcmsantacasasp.edu.br
28. Thanomsub B, Anupunppisit V, Chanphetch S, Watcharachaipong T, Poonkhum R, Srisukonth C. Effects of ozone on cell gowth and structurall changes in bacteria. J Gen Appl Microbiol. 2002 [citado 22 Abr 2016];48:193-9. Disponible en: https://www.ncbi.nlm.nih.gov/pubmed/12469318
29. Sylvester PW. Optimization of the tetrazolium dye (MTT) colorimetric assay for cellular growth and viability. Meth Mol Biol. 2011;716:157-68. doi: 10.1007/978-1-61779-012-6_9
30. Ferreira H, Lala ERP. Pseudomonas aeruginosa: An alert to the professionals of health. Rev Panam Infectol. 2010 [citado 2 Feb 2016];12:44-50. Disponible en: http:// www.revistaapi.com/wp-content/uploads/2014/03/API_02_10_G.pdf
31. Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions of acquired resistance. Clin Microbiol Infect. 2012;18:268-81. doi: 10.1111/j.1469-0691.2011.03570.x
32. Prabakaran M, Tamil Selvi S, Merinal S, Panneerselvam A. Effect of ozonation on pathogenic bacteria. Adv Appl Sci Res. 2012 [citado 2 Feb 2016];3:299-302. Disponible en: http://www.pelagiaresearchlibrary.com
33. Guerra NMM, Otenio MH, Silva MEZ, Guilhermetti M, Nakamura CV, Ueda-Nakamura T, et al. Occurrence of Pseudomonas aeruginosa in drinking water. Acta Sci Biol Sci. 2008;28:13-8. doi: 10.4025/actascibiolsci.v28i1.1053
34. Nogales CG, Ferreira MB. Lage-Marques JL, Antoniazzi JH. Comparison of the antimicrobial activity of three different concentrations of aqueous ozone on Pseudomonas aeruginosa, Staphylococcus aureus, and Enterococcus faecalis - in vitro study. Rev Esp Ozonoterapia. 2014 [citado 2 Feb 2016];4(1):9-15. Disponible en: http://www.dialnet.unirioja.es
Recibido: 4 de abril de 2017.
Aprobado: 22 de diciembre de 2017.
Dora Inés Kozusny-Andreani. Universidade Brasil. Fernandópolis - São Paulo, Brazil. Correo electrónico: doraines@terra.com.br