Introduction
Chronic Kidney Disease (CKD) is currently recognized as a major global health problem, not only because of its high prevalence and incidence, but also because of its remarkable contribution to premature mortality, especially in low and middle-income countries. (1
The Latin American region has the highest mortality rate due to CKD worldwide, and the condition is the second leading cause of years of potential life lost. (2) In El Salvador, chronic renal failure was the leading cause of potential life lost in the period of 2011-2015. (3
The epidemiological behavior of CKD in the Latin American region has distinctive characteristics. The very high prevalence and mortality rates in several Central American countries are most prominent in people under 60, especially male. 4,5 Approximately one third of those affected suffer from CKD that cannot be attributed to those etiological conditions that typically precede chronic kidney damage (mainly, hypertension and diabetes). (6
This modality, a chronic interstitial nephritis that has been called Chronic Kidney Disease of Non-Traditional Causes (CKDnT) or Chronic Kidney Disease of Unknown causes (CKDu), has been identified by the World Health Organization (WHO) as an epidemic problem that confers special connotation to this health emergency. (7,8,9) This phenomenon has also emerged in other parts of the world, (10 with particular intensity in agricultural areas of Sri Lanka, (11,12 and in southern India. 13
The epidemic has repeatedly attracted the attention of the most recent scientific literature. 14,15
In the specific case of El Salvador, a recent work describes in detail the features of the epidemic. 6 Based on the National Survey of Chronic Diseases in Adults in El Salvador (ENECA-ELS 2015), 16 the results confirm that CKD has reached alarming proportions in that country. Additionally, the findings of previous research in some Salvadoran agricultural communities 17 highlight the elevated degree of exposure of the population to agrochemicals, especially in rural areas.
Several hypotheses have been raised to explain the epidemic, 18 with two of them especially prominent.
One hypothesis proposes that herbicides are primarily responsible for the epidemic. In addition to direct contact with such products during pesticide application, subjects could have exposed to air, water or food contamination. The presence of the CKDnT modality not only among agricultural workers, but also among women and children, as well as in non-agricultural adolescents (19,20,21 and wives of pesticide handlers 22 adds plausibility to this conjecture.
The second hypothesis attributes etiological prominence to prolonged exposure to high temperatures 23 in a context of exhausting working conditions, to which dehydration is added due to insufficient or not appropriated fluid intake. (24 As it has been known for decades, (25 dehydration compromises renal function by itself, and cause, among other disorders, hyperosmolarity, hyperuricemia and accumulation of nephrotoxins. In turn, the successive subclinical episodes of acute kidney damage could lead to chronic damage of these organs.
A more comprehensive explanatory model for the epidemic has been proposed by a group of researchers from El Salvador and Cuba. 26 This proposal does not make prior assumptions but contemplates the possibility that both components contribute. This is in line with the opinion of several specialists who support a multifactorial and integrated approach, which also takes into account the social determinants of health, non-sustainable economic growth models and environmental and public health degradation. 7,27
However, remarkable controversy persists about the causes of the epidemics of CKDnT that affect agricultural communities in Central America and South Asia 28 despite the efforts of many researchers and ministries of health to involve renowned experts with the support of international public agencies. Some previous studies have focused on investigating risk factors and causal hypotheses (including heat stress and dehydration, pesticides, infectious agents and heavy metals), but still there is no consensus on this matter. 29,30
Although prevalence surveys are indispensable initial steps, it is imperative to increase the number and, above all, the quality of epidemiological investigations. Efforts should be made to go beyond the mere identification of the magnitude and location of the problem and to focus on the discovery of its causes. 31
ENECA-ELS 2015 was designed not only to describe the problem but also to encourage an analysis able to provide explanatory clues. In this article we communicate the main findings of such an analytical effort in relation to the possible role of agrochemicals in the CKD epidemic.
Methods
Type of study, universe and sample
Consideration should be given to the temporality of the exposures in a formal way and as rigorous as possible. For that reason, a case-control study was carried out, using the national sample provided by ENECA-ELS 2015.
The inclusion criteria and the probabilistic sample design applied, as well as the procedures for data collection, measurement of physical-chemical parameters, quality control, standardization procedures and data validation, are detailed in our previous work describing the epidemic. 6
To assess the association between the risk factors considered and CKD, of the 4,817 adults studied in the ENECA-ELS 2015, all persons suffering from CKD (519) were considered cases, no matter the possible origin of the disease and regardless of the peculiarities that they exhibited. Such classification was made according to the initially applied laboratory tests (essentially, Glomerular Filtration Rate and Albumin / Creatinine Ratio) and with the chronic nature of the condition confirmed by repeated measurements three months after the first measurement, according to Kidney Disease Improving Global Outcomes (KDIGO), 32 to which the Pan American Health Organization adheres. 33 The remaining participants were considered controls.
To study the associations of the diverse risk factors with CKDnT, the same procedure was carried out but restricting the analysis to the subsample of the 2898 individuals who suffered neither diabetes nor hypertension.
ENECA-ELS-2015 included the registration of hundreds of primary variables, coming from the interview and tests. For the present study, only a few of them were selected: three of a general nature (sex, age, urban-rural condition), and those that recorded past exposures to conditions associated with agrochemicals or with these products themselves. The responses showed the existence of prior contact with a high number of specific pesticides (39 in total) with paraquat standing out as the most frequently used product. This was consistent with the much greater frequency of poisoning with paraquat than with other pesticides in El Salvador. (34 We determined the length of past exposure to this specific product, to agrochemicals in general, to agrochemicals considered as nephrotoxic and to those in the different groups defined by the WHO according to the attributed risk involved in their employment. 35 The established codifications (red, yellow, blue and green) indicate degrees of greater to lesser danger of producing intoxication. These warnings concern acute damage, but the repeated injury can obviously lead to illness.
We also determined the time interval during which the following conditions associated with agrochemicals were prevalent during the lifetime of the respondents:
Daily consumption of water glasses in the last 5 years (less than 4, between 4 and 8, between 9 and 12, more than 12)
Agricultural work occupation
Work activity specifically associated with agrochemicals (pesticide application, mixing or formulation of pesticides, flagman)
Water consumption from well, spring or river
Storage of fumigation products and equipment in the residence
Agrochemical aerial fumigation in the work area or residence
Additionally, each participant was diagnosed as:
Diabetic: participant self-reported diagnosis made by a physician prior to the study, or diagnosed during the present study (blood glucose >126 mg Hg)
Having hypertension: if he or she referred that a hypertension had been diagnosed before the study by a physician or when diagnosed during the study (blood pressure ≥ 140/90 mmHg)
Suffering Chronic Kidney Disease (CKD): if at least one of the following characteristics were present:
Suffering Chronic Kidney Disease of Non-Traditional Causes (CKDnT): if suffering from CKD, but without hypertension and diabetes.
We created dichotomous variables to establish whether previous exposures to agrochemicals or to conditions associated with them occurred to some extent or not. In order to assess the possible relationship between the degree of exposure and the disease, we also created variables indicating whether the exposure had extended for at least 5 years and for at least 10 years.
Statistical analysis
For binary variables, the analysis focused on the estimation of the odds ratio (OR) for each risk factor with respect to the presence of CKD. For the continuous variables that measure periods of exposure to agrochemicals or to conditions associated with them, the mean values for cases and controls and their differences were calculated and compared. The analysis for CKDnT was carried out in the same way, but including only with participants who had neither diabetes nor hypertension.
To assess the effect of previous contact with agrochemicals on CKD, logistic regression was used to control for sex, age, urban-rural condition, water consumption, hypertension and diabetes. In order to assess the effect of previous contact with agrochemicals on CKDnT, multivariate models were used controlling for sex, age, urban-rural condition and water consumption.
For all estimates, the confidence intervals were calculated considering both the complex structure of the sample design and the appropriate weights, derived from the non-equiprobabilistic nature of the sample and from the need to calibrate for “non-response”. 36,37
The information was analyzed with the statistical package SPSS Version 24 (Complex Samples module) for Windows.
Ethical considerations
The study was designed and carried out in accordance with the Helsinki Declaration of the World Medical Association guidelines. (38 Written informed consent was obtained from all participants, with specification that their data were to be used only in the framework of the investigation. All participants for whom an abnormality was detected received clinical follow-up by the health services. The study was conducted by National Institute for Health at the request of the Ministry of Health of El Salvador (MINSAL) and the research protocol was approved by the National Committee of Clinical Research of El Salvador of the Higher Council of Public Health.
Results
As expected, we observed a strong association of CKD with both hypertension and diabetes: OR = 3.60, CI95% (2.83-4.59) and OR = 3.75, CI95% (2.92-4.82), respectively.
Table 1 show the prevalence, the OR and their confidence intervals for past exposures to agrochemicals or conditions associated with presence or absence of CKD both for the whole sample and for those without diabetes or hypertension.
Table 2 show the average length of exposure to conditions and types of agrochemicals, as well as the differences of these means between cases and controls and their confidence intervals for CKD and for no CKD both, for the whole sample and for those that suffer nor diabetes neither hypertension.
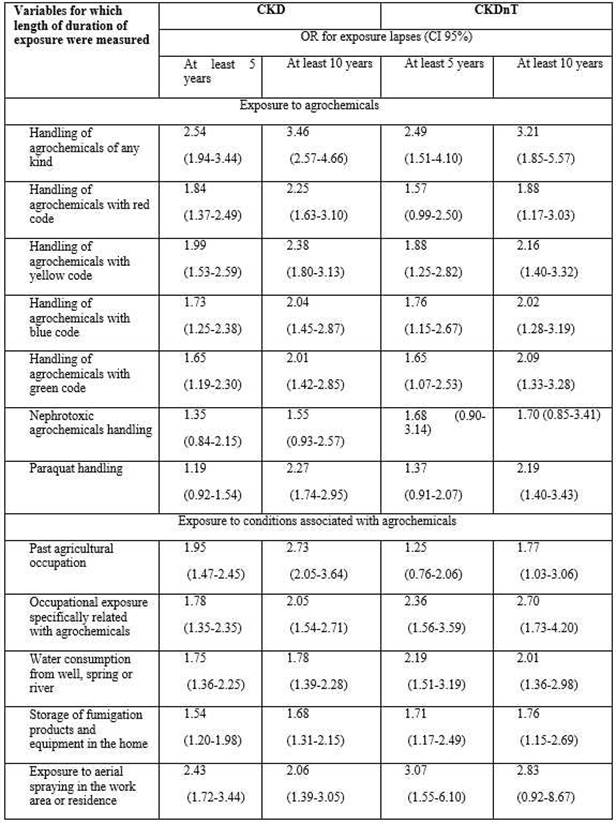
Table 3 presents the changes in OR in relation to CKD and CKDnT respectively when participants had been exposed to agrochemicals and to conditions associated with them for at least 10 years compared with at least 5 years.
After making an adjustment through logistic regression, the OR corresponding to previous contact with agrochemicals of any kind for developing either CKD or CKDnT, decreased slightly compared to the crude estimates when possible confounding factors were controlled for (sex, age, urban-rural condition and water consumption; plus hypertension and diabetes in the case of CKD); but they remained within the general pattern values greater than unity: OR = 1.26 CI95% (1.01-1.64) for CKD and OR = 1.22 CI95% (0.79-1.87) for CKDnT.
Table 1. Prevalence of exposures in the past and OR for each risk factor with respect to presenting CKD and no CKD, in the adult Salvadorian population and in those suffering neither diabetes nor hypertension. ENECA-ELS 2015
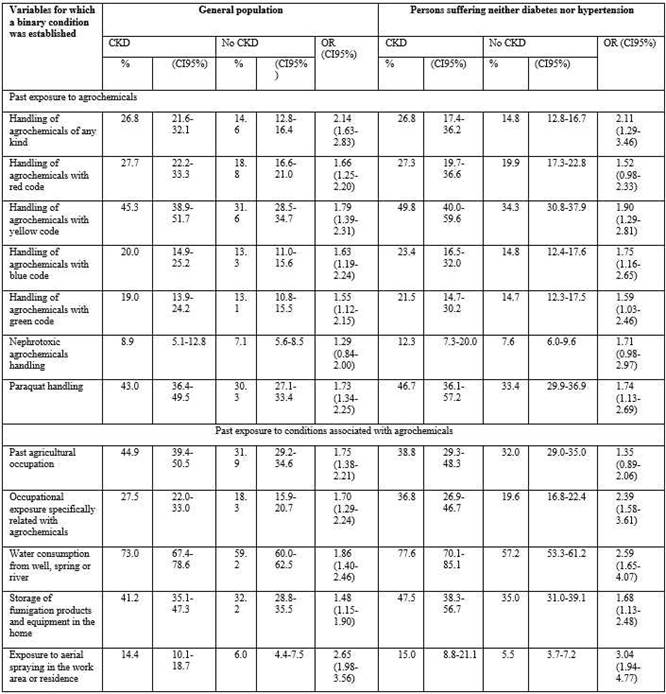
Table 2. Means and differences for years of exposure to agrochemicals and to conditions associated with them along the lifetime, with respect to presenting CKD and no CKD, in the adult Salvadorian population and in those suffering neither diabetes nor hypertension. ENECA-ELS 2015

Discussion
Numerous results published to date suggest that the population working in the agricultural communities most affected by the CKD epidemic has been directly or indirectly exposed to pesticides, herbicides and numerous toxic substances (heavy metals and metalloids), contained in some agrochemicals and fertilizers. (23,37,39 Many of these products are banned in the countries where they are produced; (40,41 however, they are used in large quantities and, often, without any or very little protection. (42 Such is the case in Central America, the region of the world where agrochemicals are used on a larger scale 43 and, in particular, in 2005-2010, El Salvador imported almost 16 million kg of pesticides, with an increase of 171% over the period. That means a very high ratio: 2.5 kg per person over this interval. 44
This report presents the results of the first worldwide epidemiological investigation on CKD and CKDnT based on a probabilistic, two stage sample of a whole nation. The large sample size in a country embroiled in social conflict, and the fact that the chronic nature of the kidney disease initially detected was validated, constitute its two most important strengths.
The association we found between exposures and disease is remarkably strong and consistent: the estimates of all odds ratios exceed unity, and ORs associated with longer past exposures were markedly higher for all of the variables studied.
Many studies that have valued the role of agrochemicals in recent years have yielded results compatible with those found in the present study. (45,46 Most of them use the concept of "statistical significance," as the systematic reviews reveal. Not always do the associations proved to be "statistically significant" (p <0.05). Taking into account these binary outcomes, the results have been incorrectly considered as indicators of whether or not there was a real effect. Consequently, in many cases it was considered that the objectively found association was a "false positive”. Although this is not the only mistake commonly made, it is the one that appears most frequently in the literature. The call to avoid all mention to “statistical significance” has reached the unanimous support of the most renowned statisticians. 47 Consequently, we did not calculate any p-value and circumvented any reference to this concept.
It is worth recalling two classic Bradford Hill warnings for causality analysis: (48 that the stronger the association, the less likely it is that third (confounding) factors can explain it and, that if there is an exposure-response relationship, it is more likely that the hypothetical risk factor influences the onset of the disease. Both conditions are consistently manifested in our study. It should also be noted that all estimates (the 74 odds ratios and the 24 mean differences) would tend to be conservative estimates of the real ones due to the “Incidence-prevalence bias”, which operates when the exposure is related, not only to incidence, but also to survival: if exposure to pesticides is causally associated with increased CKD risk (incidence), it is reasonable to assume that CKD patients who have been exposed will have shorter survival than those who have not been exposed, which will result on the OR (or prevalence ratio) tending to underestimate the relative risk (incidence ratio).
The conjecture about the role of agrochemicals gains additional credibility when it is noted that some pesticides regularly used in Central America have been documented as nephrotoxic agents for decades, 49,50,51 a fact that has been repeatedly emphasized over past years 55,53,54,55 and, highlighted again more recently, in the context of the current health emergency. 47,56
For example, glyphosate (an organophosphorus herbicide commercialized as Roundup by Bayer-Monsanto) has been considered as an agent that compromises the renal function 57,58 and, therefore, as a possible facilitating factor for the epidemic. 59,60 This dangerous herbicide is subject to direct prohibition or harsh restrictions in many regions or countries. 47 Something similar occurs with paraquat, a pesticide repeatedly described as a nephrotoxic in humans. (61,62,63 The association between paraquat, the most intensively used pesticide in El Salvador, and CKD, was also highlighted in our study.
In El Salvador, in 2013 a process was initiated to ban paraquat along with 52 other chemicals. However, despite being the product most involved in both poisonings and accidental deaths in the country, (37 the initiative was blocked through pressure exerted by the industry, and, even today, there is still no current law regulating its use.
Such realities are enough to place the agrochemicals in any hypothetical causality model constructed to explain the epidemic.
Agricultural workers in the areas where the most pronounced outbreak of the epidemic has been documented operate in precarious conditions: rest hours and opportunities for adequate hydration are minimal, there is lack of work stability and laborers are paid according to what they produce each day. They also work in an adverse climate, characterized by high ambient temperature and high humidity levels. 64,65 In addition, and related to the effects of the working conditions, there is often high consumption of non-steroidal anti-inflammatory analgesics (NSAIDs) whose nephrotoxic impact would seem out of the question. 25,66 These factors, far from contradicting the etiological role of pesticides, would potentiate their harmful effect. Such a synergy has a high degree of likelihood, especially in very vulnerable populations, given the marked socioeconomic disadvantages that they suffer. 67
Some researchers believe that dehydration and heat stress best explain the epidemic. (68,69 Along these lines, it has been argued that global warming over the last decades is “the main suspect” in mediating effects of heat stress on renal function in manual workers. (70,71 However, there is no clear proof for this hypothesis. It is noteworthy that the epidemic also manifests itself in some environments in El Salvador, where abandoned containers pollute the environment and water wells with toxic elements such as toxaphene, arsenic, glyphosate and paraquat. (66,72 Hence numerous researchers believe that a predominant role of pesticides has equal or greater merit as a possible explanation for CKDnT; 47,62 and some strongly criticize conjecture that heat stress and dehydration are the major risk factors. (73
Limitations of the study
The main limitation is that the data concerning the exposures were self-reported, which may involve underreporting and, above all, a possible memory bias. Additionally, there was a considerable degree of “non-response”: this was expected and can be partially explained by the fact that the investigation involved surveying and measuring 6,054 people in the extremely hostile conditions prevailing in El Salvador. However, this “non-response”, which resulted in the study being completed only for 4817 subjects, probably did not introduce bias, due to two reasons: first, lower response was largely due to the impaired access to housing enclaves, sometimes blocked by gangs or other informal violent groups that operate in the country, and to the dismissal of hundreds of cases when it was detected that informed consent had not been unequivocally recorded in some questionnaires.
Conclusions
The plausibility of the hypothesis that attributes causal weight to agrochemicals in the epidemic reaches a consistent, systematic and notorious empirical endorsement, complemented by previous evidences and the numerous theoretical indications that supported it. The impact of agrochemicals seems to converge decisively in the causal network, probably enhanced by heat stress and dehydration in markedly hostile social and labor conditions.