My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.26 no.3 La Habana July-Sept. 2009
RESEARCH
Evaluation of different formulations of a dengue-2 chimeric protein and outer membrane vesicles from Neisseria meningitidis in mice
Evaluación en ratones de diferentes formulaciones de la proteína quimérica de dengue-2 y las vesículas de membrana externa de Neisseria meningitidis
Iris Valdés1, Olivia Niebla1, Lisset Hermida1, Jorge Sánchez1, Laura Lazo1, Jorge Martín1, Yaremis Romero1, Yadira Rodríguez1, Maria G Guzmán2, Gerardo Guillén1
1Vaccine Division, Center for Genetic Engineering and Biotechnology, CIGB Ave. 31, PO Box 6162, Havana, Cuba
2PAHO/WHO Collaborating Center for the study of Dengue and its vector, Department of Virology, Pedro Kourí Tropical Medicine Institute, IPK PO Box 601, Havana, Cuba
ABSTRACT
New generation vaccines, particularly those based on recombinant proteins, are generally less reactogenic than traditional live attenuated vaccines. Nevertheless, in terms of immunogenicity, they require potent adjuvants to reach a proper immune response in the recipients. We had previously evaluated the potential capacity of PD5 protein (a vaccine candidate against dengue-2, composed by the P64k protein of Neisseria meningitidis, and the domain III of the dengue Envelope protein), as a vaccine candidate with Freunds adjuvant. In this work, we evaluated the adjuvant capacity of the outer membrane vesicles (OMV) from N. meningitidis on the immunogenicity of the PD5 protein. As a result, after three doses in mice, the groups immunized with three different formulations of OMV elicited high titers of antiviral and neutralizing antibodies against dengue-2 with predominant IgG1 levels. Additionally, in the protection study, the most statistical difference was obtained in one of the three groups immunized with OMV, specifically with one formulation which favors the possible association between the protein and vesicles.
Keywords: adjuvant, dengue virus, Neisseria meningitidis, outer membrane vesicles, recombinant protein.
RESUMEN
La nueva generación de vacunas, específicamente las basadas en proteínas recombinantes, son menos reactogénicas que las vacunas vivas atenuadas tradicionales. Sin embargo, en términos de inmunogenicidad, estas requieren de potentes adyuvantes para lograr la respuesta inmune adecuada. Nosotros previamente hemos evaluado la capacidad potencial de la proteína PD5 (candidato vacunal contra el virus dengue-2, compuesto por la proteína P64k de Neisseria meningitidis y el dominio III de la proteína de la Envoltura de dengue), como un candidato de vacuna con adyuvante de Freund. En el presente trabajo nosotros evaluamos la capacidad adyuvante de las vesículas de membrana externa (VME) de N. meningitidis sobre la inmunogenicidad de la proteína PD5. Como resultado, después de tres dosis en ratones, los grupos inmunizados con tres formulaciones diferentes de VME generaron altos títulos de anticuerpos antivirales y neutralizantes contra dengue-2 con niveles predominantes de anticuerpos IgG1. Adicionalmente, en el estudio de protección, la mayor diferencia estadística fue obtenida en uno de los tres grupos inmunizados con VME, específicamente en el cual se favorece la posible asociación entre la proteína y las vesículas.
Palabras clave: adyuvante, virus dengue, Neisseria meningitidis, vesículas de membrana externa, proteína recombinante.
INTRODUCTION
Dengue virus infections are a serious health problem; indeed, the cause of morbidity and mortality in the majority of tropical and subtropical regions of the world: mainly Southeast and South Asia, Central and South America, and the Caribbean (1). An estimate of 100 million human infections and several hundred cases of dengue hemorrhagic fever/dengue shock syndrome are reported annually (2).
For the development of vaccine candidates against dengue (DEN) virus, the strategies based on recombinant subunit vaccines have been used by several groups (3-14). Most of these strategies have focused on the Envelope (E) protein, which is the main target of neutralizing antibodies (15). In this sense, different candidates have been evaluated in mice with successful results (3, 4, 7, 8, 12-14). Most of them have used potent adjuvants to demonstrate the functionality of the protein; nevertheless these adjuvants cannot be used in humans (7, 8, 12, 13).
Our group has previously reported the expression, characterization and immunological evaluation of the recombinant protein PD5, which contain the domain III of the E protein from DEN-2 fused to the protein carrier P64k (16). This molecule completely protected in Macaca fascicularis monkeys against viral challenge when the Freunds adjuvant was employed in the formulation (10). However, several authors (9, 5, 6, 11, 17) employing fragments of recombinant E protein did not protect monkeys when the aluminum hydroxide was used in the formulations. Nevertheless, we had evaluated the protein PD5 formulated in aluminum hydroxide combined with the outer membrane vesicles (OMV) from Neisseria meningitidis in Chlorocebus aethiops sabaeus monkey (18), but that for mulation was not immunogenic in this species. Alternatively, in the present study we evaluated a different process of formulation to characterize the influence of OMV in the immunogenicity of recombinant protein PD5. These vesicles have been used in different heterologous antigen formulations with successful results (19-21). On the other hand, as a vaccine against the homologous pathogen, its safety and immunogenicity in humans have been demonstrated in several clinical trials (22-25).
In the current study, the formulations of the protein PD5 combined with OMV were evaluated in mice. In all cases, as previously described for the OMV formulations, the components were adsorbed in aluminum hydroxide (22, 23). The immunization schedule was performed; as well the humoral immune response and protection against the infective DEN-2 virus were determined.
MATERIAL AND METHODS
Virus strains
An inactivated preparation from suckling mice brain infected with DEN-2 strain New Guinea C (highly passaged in mice) was used as antigen for antibody detection (26).
For animal immunization and virus challenge, a preparation of infective virus (105 PFU/mL) verified by plaque assay, was employed. It was obtained by homogenization in RPMI 1640 medium (Sigma Aldrich, Ayrshire KA, UK) of suckling mice brain infected with DEN-2 strain New Guinea C.
For the neutralization assay, cell-culture supernatant harvested from Vero cells infected with the DEN-2 strain SB8553 (kindly provided by Dr. MJ Cardosa, University Sarawak, Malaysia) was used.
Recombinant protein and formulation process with the outer membrane vesicles
The design, cloning and expression of the recombinant protein were previously described (16). Briefly, the fusion protein PD5 containing the domain III, coding for amino acid 286-426, of dengue E glycoprotein from strain Jamaica, fused to the carrier protein P64k. The purified protein was kindly provided by Dr. Carlos López from the Center for Genetic Engineering and Biotechnology (CIGB) (27). Protein PD5, produced under GMP conditions and at high levels of purity, was employed for mice immunizations. The calculated percentage of purity was 97%.
Outer membrane vesicles were purified from the N. meningitidis serogroup B strain CU385 (B:4,7: P1.19,15; ST = 33) from the collection of the Finlay Institute, Havana, Cuba (23). The OMV from N. meningitidis serogroup B were obtained by extraction with deoxycholate as previously described (28).
Three formulations were performed with the purified recombinant PD5 protein and OMV. Two different concentrations of the OMV were evaluated 0.5 and 1 mg/mL, these concentrations are corresponding to total proteins present in the vesicles. The formulations were identified as PD5-OMV(0.5) and PD5-OMV(1) respectively. The different concentrations of OMV were firstly mixed with 2.5 mg/mL of PD5 protein to favors the possible association between both antigens. The resultant formulations were subsequently adsorbed on aluminum hydroxide (alum) at a final concentration of 1.44 mg/mL. As the control formulation, the simple mixture of the three components PD5, OMV and alum was performed (PD5 + OMV), in similar conditions of concentration of the OMV and PD5 to the described above.
Enzyme linked immunosorbent assay
The anti-DEN-2 antibody levels in mice sera were determined by an amplified sandwich ELISA system. Briefly, 96 well polystyrene plates (Costar, USA) were coated with 100 μL/well of a mixture of human immunoglobulins (IgG) (5 μg/mL) highly reactive to DEN virus in coating buffer (0.16% Na2CO3, 0.29% NaHCO3, pH 9.5). Plates were incubated 2 h at 37 °C and then, blocked with coating buffer containing 1% bovine serum albumin (BSA). After 1 h incubation at 37 °C, they were washed three times with PBS-T. An extract of suckling mouse brain infected with DEN-2 (100 μL/well) was used as antigen and incubated overnight at 4 °C. After three washes in PBS-T, 100 μL/ well of sera from each group were tested by serial dilutions in PBS-T, starting at 1:1 000 and incubated 2 h at 37 °C. Plates were washed as aforementioned and 100 μL/well of 1:6 000 diluted anti-mouse IgG-HRP conjugate (Amersham-Pharmacia, UK), containing fetal calf serum, were added and the plates were incubated 1 h at 37 °C. After washing again with PBS-T, 100 μL/well of O-phenilendiamine 0.04% in substrate buffer (2% Na2HPO4, 1% citric acid, pH 5.0), were added. The plates were incubated 30 min at room temperature and the reaction was stopped with 50 μL/ well of 12.5% H
Besides, the anti-mouse IgG subclasses antibody in mice sera were determined by a similar ELISA system as mentioned above, but employing 1:5 000 diluted subclass anti-mouse IgG-HRP conjugate (Amersham- Pharmacia, UK).
Plaque reduction neutralization test
Neutralizing antibody titers were measured by plaque reduction neutralization test (PRNT) in BHK-21 cells as previously described (29). The strain SB8553 was used in this test. The neutralizing antibody titer was identified as the highest serum dilution that reduced the number of virus plaques in the test by 50% or more. The monoclonal antibody 4G2 was used as positive control (30).
Mice immunization
Groups of 20 female 5-6 week-old Balb/c mice (CENPALAB, Cuba) were injected by the intraperitoneal (i.p.) route on days 0, 15, and 30 with the different formulations containing 15 μg of the PD5 purified protein and OMV with alum as adjuvant. Similarly, negative control mice received 15 μg of OMV at a final concentration of aluminum hydroxide of 1.44 mg/mL. As positive control, one dose of 105 PFU/mL of infective DEN-2 virus (strain New Guinea C), obtained from infected suckling mouse brain, was inoculated by the same route. Ten mice from each group were bled two weeks after the last dose; and sera were collected for further immunological analysis. The maintenance and care of the experimental animals used in this research complied with the Cuban Institute of Health guidelines for the humane care and use of laboratory animals.
Animal protection study
One month after the last dose, ten remaining animals (non-bled) were injected intracranially (i.c.) with 20 μL of a suspension of DEN-2 (strain New Guinea C) virusinfected suckling mouse brain containing 100 median lethal doses (LD50). Mice were observed daily for 22 days. Morbidity and mortality were recorded.
Statistical analysis Direct or transformed (Log10) data that passed the normality test (DAgostino & Pearson omnibus normality test) and showed variance homogeneity (Bartlett´s test) were analyzed by ANOVA parametric tests. Data that not fulfill normality and /or variance homogeneity test, even after transformations were analyzed by nonparametric test. The analysis of data from ELISA was assessed using a Newman-Keuls multiple comparison test. The analysis of data from PRNT was assessed using a Kruskal-Wallis non-parametric test with Dunns multiple comparison test. Data from protection assay were analyzed by the log-rank test. In all cases, the GraphPad Prism version 5.00 for Windows, GraphPad Software, San Diego California USA, www.graphpad.com was employed.
RESULTS
Antiviral antibody response after immunization
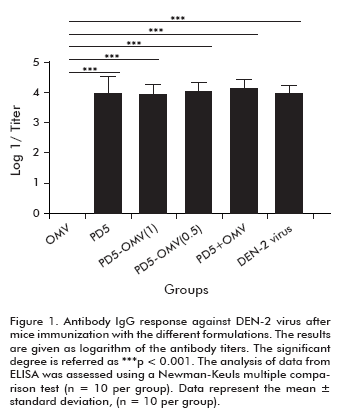
Three formulations, containing the recombinant protein PD5 and OMV, were inoculated in mice to evaluate the humoral immune response induced (as stated in Materials and methods section). Two additional groups were immunized with PD5-alum and OMV-alum, respectively. After the third dose, ten mice per group were bled to obtain the sera. To determine the presence of antiviral antibodies, each serum was evaluated by a capture ELISA system. In all groups, high levels of anti-DEN-2 antibodies were detected, with statistically significant differences compared to the negative control group (p < 0.001). Additionally, all the formulations containing the PD5 protein elicited levels of antibodies similar to those induced in the control group immunized with DEN virus (Figure 1).
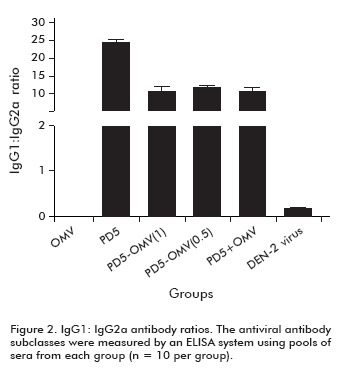
Antiviral IgG subclasses were also determined by the same capture ELISA system using pools of sera from each group. As shown in figure 2, in groups immunized with the protein PD5, the predominant pattern was IgG1 subtype. However, in the groups immunized with OMV as adjuvant, the ratio IgG1:IgG2a was lower than that observed in the PD5-alum group. In contrast, the pool sera from mice inoculated with the infective virus showed high levels of IgG2a compared to those of IgG1, reflected as the ratio IgG1:IgG2a < 1 (Figure 2).
Neutralizing antibodies after immunization
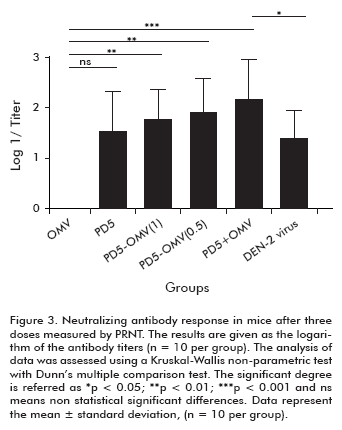
The functionality of the antibodies elicited by the different formulations was measured by PRNT. Figure 3 shows the neutralizing antibody response in each group. In contrast to antiviral antibody response, in the groups inoculated with formulations of OMV, independently of the formulation process, high levels of neutralizing antibodies were elicited with statistically significant differences with respect to the negative control group (p < 0.01) (Figure 3). In case of the viral group, a low response of neutralizing antibodies was detected.
Protection study
One month after the last dose, the 10 remaining mice from each group were inoculated by the i.c. route with a live neuro-adapted DEN-2 virus. Figure 4 shows the analysis of morbidity and mortality data following challenge and for 22 subsequent days. Animals immunized with the DEN-2 virus achieved a 100% protection, however during the observation period they showed symptoms of illness (Figure 4A and 4B).
Unlike to the results of the humoral immune response, only mice immunized with formulations PD5-OMV(1) and PD5-OMV(0.5) showed statistically significant differences with respect to the negative control group in terms of percentage of survival, (p < 0.01). The group immunized with the simple mixture of the formulation components (PD5 + OMV) did not show statistically significant difference compared to the negative control group (Figure 4B). At the end of the observation period, 40% and 20% of animals immunized with PD5-OMV(1) and PD5-OMV(0.5) survived, respectively. The group that received protein PD5 alone had 20% of survival, however did not show significant statistical differences with the OMV group (Figure 4B).
DISCUSSION
In a previous work, we had evaluated a PD5-OMV formulation absorbed in aluminum hydroxide in monkeys. As a result, the PD5-OMV formulation was not able to induce antiviral and neutralizing antibodies in this model. Besides, it did not protect in vivo protection assay of viral challenge (18). For this reason, in the present work we evaluated other formulation processes to enhance the immunogenic capacity of PD5 when it is combined with OMV.
The OMV from N. meningitidis have been widely employed (22-25) and their adjuvant capacity has been demonstrated (19-21) .
One of the proposed mechanisms through which OMV may exert this adjuvant effect is by acting as ligands for several receptors that stimulate the innate immunity such as the Toll-like receptors (31). In fact, different groups have reported vaccine formulations combining the OMV and heterologous antigens from malaria (19), Haemophilus influenzae type b (32), and pneumococcus (33), with successful results.
The current work aimed at evaluating the influence of OMV on the immunogenicity of the recombinant protein PD5. In terms of antiviral antibodies no differences were observed among the groups immunized with recombinant protein. On the other hand, the functional antibody response was higher in the groups immunized with formulations containing the protein PD5 and OMV. These evidences suggest the proper antigenic presentation of the protein PD5 in the context of OMV independently of the formulation process.
In the case of the viral control group, the neutralizing antibody titers were low. However, similarly low antibody titers have been reported by other authors for the same serotype (34), and for DEN-1 (7) and DEN-4 (14), after immunization with one viral infective dose. A possible explanation could be related with the short-lived viremia produced in mice after virus immunization by the intraperitoneal route (35), and consequently, with the induction of a limited immune response. However, this response was sufficient to induce total protection after homologous viral challenge.
Moreover, it has also been described that OMV as adjuvant modulates the immune response induced by different antigens measured by the ratio IgG1:IgG2a (36). Consistently, in the current work, the ratio IgG1:IgG2a of formulations of the protein PD5 and OMV were lower than that determined in the PD5- alum group, though the typical Th2 pattern was kept. This behavior could be explained by the soluble nature of the protein, mainly inductor of humoral immune response since the domain III is the principal target of neutralizing antibodies (15). Additionally, in all the formulations, the alum was the base adjuvant being a potent Th2 inducer.
The protection study against the homologous infective virus was the other parameter measured. Despite the difference of this animal model with respect to the human disease, significant differences in the percentages of survival compared to the negative control group were attained as other indicator of a functional immune response. In the challenge assay, only the groups immunized with the formulations based on the interaction between the PD5 protein and the OMV demonstrated statistical differences with respect to the negative control group (p < 0.01). On the contrary, when both components were simply mixed together with alum, the induced immune response was unable to protect the animals despite the highest neutralization titers detected in this group (Figure 4B).
Various candidates containing the domain III of the E protein have been evaluated by different groups and their protective capacity has been assessed in the encephalitis mouse model (3, 4, 7, 8, 13, 14). However, the best results in percentage of survival have been obtained when potent adjuvants, not suitable for human use, have been employed (7, 8, 13). On the other hand, a tandem domain III candidate against the four dengue serotypes was developed by Chen and coworkers, although in this study, the protective capacity of the humoral response was only elicited in mice; and excluded the cell-mediated mechanisms (12). Only Simmons and coworkers achieved an 80% survival rate against viral challenge with formulations in alum, but they used a very small number of animals per group in their study (4). Of the candidates mentioned above, only two have been assayed in monkeys using alum, and did not protect against viral challenge (10, 17).
In general, the percentages of survival obtained in this study could have been affected by the high severity of the challenge assay, manifested by the fact that, in spite of the 100% survival rate obtained in the virus-immunized group, several mice in this group became ill during the observation period. On the other hand, considering the similarity in the humoral immune response induced by the formulations containing PD5, we can suggest that a possible association between OMV and PD5 may have favored another mechanism of the immune system that was responsible for the protection obtained. Several groups have reported the lack of correlation between neutralizing antibodies and protection (5-8, 37-39). In this sense, cell-mediated immune mechanisms could have played a role in protecting against the virus, in fact, some modulation in the IgG subclass was detected.
The lack of correlation between neutralizing antibodies and protection was clearly evident in the virus- immunized group. Accordingly, in this case, the cellular immune response was reflected in the ratio of IgG1:IgG2a, indicating a potent protector mechanism in this animal model. In general, the percentages of survival obtained in the study were lower than those previously reported by our group (8).
Taking into account, the evidences of the present study support the OMV as one of the possible adjuvant formulation for PD5. Further studies in monkeys would be needed to confirm the capacity of PD5- OMV formulation to provide complete protection of monkeys against viral challenge.
ACKNOWLEDGMENTS
The authors are very grateful to Dr. Ricardo Silva and Dr. Harold Curiel for their critical reading and useful comments in the revision of the manuscript. This investigation received financial support from the Cuban Program for Dengue Vaccine Development.
REFERENCES
1. Kurane I. Dengue hemorrhagic fever with special emphasis on immunopathogenesis. Comp Immunol Microbiol Infect Dis 2007;30:329-40.
2. Gubler DJ. Epidemic dengue/dengue hemorrhagic fever as a public health, social and economic problem in the 21st century. Trends Microbiol 2002;10:100-3.
3. Srivastava AK, Putnak Jr, Warren RL, Hoke CH. Mice immunized with a dengue type 2 virus E and NS1 fusion protein made in Escherichia coli are protected against lethal dengue virus infection. Vaccine 1995;13:1251-8.
4. Simmons M, Nelson WM, Wu SJ, Hayes CG. Evaluation of the protective efficacy of a recombinant dengue envelope B domain fusion protein against dengue 2 virus infection in mice. Am J Trop Med Hyg 1998;58:655-2.
5. Velzing J, Groen J, Drouet MT, van Amerongen G, Copra C, Osterhaus ADME, et al. Induction of protective immunity against Dengue virus type 2: comparison of candidate live attenuated and recombinant vaccines. Vaccine 1999;17:1312-20.
6. Guzmán MG, Rodríguez R, Rodríguez R, Hermida H, Álvarez M, Lazo L, et al. Induction of neutralizing antibodies and partial protection from viral challenge in Macaca fascicularis immunized with recombinant dengue 4 virus envelope glycoprotein expressed in Pichia pastoris. Am J Trop Med Hyg 2003;69:129-34.
7. Hermida L, Rodríguez R, Lazo L, Bernardo L, Silva R, Zulueta A, et al. A fragment of the envelope protein from dengue-1 virus, fused in two different sites of the meningococcal P64k protein carrier, induces a functional immune response in mice. Biotechnol Appl Biochem 2004;39:107-14.
8. Hermida L, Rodríguez R, Lazo L, Silva R, Zulueta A, Chinea G, et al. A dengue-2 Envelope fragment inserted within the structure of the P64k meningococcal protein carrier enables a functional immune response against the virus in mice. J Virol Methods 2004;115:41-9.
9. Putnak RJ, Coller BA, Voss G, Vaughn DW, Clements D, Peters I, et al. An evaluation of dengue type-2 inactivated, recombinant subunit, and live-attenuated vaccine candidates in the rhesus macaque model. Vaccine 2005;23:4442-52.
10. Hermida L, Bernardo L, Martín J, Álvarez M, Prado I, López C, et al. A recombinant fusion protein containing the domain III of the dengue-2 envelope protein is immunogenic and protective in nonhuman primates. Vaccine 2006;24:3165-71.
11. Simmons M, Porter KR, Hayes CG, Vaughn DW, Putnak R. Characterization of antibody responses to combinations of a dengue virus type 2 DNA vaccine and two dengue virus type 2 protein vaccines in rhesus macaques. J Virol 2006;80:9577-85.
12. Chen S, Yu M, Jiang T, Deng Y, Qin C, Qin E. Induction of tetravalent protective immunity against four dengue serotypes by the tandem domain III of the Envelope protein. DNA Cell Biol 2007;26:361-7.
13. Babu JP, Pattnaik P, Gupta N, Shrivastava A, Khan M, Rao PV. Immunogenicity of a recombinant envelope domain III protein of dengue virus type-4 with various adjuvants in mice. Vaccine 2008;26:4655-63.
14. Lazo L, Zulueta A, Hermida L, Blanco A, Sánchez J, Valdés I, et al. The Dengue-4 envelope domain III fused twice within the meningococcal P64k protein carrier induces partial protection in mice. Biotechnol Appl Biochem 2009;52:265-71.
15. Chambers TJ, Chang S, Galler R, Rice C. Flavivirus genome organization, expression and replication. Annu Rev Microbiol 1990;44:649-88.
16. Zulueta A, Hermida L, Lazo L, Valdés I, Rodríguez R, López C, et al. The fusion site of envelope fragments from each serotype of dengue virus in the P64k protein, influence some parameters of the resulting chimeric construct. Biochem Biophys Res Commun 2003;308:619-26.
17. Bernardo L, Izquierdo A, Álvarez M, Rosario D, Prado I, López C, et al. Immunogenicity and protective efficacy of a recombinant fusion protein containing the domain III of the dengue 1 envelope protein in non-human primates. Antiviral Res 2008;80:194-9.
18. Valdés I, Hermida L, Martín J, Menéndez T, Gil L, Lazo L, et al. Immunological evaluation in nonhuman primates of formulations based on the chimeric protein P64k-domain III of dengue 2 and two components of Neisseria meningitidis. Vaccine 2009;27:995-1001.
19. Lowell GH, Ballou WR, Smith LF, Wirtz RA, Zollinger WD, Hockmeyer WT. Proteosome-lipopeptide vaccines: enhancement of immunogenicity for malaria CS peptides. Science 1988;240:800-2.
20. Wetzler LM. Immunopotentiating ability of neisserial major outer membrane proteins. Use as an adjuvant for poorly immunogenic substances and potential use in vaccines. Ann N Y Acad Sci 1994;730:367-70.
21. Fusco PC, Michon F, Laude-Sharp M, Minetti CA, Huang CH, Heron I, et al. Preclinical studies on a recombinant group B meningococcal porin as a carrier for a novel Haemophilus influenzae type b conjugate vaccine. Vaccine 1998;16:1842-9.
22. Bjune G, Hoiby EA, Gronnesby JK, Arnesen O, Fredriksen JH, Halstensen A, et al. Effect of outer membrane vesicle vaccine against group B meningococcal disease in Norway. Lancet 1991;338:1093-6.
23. Sierra GV, Campa HC, Varcacel NM, Garcia IL, Izquierdo PL, Sotolongo PF, et al. Vaccine against group B Neisseria meningitidis: protection trial and mass vaccination results in Cuba. NIPH Ann 1991;14:195-210.
24. Peeters CC, Rumke HC, Sundermann LC, Rouppe van der Voort EM, Meulenbelt J, Schuller M, et al. Phase I clinical trial with a hexavalent PorA containing meningococcal outer membrane vesicle vaccine. Vaccine 1996;14:1009-15.
25. Oster P, Lennon D, OHallahan J, Mulholland K, Reid S, Martin D. MeNZB: a safe and highly immunogenic tailor-made vaccine against the New Zealand Neisseria meningitidis serogroup B disease epidemic strain. Vaccine 2005;23:2191-6.
26. Clarke DH, Casals J. Techniques for hemagglutination and hemagglutination inhibition with arthropod-borne viruses. Am J Trop Med Hyg 1958;7:561-73.
27. López C, Sánchez J, Hermida L, Zulueta A, Marquez G. Cysteine mediated multimerization of a recombinant dengue E fragment fused to the P64k protein by Immobilized metal ion affinity chromatography. Protein Expr Purif 2004;34:176-82.
28. Fredriksen JH, Rosenqvist E, Wedege E, Bryn K, Bjune G, Froholm LO, et al. Production, characterization and control of MenBvaccine Folkehelsa: an outer membrane vesicle vaccine against group B meningococcal disease. NIPH Ann 1991;14:67-79.
29. Morens DM, Halstead SB, Repik PM. Simplified plaque reduction assay for dengue viruses by semimicro methods in BHK 21 cells: comparison of the BHK suspension test with standard plaque reduction neutralization. J Clin Microbiol 1985;22:250-4.
30. Kaufman BM, Summers PL, Dubois DR, Eckels KH. Monoclonal antibodies against dengue 2 virus E-glycoprotein protect mice against lethal dengue infection. Am J Trop Med Hyg 1987;36:427-34.
31. MacLeod H, Bhasin N, Massari PLM. Role of protein tyrosine kinase and Erk1/2 activities in the toll-like receptor 2-induced cellular activation of murine B cell by Neisserial porin. Clin Vaccine Immunol 2008;15:630-37.
32. Donnelly JJ, Deck RR, Liu MA. Immunogenicity of a Haemophilus influenzae polysaccharide-Neisseria meningitides outer membrane protein vaccine. J Immunol 1990;145:3071-9.
33. Giebink GS, Koskela M, Vella PP, Harris M, Le CT. Pneumococcal capsular polysaccharidemeningococcal outer membrane protein complex conjugate vaccines: immunogenicity and efficacy in experimental pneumococcal otitis media. J Infect Dis 1993;167:347-55.
34. Bernardo L, Yndart A, Vazquez S, Morier L, Guzman MG. Antibody Responses to Asian and American Genotypes of Dengue 2 Virus in Immunized Mice. Clin Diagn Lab Immunol 2005;12:361-2.
35. Brinton MA, Perelygin AA. Genetic resistance to flaviviruses. Adv Virus Res 2003;60:43-85.
36. Pérez O, Lastre M, Lapinet J, Bracho G, Díaz M, Zayas C, et al. Immune response induction and new effector mechanisms possibly involved in protection conferred by the Cuban anti-meningococcal BC vaccine. Infect Immun 2001;69:4502-8.
37. Deubel V, Kinney RM, Exposito JJ, Cropp CB, Vorndam AV, Monath TP, et al. Dengue 2 virus envelope protein expressed by a recombinant vaccinia virus failed to protect monkeys against dengue. J Gen Virol 1988;69:1921-9.
38. Eckels KH, Dubois DR, Summers PL, Schlesinger JJ, Shelly M, Cohen S, et al. Immunization of monkeys with baculovirus-dengue type-4 recombinants containing envelope and nonstructural proteins: evidence of priming and partial protection. Am J Trop Med Hyg 1994;50:472-8.
39. Putnak R, Barvir DA, Burrous JM, Dubois DR, DAndrea VM, Hoke CH, et al. Development of a purified, inactivated, dengue-2 virus vaccine prototype in Vero cell: immunogenicity and protection in mice and rhesus monkeys. J Infect Di 1996;174:1176-84.
Received in July, 2009.
Accepted for publication in September, 2009.
Iris Valdés. Vaccine Division, Center for Genetic Engineering and Biotechnology, CIGB Ave. 31, PO Box 6162, Havana, Cuba. E-mail: iris.valdes@cigb.edu.cu