My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.26 no.3 La Habana July-Sept. 2009
REPORT
Virus-like particles of the Rabbit Hemorrhagic Disease Virus obtained in yeast are able to induce protective immunity against classical strains and a viral subtype circulating in Cuba *
Omar Farnós1, Erlinda Fernández1, Maylin Chiong1, Marisdania Joglar1, Dalia Rodríguez2, María P Rodríguez1, Elsa Rodríguez1, Ernesto M González1, Odaysa Valdés2, Marisela Suárez1, Carlos Montero1
1Animal Biotechnology Division, Center for Genetic Engineering and Biotechnology, CIGB. Ave. 31 / 158 and 190, Cubanacán, Playa, PO Box 6162, CP 10600, Havana, Cuba
2Diagnose and Investigation, National Center for Epizootiology Ave. 51 # 33212, Arroyo Arenas, Havana, Cuba
ABSTRACT
Four epizootics caused by rabbit hemorrhagic disease virus (RHDV) have been recorded in Cuba from 1993 to 2005. Each time, thousands of animals have died or have been slaughtered to avoid the spread of the disease. Cell culture systems allowing the in vitro replication of RHDV are not available to date. Moreover, the amount of the recombinant capsid protein (VP60) obtained in heterologous expression systems does not commonly exceed a few tens of milligrams per liter of culture. In this paper, we report the expression of VP60 in two strains of the Pichia pastoris yeast with the highest expression levels obtained so far. VP60 was glycosylated, associated to the yeast cell disruption pellet at a concentration of 1.5 g/L in the first case, or soluble in the intracellular fraction at approximately 300 mg/L, following a different cloning strategy. These recombinant variants showed similar antigenic properties to those of the native protein, as determined by monoclonal antibodies. The soluble VP60 showed a higher number of protective epitopes, due to the formation of multimers that were similar in size and structure to the native RHDV capsids. Both antigens induced potent RHDV-specific immune responses in experimental animals. The antibodies produced were able to inhibit the in vitro hemagglutination of a viral strain isolated during the last outbreak in Cuba. A molecular and antigenic characterization of this strain was also carried out and led to its classification as a member of the highly pathogenic RHDVa subtype. Both recombinant antigens induced a specific, protective and long-lasting immune response against classical strains and also against the RHDVa subtype.
INTRODUCTION
Rabbit Hemorrhagic Disease is highly lethal and contagious and was first detected in China in 1984 (1). Mortality rates in adult rabbits range from approximately 40 to 90%. Infected rabbits commonly die within the first 24-72 hours post-infection due to the occurrence of intravascular disseminated coagulation and hemorrhages in the liver, spleen, kidneys and lungs (2, 3). Cell death caused by apoptosis has been observed in macrophages and in hepatic and vascular endothelial tissues (4). The etiological agent, Rabbit Hemorrhagic Disease Virus (RHDV) (5), is a member of the Lagovirus genus and belongs to the Caliciviridae (6) family. The virus is highly resistant to the environment, spreads rapidly and is transmissible by nasal, conjunctival and oral routes (7).
At present, RHDV is considered endemic in Europe and East Asia (8), in which viral outbreaks affect the productive sector and produce the instability of ecosystems that depend on wild rabbit populations (9). Outbreaks have also occurred in Africa and in the Americas in countries such as Mexico, the United States, Cuba and Uruguay (10-14). Cuba is considered the most strongly affected country in the American region, with four epizootics reported in the years 1993, 1997, 2000-2001 and 2004-2005. As consequence, thousands of rabbits have died or have been slaughtered each time.
Culture systems for the in vitro replication of RHDV are not available to date. Therefore, commercial vaccines for its prevention consist exclusively of formulations prepared from the organs of rabbits infected with the virus (15). As the use of these preparations for high-scale vaccination involves biosafety and epidemiological risks, there have been efforts to generate subunit vaccines based on the heterologous expression of the viral capsid protein. This protein has been obtained in a variety of systems such as Escherichia coli (16), insect cell cultures (17-19), Saccharomyces cerevisiae (20) and transgenic plants (21, 22). However, the expression levels obtained in these systems have not exceeded tens of milligrams per liter of culture. At the same time, vaccine production based on replicative viral vectors has also been restricted due to regulatory issues (23).
In this paper, we describe the molecular and antigenic characterization of a Cuban RHDV isolate and the construction of two recombinant Pichia pastoris strains, searching for a high expression level of VP60 from RHDV. The structural analysis and the corroboration of the antigenic determinants present in these two protein variants were also accomplished. An immunization trial was conducted using different preparations based on VP60 and several administration routes, to study the generation of the earliest IgG specific response, the time course of antibody titers and their persistence for two years. The protective titers were measured by ELISA and hemagglutination inhibition assays using a representative strain of the virus or the isolate characterized as RHDVa. The impact of these results in vaccine production issues and in the design of rational immunization strategies is discussed.
RESULTS AND DISCUSSION
Molecular and antigenic characterization of a Cuban isolate of the Rabbit Hemorrhagic Disease Virus
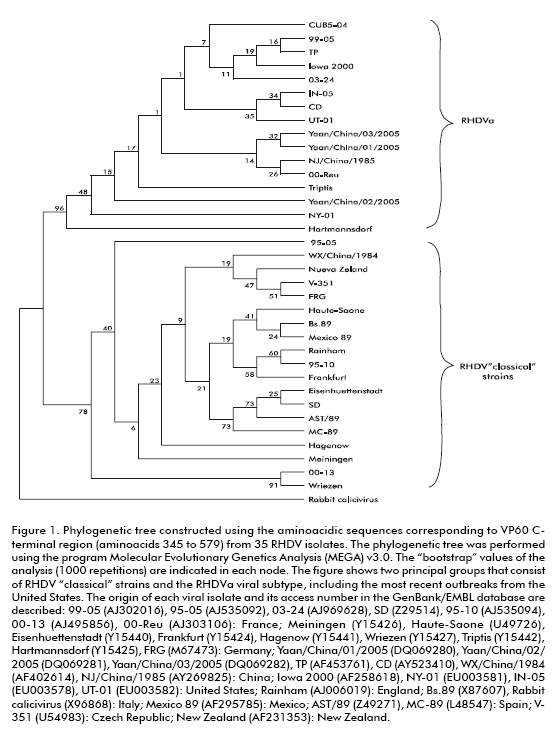
The RHDV Cuban strain CUB5-04 was isolated from liver samples of a rabbit that died in Havana province during the last epizootic, occurring in the years 2004- 2005. The 1740 bp coding region of the CUB5-04 VP60 capsid protein and its deduced amino acid sequence were included in the GenBank/EMBL database, under the access number DQ841708. The last 234 amino acids, corresponding to the C-terminal portion (that include the most variable region) of the protein, were compared with the VP60 analogous region from different international isolates. The sequence alignment showed the highest identity levels with viral strains that belong to an antigenic variant called RHDVa (24). This subtype is characterized by a high pathogenicity and by antigenic variations compared to the RHDV with the monoclonal antibody 3B12 that recognizes a protective epitope that is only present in the RHDVa subtype. The phylogenetic analysis performed with the VP60 from CUB5-04 and other 32 RHDV strains, showed that the Cuban strain shared genetic variations with the classical strains that were also present in the RHDVa isolates (Figure 1). These results confirm that the viral strain isolated during the last Cuban epizootic of the Rabbit Hemorrhagic Disease belongs to the RHDVa subtype.
Expression and characterization in pichia pastoris of the rabbit hemorrhagic disease virus VP60 capsid protein
Two different expression plasmids were constructed to obtain the secreted or intracellular expression of RHDV VP60 from the AST/89 Spanish strain in the P. pastoris yeast. The VP60 gene was cloned after the sucrose invertase secretion signal (SUC2) of S. cerevisiae in the pPS7 vector to obtain the pPSVP60 expression plasmid. In a different approach, the VP60 gene was cloned without a secretion signal in the pNAO vector to generate the expression plasmid pNAOVP60. Both plasmids contained the VP60 gene under the transcriptional regulation of the AOX1 promoter. The P. pastoris MP36 strain was then transformed by electroporation with pPSVP60 or pNAOVP60 and the recombinant yeast strains PVP11 and PVP12 were obtained, respectively. Cells from both strains were grown in 5L bioreactors and VP60 expression was confirmed by SDS-PAGE and Western blot analyses.
The PVP11 strain phenotype was identified as Mut+ by Southern blot. The analysis of the PVP11 cell disruption pellet showed the expected protein band of approximately 60 kDa as well as a broader band of about 90 kDa that corresponds to a VP60 glycosylated fraction. The study under nonreducing conditions indicated the formation of VP60 multimeric structures with a molecular weight of over 220 kDa. No recombinant proteins were detected in the culture and cell disruption supernatants in this strain (Figure 2A). The characterization of the solubilized recombinant protein by size exclusion high efficacy liquid chromatography (sec-HPLC) showed a first maximum that eluted with a retention time of approximately 16 minutes. This time was close to that of the recombinant particulate surface antigen of the Hepatitis B virus studied under the same conditions (TSK G-5000 PW column, equilibrated with PBS) at a flow rate of 0.4 mL/min (26). A second fraction eluted later with a retention time of about 29 minutes. VP60 was detected in both fractions by immunodot using a hyperimmune serum to RHDV (Figure 2B).
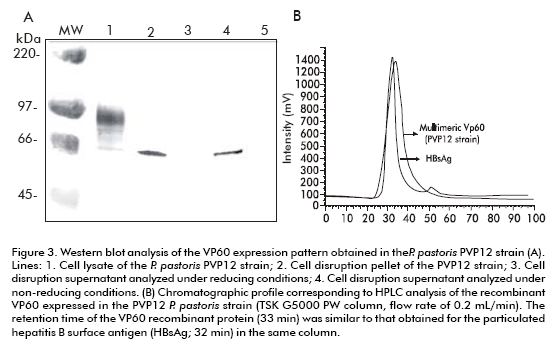
The recombinant VP60 expressed by the PVP12 strain was obtained intracellularly and was soluble with a molecular weight of 60 kDa after 12 hours of induction with methanol. No recombinant protein was found to be associated to the cell disruption pellet of this strain. The analysis of the cell disruption supernatant under non-reducing conditions suggests the formation of high molecular weight structures that were unable to enter the gel (Figure 3A). The protein eluted in a single, homogeneous, and well-defined peak with a retention time of 33 minutes, as shown by the chromatographic profile in a TSK G-5000 PW column, equilibrated with PBS. This retention time was also similar to that obtained for HBsAg (> 1500 kDa) in this column, with a flow of 0.2 mL/min. This protein, purified by size-exclusion chromatography, was characterized by ratezonal and equilibrium sucrose gradient ultracentrifugation (10-30% for rate-zonal and 20- 70% for equilibrium gradient centrifugation). VP60 was detected in a predominant peak with an estimated sedimentation coefficient of 120S that was slightly lower than that obtained for native RHDV (153S for the Bs.89 Italian isolate). This sedimentation coefficient was similar to that described for the formation of virus-like particles (VLPs) in insect cell cultures (118S) (27). The buoyant density of recombinant VP60 was estimated to be in the range of 1.30-1.32 g/mL, which agrees with the values previously described for VLPs obtained in insect cell cultures. These results suggest the formation of multimeric structures that are similar in mass and shape to native RHDV.
The formation of multimeric structures by the recombinant VP60 protein from P. pastoris was confirmed by transmission electron microscopy. The analysis of VP60 solubilized from the disruption pellet of the PVP11 strain showed the presence of multimeric structures that resemble protein aggregates rather than ordered capsids (Figure 4A). This has been previously described for particulate antigens obtained at high levels associated with the cell debris fraction in this P. pastoris strain (28, 29). In contrast, the analysis of purified VP60 from the PVP12 strain evidenced the formation of particles with an average diameter of 30- 35 nm, similar to empty RHDV capsids (Figure 4B).
The expression levels of VP60 in PVP11 and PVP12 strains were estimated by a sandwich ELISA in 1.5 g per liter of culture and 480 µg per liter of the disruption supernatant, respectively. These levels were quite higher than those obtained for this protein in expression systems as bacteria (16), insect cells (17-19), S. cerevisiae (20) and transgenic plants (21, 22).
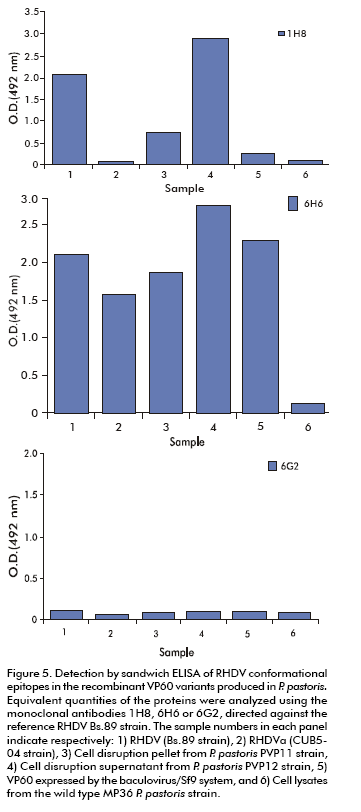
Analysis of conformational epitopes in the recombinant VP60 variants
The study of the antigenic profile was performed in the soluble and insoluble VP60 variants using a sandwich ELISA, immunodot, and monoclonal antibodies 1H8, 6H6, and 6G2 that recognize the conformational epitopes present in the RHDV native capsid (30). As capture antibodies we used hyperimmune sera raised against the classical AST/89 strain or against the Cuban RHDVa subtype. In these assays, the epitope recognized by mAb 1H8 (only present in assembled capsids) was detected in the RHDV Bs.89 classical strain and in the soluble VP60 purified by size exclusion chromatography from the PVP12 strain, with absorbance values of over 1.5 units. This epitope was also present in the VP60 obtained in insect cultured cells and was only slightly detected in multimers from the PVP11 P. pastoris strain. The epitope recognized by mAb 6H6 was detected in both recombinant proteins from P. pastoris. MAb 6G2 did not show reactivity in sandwich ELISA against these proteins due to the fact that it recognizes a buried epitope located at the N-terminus of VP60 in assembled capsids or protein aggregates. This epitope was only found in immunodot experiments. Similar results were obtained when using hyperimmune serum raised against CUB5-04 (Figure 5).
Assessment of purification and recovery strategies of recombinant VP60 VLPs from the Pichia pastoris PVP12 strain
We took into account that in our lab a significant amount of soluble, purified VP60 is commonly lost after concentration, sterile filtration and/or freezing-thawing procedures. In previous works with the recombinant Norwalk virus capsid (31), several attempts have led to the conclusion that variations in the pH used for disruption or purification processes, as well as the inclusion of preservatives such as sucrose, sorbitol and trehalose in the final aqueous suspension may enhance VLPs stabilization, decrease aggregation and achieve overall superior protein yields. We then tested a wide range of pH (from 3.0 to 8.0) and the excipients mentioned above to test their ability to inhibit the aggregation of VP60-VLPs under conditions known to induce aggregation and protein loss. In our experiment, the results showed changes in the chromatographic pattern (in sec-HPLC, using a TSK G-3000 column) in relation to the standard purification procedure previously established at pH 7.0. These changes were recorded at pH 5.0 and partially at 6.0. At pH 5.0 the recovery of VP60 was also somehow affected. At pH 8.0, following a 10-fold concentration and freezing/ thawing procedures, the total amount of protein was noticeably reduced. Interestingly, although recombinant multimers were recovered at pH 3.0, the experiment was discarded because of disturbances detected in the purification column, possibly due to aggregation. In contrast, an entire process conducted at pH 6.0 or 7.0 produced only moderate levels of protein loss, probably due to a reduction in aggregation events and to the stabilization of VLPs, which is suspected to be pH-dependent (Figure 6A). In all cases, the semiquantification of the VLPs was conducted by immunodot using VP60 from Sf9 cells as the standard. Furthermore, although differences found with the use of excipients were not specially marked, preliminary findings suggest that the inclusion of trehalose as a stabilizing agent is a useful tool in reducing protein loss (data not shown). VLPs integrity was evaluated with the use of monoclonal antibodies after the purification and stressing steps. In all cases, with a range of recognition degrees, the protein was detected by mAbs 1H8 and 6H6. Protein recognition was slightly affected in the final samples corresponding to pH 5.0, during the disruption and purification processes (Figure 6B).
Evaluation of the protective capacity of antibodies generated by recombinant VP60 from PVP11/PVP12 strains against the classical RHDV and against the Cuban RHDVa subtype
We further investigated the immunogenicity in rabbits of the recombinant VP60 variants obtained from P pastoris. The protective capacity of antibodies elicited with these antigens was also evaluated in vitro by hemagglutination inhibition assays, against both the classical RHDV Bs.89 isolate and the subtype isolated in Cuba. New Zealand rabbits were randomly distributed into seven experimental groups of 3-5 animals each and then immunized with the recombinant VP60 insoluble/soluble variants obtained from the PVP11 and PVP12 strains, with VP60 from insect culture cells, with a RHDV-inactivated vaccine and with a placebo. The recombinant antigens were emulsified in Montanide 888 and subcutaneously administered in two doses of 50 μg each on days 0 and 21 of the experiment, except for an additional group orally immunized with VP60 contained in the disruption pellet of PVP11, with three doses of 0.5 µg on days 0, 21 and 40. The RHDV-inactivated vaccine was subcutaneously applied as a single dose on day 0 of the experiment according to the manufacturers instructions. Anti-RHDV humoral immune responses were evaluated by a competition ELISA that assesses the ability of the problem serum to compete in binding to the RHDV with a polyclonal hyperimmune specific serum absorbed onto the solid phase.
All vaccinated rabbits elicited antibody levels that were able to compete in binding to the classical RHDV. Groups immunized with purified VP60 from the PVP12 strain or with the VP60 protein from insect cultured cells rapidly developed inhibition percentages of over 85% on day 14 of the experiment. Similar inhibition values were detected after day 40 post-immunization in rabbits subcutaneously injected with the PVP12 raw disruption supernatant or with the VP60 contained in the PVP11 cell debris fraction. These levels were detected after day 60 in animals vaccinated with the inactivated Cunipravac-RHDTM vaccine. The inhibition percentages were maintained until day 365 after immunization in animals inoculated with the PVP12 raw disruption supernatant and until day 638 (up to 21 months) in the rest of the groups. In contrast, rabbits immunized with the PVP11 disruption pellet by the oral route developed lower levels of specific antibodies, which were maintained for a shorter period of time (Table 1) although the antibodies were able to inhibit the in vitro hemagglutination of RHDV. Also, antibodies generated in vaccinated rabbits were able to compete in binding to the Cuban viral isolate CUB5-04 (RHDVa subtype). The inhibition values detected in these animals were above 85%, with the exception of the rabbits orally immunized with the cell disruption pellet from the PVP11 strain and with the Cunipravac-RHDTM inactivated vaccine (Table 2).
The ability of these antibodies to inhibit the in vitro hemagglutinating activity of the Cuban RHDVa isolate was also demonstrated (32). This is a key element that correlates with the in vivo protection against RHDV (21). All animals vaccinated with the recombinant variants of VP60 developed these high antibody levels. The highest hemagglutination inhibition titers (ranging from 1/2560 to 1/40960) were elicited in animals immunized with the purified VP60 from the PVP12 strain. In contrast, the sera of rabbits immunized with the viral inactivated Cunipravac-RHDTM vaccine, showed no hemmaglutination inhibition titers. This result agreed with previous reports showing variable degrees of efficacy against the RHDVa subtype (24, 33, 34) and evidenced that antibodies generated against the classical RHDV strains may not be necessarily able to fully protect rabbits against the RHDVa subtype. This issue was overcome by using the recombinant variants expressed in the two strains of P. pastoris.
In general, this report presents the characterization of two different variants of the recombinant VP60 capsid protein from RHDV expressed at useful levels in the P. pastoris yeast. Our experiments support the conclusion that the VP60 obtained from the PVP12 strain as a soluble multimer is the most attractive candidate so far for a simple and inexpensive scaled up production and implementation of a recombinant subunit vaccine against RHDV.
ACKNOWLEDGMENTS
The authors would like to acknowledge the assistance of Galina Moya Fajardo from the Physical-Chemistry Division of CIGB. Viviana Falcón and Ivón Menéndez from the Electron Microscopy department of the CIGB are also acknowledged for excellent assistance. PastorAlfonso and the technical staff from the National Center for Animal and Plant Health (CENSA) are recognized for their inestimable cooperation in the immunization experiments in rabbits. We would especially like to thank Dr. Lorenzo Capucci and the Institute Zooprofilattico Sperimentale della Lombardia e dell Emilia, Italy, for kindly providing inactivated the RHDV (Bs.89 strain) antigen and the monoclonal antibodies used.
REFERENCES
1. Liu SJ, Xue HP, Pu BQ, Quian NH. A new viral disease in rabbits. Anim Husb Vet Med 1984;16:253-5.
2. Marcato PS, Benazzi C, Vecchi G, Galeotti M, Della Salda L, Sarli G, et al. Clinical and pathological features of viral haemorrhagic disease of rabbits and the European brown hare syndrome. Rev Sci Tech 1991;10:371-92.
3. Ferreira PG, Costa-e-Silva A, Oliveira MJ, Monteiro E, Cunha EM, Aguas AP. Severe leukopenia and liver biochemistry changes in adult rabbits after calicivirus infection. Res Vet Sci 2006;80:218-25.
4. Alonso C, Oviedo JM, Martín-Alonso JM, Díaz E, Boga JA, Parra F. Programmed cell-death in the pathogenesis of rabbit hemorrhagic disease. Arch Virol 1998;143:321-32.
5. Parra F, Prieto M. Purification of a calicivirus as the causative agent of a lethal hemorrhagic disease in rabbits. J Virol 1990;64:4013-5.
6. Green KY, Ando T, Balayan MS, Berke T, Clarke IN, Estes MK, et al. Taxonomy of the caliciviruses. J Infect Dis 2000;181:322-30.
7. Xu ZJ, Chen WX. Viral haemorrhagic disease in rabbits: a review. Vet Res Commun 1989;13:205-12.
8. Cooke BD. Rabbit hemorrhagic disease: field epidemiology and the management of wild rabbit populations. Rev Sci Tech 2002;21:347-58.
9. Calvete C. Modeling the effect of population dynamics on the impact of rabbit hemorrhagic disease. Conserv Biol 2006;20:1232-41.
10. Gregg DA, House C, Meyer R, Berninger M. Viral haemorrhagic disease of rabbits in Mexico: epidemiology and viral characterization. Rev Sci Tech 1991;10: 35-451.
11. Toledo M, Seoane G, Serrano E, Encinosa A, Delgado C, González S, et al. Estudio epizootiológico y control de la enfermedad hemorrágica viral del conejo en la República de Cuba. Rev Cub Cienc Vet 1995;24:5-9.
12. Neilan JG, Lu Z, Ward G, Lubroth J, Rock DL, Kutish GF. The genome of rabbit hemorrhagic disease virus (Iowa 2000) from North America. EMBL/GenBank/ DDBJ databases direct submission 2000.
13. Campagnolo ER, Ernst MJ, Berninger ML, Gregg DA, Shumaker TJ, Boghossian AM. Outbreak of rabbit hemorrhagic disease in domestic lagomorphs. J Am Vet Med Assoc 2003;223:1151-5.
14. McIntosh MT, Behan SC, Mohamed FM, Lu Z, Moran KE, Burrage TG, et al. A pandemic strain of calicivirus threatens rabbit industries in the Americas. J Virol 2007;4:96.
15. Argüello-Villares JL. Viral haemorrhagic disease of rabbits: vaccination and immune response. Rev Sci Tech 1991;10:459-80.
16. Boga JA, Casais R, Marin MS, Martin- Alonso JM, Carmenes RS, Prieto M, et al. Molecular cloning, sequencing and expression in Escherichia coli of the capsid protein gene from rabbit haemorrhagic disease virus (Spanish isolate AST/89). J Gen Virol 1994;75:2409-13.
17. Laurent S, Vautherot JF, Madelaine MA, Le Gall G, Rasschaert D. Recombinant rabbit hemorrhagic disease virus capsid protein expressed in baculovirus self-assembles into virus like particles and induces protection. J Virol 1994;68:6794-8.
18. Marín MS, Martín-Alonso JM, Pérez- Ordoyo García LI, Argüello-Villares JL, Casais R, Venugopal K, et al. Immunogenic properties of rabbit haemorrhagic disease virus structural protein VP60 expressed by a recombinant baculovirus: an efficient vaccine. Virus Res 1995;39:119-28.
19. Gromadzka B, Szewczyk B, Konopa G, Fitzner A, Kesy A. Recombinant VP60 in the form of virion-like particles as a potential vaccine against rabbit hemorrhagic disease virus. Acta Biochim Pol 2006;53:371-6.
20. Boga JA, Martín-Alonso JM, Casais R, Parra F. A single dose immunization with rabbit haemorrhagic disease virus major capsid protein produced in Saccharomyces cerevisiae induces protection. J Gen Virol 1997;78:2315-18.
21. Fernández-Fernández MR, Mourino M, Rivera J, Rodríguez F, Plana-Duran J, García JA. Protection of rabbits against rabbit hemorrhagic disease virus by immunization with the VP60 protein expressed in plants with a potyvirus based vector. Virology 2001;280:283-91.
22. Gil F, Titarenko E, Terrada E, Arcalis E, Escribano JM. Successful oral prime-immunization with VP60 from rabbit haemorrhagic disease virus produced in transgenic plants using different fusion strategies. Plant Biotechnol 2006;4:135-43.
23. Morenweiser R. Downstream processing of viral vectors and vaccines. Gene Ther 2005;12:S103-10.
24. Capucci L, Fallacara F, Grazioli S, Lavazza A, Pacciarini ML, Brocchi E. A further step in the evolution of rabbit hemorrhagic disease virus: the appearance of the first consistent antigenic variant. Virus Res 1998;58:115-26.
25. Grazioli S, Agnoletti F, Scicluna MT, Masoero N, Guercio A, Fallacara F, et al. Rabbit haemorrhagic disease virus (RHDV) subtype A (RHDVa) is replacing the original strain in some Italian regions. In: Brocchi E, Lavazza A. (eds), Fifth International Congress of the European Society for Veterinary Virology, Brescia, Italy, p. 202-3.
26. Hardy E, Martinez E, Diago D, Diaz R, Gonzalez D, Herrera L. Large-scale production of recombinant hepatitis B surface antigen from Pichia pastoris. J Biotechnol 2000;77:157-67.
27. Sibilia M, Boniotti MB, Angoscin P, Capucci L, Rossi C. Two independent pathways of expression lead to self-assembly of the rabbit hemorrhagic disease virus capsid protein. J Virol 1995;69:5812-5.
28. Rodríguez M, Rubiera R, Penichet M, Montesinos R, Cremata J, De la Fuente J. High level expression of the B. microplus Bm86 antigen in the yeast Pichia pastoris forming highly immunogenic particles for cattle. J Biotechnol 1994;33:135-46.
29. García-García JC, Montero C, Redondo M, Vargas M, Canales M, Boué O, et al. Control of ticks resistant to the immunization with Bm86 in cattle vaccinated with the recombinant antigen Bm95 isolated from the cattle tick, Boophilus microplus. Vaccine 2000;18:2275–87.
30. Capucci L, Frigoli G, Roenshold L, Lavazza A, Brocchi E, Rossi C. Antigenicity of the rabbit hemorrhagic disease virus studied by its reactivity with monoclonal antibodies. Virus Res 1995;37:221-38.
31. Kismann J, Ausar SF, Foubert TR, Brock J, Switzer M, Detzi ED, et al. Physical Stabilization of Norwalk virus-like particles. J Pharm Sci 2008;7:4208-18.
32. Farnós O, Fernández E, Chiong, M, Parra F, Joglar M, Méndez L, et al. Biochemical and structural characterization of RHDV capsid protein variants produced in Pichia pastoris: Advantages for immunization strategies and vaccine implementation. Antiviral Res 2009;81:25-36.
33. Schirrmeier H, Reimann I, Kollner B, Granzow H. Pathogenic, antigenic and molecular properties of rabbit haemorrhagic disease virus (RHDV) isolated from vaccinated rabbits: detection and characterization of antigenic variants. Arch Virol 1999;144:719-35.
34. Lavazza A, Cerrone A, Agnoletti F, Perugini G, Fioretti A, Botti G, et al. An update on the presence and spreading in Italy of rabbit haemorrhagic disease virus and of its antigenic variant RHDVa. In: Proc., 8th World Rabbit Congress2004 September 7-10, Mexico, Puebla City, p. 62-68.
*This work received the Award of the National Academy of Sciences of Cuba for the year 2008.
Omar Farnós. Animal Biotechnology Division, Center for Genetic Engineering and Biotechnology, CIGB. Ave. 31 / 158 and 190, Cubanacán, Playa, PO Box 6162, CP 10600, Havana, Cuba. E-mail: omar.farnos@cigb.edu.cu