My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.26 no.4 La Habana Oct.-Dec. 2009
REPORT
Association between enterovirus infection and type 1 diabetes: new findings from Cuban studies
Luis Sarmiento1, Eduardo Cabrera2, Oscar Díaz2, Magile Fonseca1, Gisela Molina2, Ileana Cuba2, Pedro Más1, Lai Heng1,Oscar Díaz2, Sonia Resik1
1 Department of Virology, “Pedro Kourí” Tropical Medicine Institute, IPK
2 Department of Immunology of Diabetes, National Institute of Endocrinology, Havana, Cuba
ABSTRACT
Enteroviruses have been examined for their possible role in the etiology of type 1 diabetes mellitus (T1DM) for nearly 40 years; however, the evidence remains inconclusive so far. This article provides new findings from Cuban studies suggesting the involvement of enteroviruses, either with the clinical T1DM onset or the pre-clinical stages of the β- cell autoimmunity in populations with low incidence of diabetes and high enteroviral circulation. Potential mechanisms of pathogenesis such as direct β- cell lysis and molecular mimicry are also discussed.
Keywords: type 1 diabetes mellitus, autoimmunity, autoantibodies, enterovirus
INTRODUCTION
Type 1 diabetes mellitus (T1DM) results from autoimmune destruction of pancreatic islet β cells. One of its major hallmarks is the presence of islet cell autoantibodies (ICA), present in over 90% of cases and defined by indirect immunofluorescence or by reactivity to three molecularly defined islet autoantigens: insulin autoantibodies (IAA), glutamic acid decarboxylase autoantibodies (GADA) and insulinoma-associated-antigen-2 autoantibodies (IA-2A) (1).
Although the discovery of islet cell autoantibo-dies in 1974 shaped thinking on the pathogenesis of T1DM, leading to its classification as autoimmune in nature, the etiology of the disease remains unknown (1). Disease-associated genes are clearly important, but numerous studies, especially those on monozygotic twins, show that heritable factors alone account for only 30-50% of disease susceptibility. Therefore, attention has been focused on environmental factors that might trigger and/or accelerate disease (2).
Several studies have suggested that enteroviruses are one of the major environmental agents contribu-ting to the increased T1DM incidence observed over the last few decades. However, the majority of those studies have been carried out in countries with either exceptionally high or intermediate incidence of T1DM (3). The purpose of this article is to provide further support for the association between enterovirus infections and T1DM in a population with low incidence of T1DM (4) and high circulation of enteroviruses (5).
MATERIALS AND METHODS
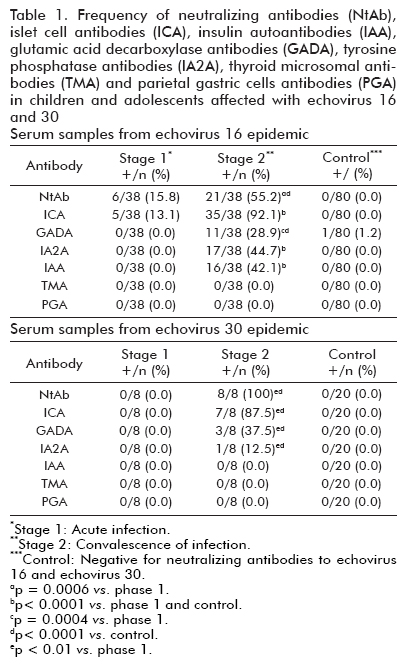
Type 1 diabetes islet associated antibodies in subjects infected by echovirus 16 and echovirus 30
We obtained 38 serum samples from children and adolescents in the acute (five/five to six days after the beginning of the infection) and convalescent (30/29 to 35 days after the beginning of the infection) stage from a large-scale echovirus 16 (E16) meningitis epidemic throughout the country in the year 2000. In addition, eight paired sera randomly selected from echovirus 30 (E30) meningitis epidemic occurred in Cuba in 2001 were included in the study. Sera from infected children were matched with at least two control sera (selected from 300 healthy children) for age, sex, date of collection, and location. All subjects involved in the study had no family history of diabetes and were screened for diabetes-associated antibodies (ICA, GADA, IAA,IA2A) and neutralizing antibodies against different enterovirus serotypes (Echoviruses: E4, E6, E9, E11,E16, E30 and Coxsackieviruses: CVA9, CVB1, CVB2,CVB3, CVB4, CVB5, CVB6) using international work-shop-validated assays (6, 7).
Thyroid Microsomal Antibodies (TMA) and Parietal Gastric Cells Antibodies (PGA) measurement were only carried out in subjects infected in order to show the specific pancreatotoxic activity of this infection (6,7).
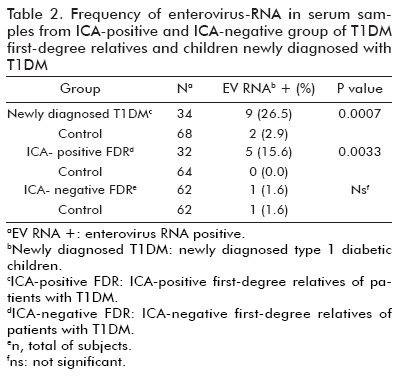
Enterovirus RNA in serum of children with newly diagnosed T1DM and islet cell autoantibody-positive subjects
Enterovirus genome detection was carried out using a Reverse Transcriptase-Nested Polymerase Chain Reaction assay of serum specimens from: (I) 34 children at T1DM onset, (II) 32 ICA positive T1DM first-degree relatives, (III) 31 ICA negative T1DM first-de-gree relatives and (IV) 194 healthy subjects who were verified negative for ICA and had no family history of diabetes. Two control sera were matched with each serum from group I, II and III for age, sex, date of collection and location. Urianalysis for the detection of ketones was performed on samples from children with newly diagnosed T1DM at initial presentation, prior to the administration of insulin. Severe diabetic ketoacidosis was defined as pH, 7.1 and bicarbonate, 5 mmol/L (8).
Antibodies to human glutamic acid decarboxylase in sera from enterovirus-immunized rabbit
Enterovirus antisera were prepared by immunizing New Zealand white rabbit with enteroviral antigens obtained from 17 strains of enterovirus serotypes most frequently isolated during the last 30 years in Cuba (Echoviruses: E1, E2, E3, E4, E6, E9, E11, E16, E30 and Coxsackieviruses: CVA7, CVA9, CVA16, CVA24,CVB1, CVB3, CVB5, CVB6). Antigens from moninoculated cells where utilized as negative controls. Rabbit sera were screened for three human islet autoantigens: GADA, IAA and IA2A using international workshop-validated assays (9).
Statistical Analysis
Statistical differences of frequencies among groups were analyzed by the Chi-square or Fisher’s exact test. A p value of less than 0.05 was considered to be statistically significant.
RESULTS
Type 1 diabetes islet associated antibodies in subjects infected by echovirus 16 and echovirus 30
Notably, in the convalescent (but not the acute stage of the infection), ICA seroconversion was demonstrated, as well as the emergence of IAA and GADA and IA2A concentrations. The islet cell autoimmunity is clearly epidemic and infection-associated, since no serum samples from uninfected subjects serologically negative for neutralizing antibodies to E16 and E30 had ICA, and only one was GADA positive (similar to reported background population prevalence of GADA). The presence of T1DM islet associated antibodies in subjects does not seem to reflect a general response of autoantibodies since none of the serum samples obtained during the infection were positive for TMA or PGA (Table 1).
Only a patient who developed pancreatic autoimmunity and T1DM after E30 epidemic was reported. After the meningitis episode this patient gained 5 kg of weight in the subsequent 6 months. Eight months after the infection the subject started to show symptoms suggesting diabetes such as polyuria, polydipsia, increased appetite and weight lost (3 kg). At this period, fasting and postprandial blood glucose concentrations were 12.1 and 18.9 mmol/L, respectively. ICA and IA2A were detected post-infection in the adolescent with newly diagnosed T1DM. In addition, the analysis of neutralizing antibodies to enterovirus serotypes most frequently isolated during the last 30 years in Cuba showed high titres of neutralizing antibodies only to E30 (1:80) suggesting that this virus was the agent who caused the aseptic meningitis. However, the adolescent was found to carry the HLA DR 15(DR2)/DR7 and DQ 2/DQ6 (DQ1) genotype which is not associated with an increased risk for T1DM.
Enterovirus RNA in serum of children with newly diagnosed T1DM and islet cell autoantibody-positive subjects
The rate of enterovirus sequences detection in newly diagnosed T1DM children (9/34, 26.5%) and ICA-positive first-degree relatives (5/32, 15.6%) was significantly higher than their corresponding matched controls (2/68, 2.9%, p = 0.0007 and 0/64, 0.0%, p = 0.0033, respectively). There was no significant difference in the frequency of enterovirus RNA detection between ICA-negative T1DM first degree relatives and their matched-controls (1.6%, 1 out of 62 vs. 1.6%, 1 out of 62) (Table 2).
Interestingly, subjects at T1DM onset with occurrence of enterovirus RNA were more likely to suffer severe diabetic ketoacidosis (66.6%, 6 out of 9 vs. 20.0%, 5 out of 25; p = 0.0328). However, the presence of ketosis and mild diabetic ketoacidosis at diagnosis of T1DM was equally distributed among positive and negative subjects for enterovirus RNA.
Antibodies to human glutamic acid decarboxylase in sera from enterovirus-immunized rabbit
Our results revealed the presence of antibodies to β-cell autoantigen glutamic acid decarboxylase (GAD 65) in the hyperimmune rabbit sera immunized with E9, E11, E16, E30, CVB1, CVB3 and CVB5. IAA or IA2A were not detected in any of the sera studied. Sera from rabbits immunized with negative control antigen preparations were negative for the three islet-related autoantibodies.
RELEVANCE OF THE STUDY
The opportunistic study of enterovirus epidemics in Cuba and their relationship to islet cell autoimmunity and T1DM showed that various types of enterovirus were associated with a varying degree in the acute development of islet cell autoimmunity. Thus, our fin-dings provide the first evidence of association of E16 and E30 with the appearance of pancreatic autoimmunity worldwide and suggest that the induction of islet cell associated antibodies seems to occur right after episodes of enterovirus infections.
Moreover, the epidemic echovirus strains showed subtle differences in the nature of the islet cell autoimmunity they induced. The isolation of viruses with proven but varying potency in their ability to induce islet autoimmunity in man suggests that there is heterogeneity of the immune response against β-cell antigens after echovirus infection.
Our results indicate a strong temporal relationship between islet autoantibodies and exposure to echovirus, and prompt further important questions, such as the longevity of post-echovirus islet autoimmunity or whether the autoimmune response is transient and wanes. Although, no follow-up of these children for diabetes has been done, we predict that the post-echovirus islet autoimmunity detected in the original epidemics will not show persistence, and it is unlikely to cause T1DM onset. This assertion is based on the fact that T1DM incidence in Cuba is low (3/100.000) (4). Thus, while β-cell autoimmunity may be triggered in anyone, the HLA genes may be crucial for progression from autoimmunity to T1DM.
It is worth noting that the present study has shown a significantly higher frequency of enterovirus RNA in serum from T1DM children at the onset of disease, as well as in ICA positive first-degree relatives compa-red to healthy control subjects and ICA negative first-degree relatives. It suggests that enterovirus infection is associated with T1DM onset and β-cell autoimmunity in subjects at risk of developing T1DM. However, the emergence of pancreatic autoimmunity and T1DM after meningitis infection by E30 in adolescent ca-rrying specific low-risk genotypes for T1DM suggests that enterovirus can contribute to the development of T1DM in subject with a low genetic risk.
The high occurrence of severe diabetic ketoacidosis at onset in subjects with enterovirus RNA-positive suggests that enteroviruses may cause direct cytolytic damage against β-cells rather than a chronic autoimmune process. However, destruction of pancreatic β-cells by a virus-directed immune response against infected β-cells should not be excluded since an association between the presence of enterovirus infection and the occurrence of islet related autoantibodies without diabetes was also found. Indeed, the presence of GADA in sera from enterovirus-immunized rabbits suggests the existence of common determinants among enterovirus serotypes and the human β-cell autoantigen GAD 65 and support the hypothesis that molecular mimicry is a possible mechanism involved in the development of β-cell autoimmunity induced by enteroviruses.
CONCLUSIONS
Our findings suggest that enterovirus infections are associated with T1DM onset and β-cell autoimmunity in populations with low incidence of diabetes and high enteroviral circulation and are in agreement with the idea that the molecular mimicry and direct cytolytic damage against β -cells are possible mechanisms involved in the development of β -cell autoimmunity induced by enteroviruses.
If enteroviruses associate with T1DM: How could a low incidence of T1DM coexisting with a high circulation of enteroviruses be explained in the Cuban situation?
Two different, yet non-mutually, exclusive hypotheses could explain this paradox. Firstly, the genetic T1DM susceptibility background within populations (Differences in high risk HLA prevalence and other genetic loci). This points out the possibility that enterovirus infection could be “more dangerous” with regard to the diabetogenic potential in those populations carrying specific high-risk genotypes. Probably the low frequency of T1DM susceptibility genes in Cuban population (10) could justify the low incidence of T1DM although enterovirus infections are highly prevalent in Cuba (11). Secondly, the so-called “Hygiene Hypothesis” according to which reduced rate of infection contributes to increased T1DM incidence (12). Thus, the more the population is exposed to enteroviruses (e.g. Cuba) the higher the probability to be immunized against subsequent “diabetogenic” strain exposition. The other way around also occurs in the European population in which a low frequency of enterovirus infections in the background population would increase the susceptibility of young children to the diabetogenic effect of such viruses.
In conclusion, although direct proof for enteroviruses as causative agents for T1DM is difficult to verify, our findings support the role of enterovirus in the pathogenesis of T1DM and its confirmation might contribute to the development of preventive strategies in the T1DM.
REFERENCES
1. Haller MJ, Atkinson MA, Schatz D. Type 1 diabetes mellitus: Etiology, presentation, and management. Pediatr Clin N Am 2005;52:1553-78.
2. Rewers M, Norris J, Dabelea D. Epidemiology of type 1 diabetes. In: George S Eisenbarth, ed. Immunology of type 1 diabetes. New York: Eurekah.com and Kluwer Academic/ Plenum Publishers;2004, p. 219-46.
3. Sarmiento L. Enterovirus - Induced Type 1 Diabetes. In: Luis Sarmiento-Pérez and Eduardo Cabrera-Rode (editors). Enterovirus infections and type 1 Diabetes Mellitus. New York: Nova Science Publishers; 2007,p.125-65.
4. Collado F, Díaz O, Hernández I. Epidemiologic behaviour of insulin dependent diabetes mellitus in children younger than 15 years of old. Cuba, 1990-1993. Rev Cub Endocrinol 1993;8:119-25.
5. Sarmiento L. Enteroviral meningitis and emergence of rare enterovirus types: Cuban experience”, In: Strong P (editor), Focus on Meningitis Research, (Nova Science Publishers, New York), 2004, p.1-14.
6. Cabrera-Rode E, Sarmiento L, Tiberti C, Molina G, Barrios J. Hernandez D, et al. Type 1 diabetes islet associated antibodies in subject infected by echovirus 16. Diabe-tologia 2003; 46(10):1348-53.
7. Cabrera-Rode E, Sarmiento L, Molina G, Pérez C, Arranz C, Galvan J, et al. Islet cell related antibodies and Type 1 diabe-tes associated with echovirus 30 Epide-mic: A case report. J Med Virol 2005;76(3): 373-7.
8. Sarmiento L, Cabrera-Rode E, Lekuleni L, Cuba I, Molina G, Fonseca M, et al. Occurrence of enterovirus RNA in serum of children with newly diagnosed type 1 diabetes and islet cell autoantibody-positive subjects in a population with incidence of type 1 diabetes. Autoimmunity 2007; 40(7):540-5.
9. Sarmiento L, Cabrera-Rode E, Mas P, Díaz-Horta O. Antibodies to human glutamic acid decarboxylase in sera from enterovirus-Immunized Rabbit. Autoimmunity 2007;40(7):546-7.
10. Alegre R, Moscoso J, Martinez-Laso J, Martin-Villa M, Suarez J, Moreno A, et al. HLA genes in Cubans and the detection of Amerindian alleles. Mol Immunol 2007;44:2426-35.
11. Sarmiento L. Human enterovirus in-fections. In: Luis Sarmiento-Pérez and Eduardo Cabrera-Rode (editors). Entero-virus Infections and Type 1 Diabetes Mellitus. New York: Nova Science Publishers; 2007, p.93-123.
12. Hintermann E , Christen U. Viral infection - A cure for type 1 diabetes? Curr Med Chem 2007;14:2048-52.
Luis Sarmiento. Department of Virology, “Pedro Kourí” Tropical Medicine Institute, IPK, Havana, Cuba. E-mail: sarmiento@ipk.sld.cu