My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.27 no.3 La Habana July-Sept. 2010
RESEARCH
Characterization of the immune response generated by intramuscular administration of nucleocapsid and surface antigens of Hepatitis B
Caracterización de la respuesta immune generada por administración intramuscular de los antígenos de superficie y nucleocápsida del virus de la Hepatitis B
Yadira Lobaina1, Daymir García1, Diane Rodríguez2, Yanaisa La O2, Julio C Aguilar1
1 Hepatitis B Department, Biomedical Research Unit,
2 Animal Facilities, Center for Genetic Engineering and Biotechnology, CIGB Ave. 31 / 158 and 190, Cubanacán, Playa, PO Box 6162, Havana, Cuba
ABSTRACT
Multiple immunization studies have been carried out in mice and humans evaluating formulations based on the surface antigen of Hepatitis B virus (HBV) in combination with different adjuvants. Such studies explore the use of new vaccine candidates for the prevention or therapy of hepatitis B infection. We have previously reported the preclinical and clinical development of a novel nasal formulation containing the surface and nucleocapsid antigens of the HBV. The present work explores the immune response elicited in mice when this formulation was administered adsorbed in alum by intramuscular route. Mice were immunized with different antigenic proportions and the immune response was evaluated by ELISA in order to study the total IgG and the main IgG subclasses in sera. The lymphoproliferation capacity of spleen cells stimulated with each antigen was assayed. The results indicate that the formulation is very immunogenic when administered by intramuscular route, eliciting potent humoral immune responses. We also demonstrated that the inclusion of the nucleocapsid antigen in the formulation favored a deviation to a Th1 pattern of response, showed at IgG subclasses and lymphoproliferation results. The evaluation of this candidate using the parenteral route suggests the potential reduction of the number of doses and the quantity of antigen per dose with respect to the conventional HBV vaccine. The possible use of this formulation as prophylactic or therapeutic vaccine candidates was also discussed.
Key words: HBV, parenteral, vaccine.
RESUMEN
Múltiples estudios de inmunización se han llevado a cabo, tanto en ratones como en humanos, evaluando formulaciones basadas en el antígeno de superficie del virus de la hepatitis B (VHB) en combinación con diferentes adyuvantes. Estos estudios exploran el empleo de nuevos candidatos vacunales en la prevención o terapéutica de la infección por hepatitis B. Nuestro grupo ha reportado previamente el desarrollo preclínico y clínico de una novedosa formulación para uso nasal que contiene los antígenos de la superficie y nucleocápsida del VHB. El presente trabajo estudia la respuesta inmune generada en ratones cuando dicha formulación es administrada adyuvada en alúmina por la ruta intramuscular. Con este objetivo se inmunizaron ratones con diferentes proporciones de antígenos y la respuesta de anticuerpos IgG y subclases de IgG inducida en suero se evaluó por ELISA. Adicionalmente se midió la capacidad de linfoproliferación de esplenocitos estimulados con cada antígeno. Los resultados obtenidos indican que la formulación administrada por ruta intramuscular es muy inmunogénica, generando una potente respuesta humoral. Además se demuestra que la inclusión en la formulación del antígeno de la nucleocápsida favorece la desviación hacia un patrón de respuesta Th1, lo cual se muestra a nivel de las respuestas de subclases de IgG y linfoproliferación. La evaluación de este candidato por la ruta parenteral sugiere una potencial reducción del número de dosis, así como la cantidad de antígeno por dosis con respecto a la vacuna anti-VHB convencional. Adicionalmente se discute el posible empleo de esta formulación como candidato vacunal profiláctico o terapéutico.
Palabras clave: VHB, parenteral, vacuna.
INTRODUCTION
The infection by Hepatitis B Virus (HBV) remains as an important health problem at a global scale in spite of the existence of very effective vaccines since the 1980´s. Two billion people alive today show evidence of past or current infection and more than 350 million people are persistently infected. The state of chronicity correlates with an increased risk of developing liver cirrhosis, hepatocellular carcinoma and other complications like portal hypertension and liver failure. As a consequence one million people die each year worldwide (1)
HBV is a unique DNA virus that replicates in hepatocytes, such cells produce and secrete Hepatitis B surface, e antigens (HBsAg and HBeAg, respectively) and intact virions (HBV-DNA) in the circulation (2). The HBsAg and the core antigen (HBcAg) are the main structural antigens of HBV. Both antigens constitute potent immunogens for experimental animals as well as in humans acutely infected with HBV (3). HBsAg constitutes the main protective antigen and the basis of all prophylactic available vaccines, which has been proven over the last 20 years to be safe and effective against HBV infection (1). However, there remains a need for development of more potent hepatitis B vaccines, both for use as prophylactic vaccines in poor responders to the current HBV vaccines (4) and for use in therapeutic HBV vaccination (5, 6).
In the therapeutic scenario the results coming from multiple clinical trials using candidates based on HBsAg as unique antigen in the formulation had showed that more powerful antigen formulations and novel adjuvant strategies are required to overcome the state of unresponsiveness of chronic patients (6, 7). Numerous studies in HBV-transgenic mice pointed out that the HBcAg is an antigen of choice to increase the number of epitopes and the variety of the immune response, even when used in combination with other antigens such as HBsAg (8, 9). These studies also confirmed the prominent role of HBcAg as a pro-Th1 antigen (10).
Our group has been working in the design of a candidate based in the combination of HBsAg and HBcAg antigens (11-13). Our strategy took advantage of the unique immunological properties of the HBcAg (14) and also explored new routes of administration and combinations of routes as a way to develop more potent and wider immune responses (12, 15).
On the other hand, HBV vaccines capable of circumventing the non-responder/inadequate responder status of some vaccinees remain to be a specific requirement for the prophylactic scenario.
It is reported that about 4% of vaccinated newborns with conventional HBV vaccines do not reach a protective antibody level (³ 10 mIU/mL) at seroconversion and 0.4% is a non-responder even after receiving a fourth dose of vaccine (true non-responders (TNR)); while 3.6% achieved an antibody level ³ 10 mIU/mL (slow responders (SR)) only when reboosted with a fourth dose. Among children achieving protective antibody levels at seroconversion, low responders (LRs) had an antibody level between 11 and 40 mIU/mL at the seroconversion (5% of the total population), while responders (Rs) had an antibody level ³ 40 mIU/mL (16, 17).
Otherwise, in healthy adults a variegated antibody response following standard HBV vaccination is observed, with 5-10% showing no response. It is suggested that non-responsiveness may be associated with antigen-specific HLA determined deficiency in the T-cell repertoire and not only with defective antigen presentation or HLA class II affinity for hepatitis surface antigen (HBsAg) derived peptide (18).
In the present work we evaluated the immunological response elicited in BalbC mice when administered with a formulation containing HBV surface and core antigens adjuvated in alum by intramuscular route and we also explored different proportions of the antigens in the formulation. This new approach could be used in the prophylaxis of non-responder/inadequate responder populations, and also in the development of therapeutic strategies.
MATERIALS AND METHODS
Antigens
HBsAg was produced to more than 95% purity at the Center for Genetic Engineering and Biotechnology production facilities (CIGB, Havana, Cuba) as a component of the commercial HBV prophylactic vaccine, Heberbiovac-HB. HBsAg for this vaccine is expressed and purified from the yeast Pichia pastoris. HBcAg was expressed in E. coli strain W3110, which had previously been transformed with a plasmid containing the entire core antigen gene under the control of a tryptophan promoter. The resulting HBcAg had a purity of >95% and measured 28 nm as characterized by electron microscopy. The formulation containing both antigens was obtained by simple mixture in phosphate saline buffer (PBS) (0.1 mol/L NaCl, 2 mmol/L KCl, 10 mmol/L, Na2HPO4, 1 mmol/L KH2PO4, pH 7.2).
Immunization schedules
An immunization schedule was carried out using groups of 10 BalbC mice, females, from 8 to 12 weeks old each. The intramuscular (i.m.) immunization route was used, administering a volume of 100 µL per animal in the quadriceps muscle. Formulations using different proportions (2:1, 1:1, 1:2, 0.5:0.5, respectively) of the simple mix of the 2 µg HBsAg and (x) µg HBcAg antigens adjuvated in 0.5 mg/mL of alum, were administered. An immunization group exploring the reduction of the number of doses with the 1:1 proportion was also included. Groups immunized with each antigen separately in alum, Heberbiovac-HB commercial vaccine, and an alum placebo group was also used. In general the doses were administered the days 0, 15 and 30. The sera were collected 10 days after each dose via retro-orbital plexus.
Another set of experiments were carried out evaluating, in similar immunization schedules, the former formulation but exploring minor dose of the HBsAg in the mix (between 2 to 0.25 µg). The schedule of dose used in this study was 0, 30 and 60 days. All experiments were conducted in accordance with institutional guidelines.
Enzyme-linked immunosorbent assay for determining total and subclasses IgG response
Specific IgG against each antigen in the formulation were evaluated by an enzyme-linked immunosorbent assay (ELISA). Briefly, high binding plates (Costar, USA) were coated with 100 µL of the specific antigen at 5 µg/mL in coating buffer (11 mM Na2CO3, 35 mM NaHCO3, pH 9.6) an incubated overnight at 4 ºC. Plates were blocked with 2% (w/v) skimmed milk in phosphate saline buffer (0.1 M NaCl, 2 mM KCl, 10 mM Na2HPO4, 1 mM KH2PO4, pH 7.2) (PBS) for 1 h at 37 ºC. Subsequently, the plates were incubated with the serum samples diluted with 1% (w/v) skimmed milk, 1% (v/v) Tween 20 in PBS, for 2 h at 37 ºC. The anti-mouse IgG peroxidase conjugate (SIGMA, USA) was incubated 1 h at 37 ºC. Subsequently the plates were incubated with the substrate solution (52 mM Na2HPO4, 25 mM citrate, 1 mg/mL OPD, 0.1% (v/v) H2O2) for 15 min at room temperature. Washes with 0.05% (v/v) Tween 20 in PBS solution were carried out between each step three to five times. The reaction was stopped with 3M H2SO4 solution. Finally the plates were read to 492 nm in a microtiter plate reader (Sensident Scan, Merck).
The IgG subclass evaluations were done using the kit ISO-2 Mouse Monoclonal Antibody Isotyping Reagents and following the manufactured recommendations (SIGMA, USA).
Positive samples for antibody titers were detected using cut off values of twice the optical density (OD) of negative controls (preimmune serum). Each sample was analyzed using an Excel program able to interpolate the OD values on the standard curve consisted in a pool of hiperimmune sera of known titers. This standard curve was included in each individual plate. Finally, the obtained results of total IgG and subclasses were represented as logarithm of geometric mean of titer (GMT) for each group of treatment (with a confidence interval of 95%).
Lymphoproliferation assays
Unfractionated splenocytes individual suspensions were prepared per mice and incubated (105 cells/well) for 4 days at 5% CO2 and 37 ºC in presence of rHBsAg expressed in Pichia pastoris (provided by Heber- Biotec) or rHBcAg (CIGB, Cuba) at 0.1 µg/mL. Cells incubated with concanavalin A (SIGMA, USA) was used as positive control and cells incubated with complete RPMI medium were employed as negative controls. All proliferation assays (LPA) were performed in triplicate in 96-well plates and (3H)thymidine (3HTdR; 0.5 µCi/well; specific activity, 2.0 Ci/mmol/L; Amersham International, Buckinghamshire, UK) was added 12 h before harvesting. Results are expressed as stimulation index (SI), which represents the ratio between the mean of scintillation per minute (spm) obtained in the presence and absence of antigen. SI values ³ 3 were regarded as positive.
Statistical procedures
The statistical treatment of titers was carried out using the F test to evaluate variance homogeneity followed by the T test in case of two group´s comparisons. For multiple comparisons among groups, the results were analyzed using the program GraphPad Prism version 4.00 (GraphPad Software, USA). One-way Anova and Newman Keuls tests were run for parametric analyses, and Kruskal Wallis and Dunn´s tests for the non parametric ones.
RESULTS
The present work describes the first immunological evaluation of a formulation containing the HBsAg and HBcAg adjuvated in alum administered by intramuscular route. It was designed as a study evaluating different doses and proportions of the antigens in the formulation and also studying a possible reduction of the number of doses and the influence of these parameters in the humoral immune responses elicited in BalbC mice. The humoral response was evaluated measuring total IgG titers specific for each antigen in the formulation. The main IgG subclasses were evaluated specifically against the HBsAg, which constitutes the main protective antigen of the current preventive vaccine. The lymphoproliferative response against both antigens in total spleen cells was evaluated in some groups.
Antigen-specific humoral immune response in sera
HBsAg-specific IgG response in sera
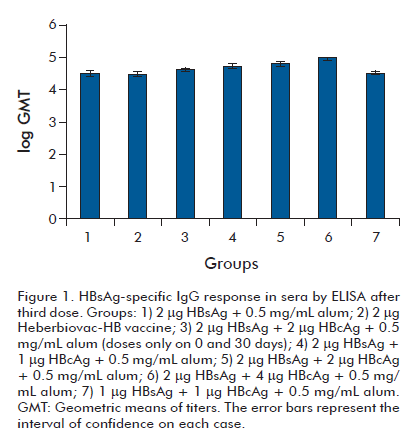
HBsAg-specific total IgG response was evaluated in sera after each dose. Seroconversion was not observed in any tested serum ten days after the first dose; this behavior is typical of the HBsAg, that needs the T-cell cooperation for the production of a specific response in sera. The IgG response obtained after the third dose was high for all groups (GMT ³ 104) (Figure 1).
In general, a trend to elicit a superior anti-HBsAg IgG response was observed for the groups that included HBcAg in the formulation when compared with the control group in which the commercial vaccine was employed (group 2). This trend became statistically significant in the case of the groups immunized with the variants (1:1) and (1:2) (groups 5 and 6, (p 0.05) and (p 0.001), respectively). The response obtained for the group that received only two doses of the formulation (1:1) with 2 µg of each antigen (group 3) did not differ statistically from the response generated by the control group. Similar behavior was observed for the response elicited by the group immunized with the proportion (1:1) with 1 µg of each antigen (group 7). The group immunized with the proportion 1:2 (group 6) generated the higher anti- HBsAg specific IgG response, differing from the rest, with the exception of group 5.
In the experiment exploring minor doses of the HBsAg in the mixture the HBsAg-specific IgG responses after three doses were similar (p > 0.05) for all immunized groups with 2 or 1 microgram of each formulation (HBsAg in alum or the formulation containing HBsAg and HBcAg in alum) (Figure 2). These results indicated that using the formulation containing HBsAg in alum (resembling the anti-HBV commercial available vaccines) could reduce the dose to 1 mg maintaining a similar IgG response; however there was a significant reduction (p > 0.05) in the anti-HBsAg IgG titer in the group containing 0.5 µg of HBsAg (group C). The results of HBsAg-specific IgG response after three doses evidenced that the adjuvant effect of 2 µg of HBcAg allows a fourfold reduction in the HBsAg dose without affecting the IgG response compared to groups A and E.
HBcAg-specific IgG response in sera
For both experiments the HBcAg-specific total IgG response were evaluated in sera after each dose. Differing from the HBsAg-specific IgG response, 10 days after the first dose seroconversion and high titers (GMT in levels close to 104) were observed in all tested serum. The IgG responses obtained after the third dose were high for all groups (GMT ³ 105) (data not shown).
Response of IgG subclasses
The specific IgG subclass response was evaluated alter the third dose (Figure 3). The results of an intra-group analysis showed that the serum IgG1 responses generated by all treatment groups were higher than their respective IgG2a and IgG2b (p £ 0.001), however, we observed a higher IgG2a and IgG2b responses for the groups immunized with the formulations containing HBcAg (Figure 3).
Group 6 generated a significantly superior HBsAg-specific IgG1 and IgG2a response compared to the response generated by the conventional vaccine (group 2) (p < 0.05 and p < 0.01, respectively).
Lymphoproliferative response
One month after the third dose the proliferative response was evaluated stimulating the cells with each antigen (HBsAg and HBcAg).Some groups of the study were selected with this purpose. In the case of the HBsAg-specific response (Figure 4), a positive response for groups 2 and 6, with a trend to generate a better proliferative response for the last was observed.
The evaluation of the HBcAg-specific proliferative response (Figure 4) showed a higher response for group 6 immunized with the formulation containing HBsAg and HBcAg compared with group 8, immunized with the HBcAg alone.
DISCUSSION
Although an effective prophylactic vaccine against hepatitis B is commercially available since the 1980´s, there remains a specific requirement for HBV vaccines capable of circumventing the non-responder or low-responder status of some vaccines.
Non-responsiveness to hepatitis B virus (HBV) vaccine in adults is associated with multiple causes, such as the HLA-C4AQ0, DRB1*0301 or DQB1*02 haplotypes (17), the insufficient production of the specific B-cell repertoire or helper T-cell function (19), among others.
Although it has been recognized the concept of cellular and cytokine mediated HBV clearance (20, 21), in the prophylactic scenario, the HBsAg-specific antibody response is considered the criterion of protection until now. That is why one of the challenges in the new Hepatitis B vaccine development is to increase anti-HBsAg antibody titers in non-/low-responders to conventional vaccines.
The inclusion of HBcAg in the vaccine formulation, a second HBV antigen regarded as middle protective and with immunoenhancing and immunomodulatory properties (10-12), provided the formulation with special features. The bi-valence of the combined formulation allows the generation of immune response against HBcAg, which is highly immunogenic, and activates nonspecifically several cell types (14, 22). The generation of a broader and more potent humoral and cellular immune response could also helps to circumvent the non-responder status to HBsAg.
In the present work, the immunogenicity of the formulation containing HBV surface and nucleocapsid antigens administered by intramuscular route of immunization has been optimized evaluating several doses for each antigen and different immunization schedules. Similar to the results reported for the nasal use of this formulation (11, 12), parenteral administration generates higher humoral responses when compare to the conventional vaccine. The immunoenhancing and immunomodulatory properties of the HBcAg were also demonstrated employing the intramuscular route, both at humoral and cellular levels, in agreement with a previous report (10).
The formulation containing the proportion HBsAg/ HBcAg (1:2) showed to be the most immunogenic.
In the first immunization schedule it was demonstrated that the inclusion of the HBcAg in the formulation allows the reduction of the number of doses established in conventional HBV vaccination protocols from three to two doses. This could make an impact in the reduction of injections to be completely protected against HBV infection. There is an important percentage of unsafe injections, mainly in developing countries (23). Limiting the number of injections will reduce the transmission risk of several diseases (24). The presence of HBcAg in the formulation will also contribute to the general protective capacity of the resulting formulation.
In addition, the inclusion of HBcAg enables the reduction of the HBsAg dose level based on the fact that a similar specific IgG response were observed for the groups immunized with the commercial vaccine compared to the group immunized with the combined formulation at half dose level (groups 2 and 7). This last result was reproduced in the second experiment, indicating that the presence of HBcAg in the formulation allowed a fourfold reduction in the HBsAg dose. This could potentially reduce the cost of the immunization based on the dual adjuvant and immunogen role of HBcAg.
The obtained HBsAg-specific IgG subclasses responses are in line with previous reports obtained in BalbC and C57/Bl6 mice (10-12), indicating that the presence of HBcAg modulated the humoral response for co-administered antigens in a pro-Th1 pattern. It has been previously reported that these properties of the HBcAg are related to the presence of trace amounts (5-20 ng/µg protein) of bacterial RNA encapsidated in the core particle (10). In the present work, we point out that the strong Th1-immunomodulating properties previously reported for nasal route could be modulated by the presence of alum in the formulations described. However, significant differences were still detected in the HBsAg-specific IgG2a response between the control group and the group receiving the higher dose of HBcAg, corresponding to formulation HBs/ HBc (1:2) (group 6). A similar behavior was seen for HBsAg-specific proliferative capacity in total spleen cells, in this case there was also a trend to generate a higher response for the combined formulation.
In general, the generated HBcAg specific immune response was high for all groups, in accordance with the immunological characteristic reported for this antigen (14). Interestingly, the results reported previously for nasal administrations (11, 12), were reproduced using a parenteral immunization route, indicating that a synergistic immunoenhancing effect occurs when the HBsAg/HBcAg formulation is administered.
The present work demonstrates that the use of the combined formulation by intramuscular route is effective generating a potent humoral immune response, opening the possibility of administration by different routes. Moreover, the possibility to administer the formulation employing nasal and parenteral routes allows the development of co-administration or combined administration strategies, useful in difficult preventive settings or therapeutic vaccination.
Presented results suggest that the formulation containing HBsAg and HBcAg administered by parenteral routes could be potentially effective for vaccinating low/non-responders, such as immuno-compromised patients, patients exposed to hemodialysis treatment and old people, among others. On the other hand, such formulation could be applied to Hepatitis B chronic patients alone or combined with mucosally administered formulation.
The development of this potent HBV vaccine formulation with attractive characteristics both for prevention or therapeutic use, could be a future tool to control chronic infected population´s growth around the world.
ACKNOWLEDGMENTS
The present work was financed by The Center for Genetic Engineering and Biotechnology, Havana, Cuba.
STATEMENT OF INTERESTS
All the authors are employees of the Center for Genetic Engineering and Biotechnology, which owns a patent (Method for obtaining antigenic aggregates and the use thereof in formulations. PCT/CU01/00009, 2001) on the parenteral use of the formulation.
REFERENCES
1. Hilleman MR. Overview of the pathogenesis, prophylaxis and therapeusis of viral hepatitis B, with focus on reduction to practical applications. Vaccine 2001;19:1837-48.
2. Hoofnagle JH, Doo E, Liang TJ, Fleischer R, Lok ASF. Management of hepatitis B: summary of a clinical research workshop. Hepatology 2007;45:1056-75.
3. Mandell GL, Bennet JE, Dolin R. Principles and practice of infectious diseases 5th edn (CD Rom). New York: Churchill Livingstone, 2000.
4. Page M, Jones CD, Bailey C. A novel, recombinant triple antigen hepatitis B vaccine (Hepacare). Intervirol 2001;44(2-3): 88-97.
5. Yalcin K, Danis R, Degertekin H, Alp MN, Tekes S, Budak T. The lack of effect of therapeutic vaccination with a pre-S2/S HBV vaccine in the immune tolerant phase of chronic HBV infection. J Clin Gastroenterol 2003;37:330-5.
6. Vandepapeliere P, Lau G, Leroux-Roels G, Horsmans Y, Gane E, Tawandee T, et al. Therapeutic vaccination of chronic hepatitis B patients with virus suppression by antiviral therapy: A randomized, controlled study of co-administration of HBsAg/AS02 candidate vaccine and lamivudine. Vaccine 2007;25:8585-97.
7. Wettendorff M. Therapeutic vaccination. Virus Res 2002;82:133-40.
8. Webster GJM, Bertoletti A. Quantity and quality of virus specific CD8 cell response: relevance to the design of a therapeutic vaccine for chronic HBV infection. Mol Immunol 2001;38:467-73.
9. Bertoletti A, Naoumov NV. Translation of immunological knowledge into better treatments of chronic hepatitis B. J Hepatol 2003;39:115-24.
10. Riedl P, Buschle M, Reimann J, Schirmbeck R. Binding immune-stimulating oligonucleotides to cationic peptides from viral core antigen enhances their potency as adjuvants. Eur J Immunol 2002;32:1709-16.
11. Lobaina Y, Palenzuela D, Pichardo D, Muzio V, Guillen G, Aguilar JC. Immunological characterization of two hepatitis B core antigen variants and their immunoenhancing effect on co-delivered hepatitis B surface antigen. Mol Immunol 2005;42:289-94.
12. Aguilar JC, Lobaina Y, Muzio V, García D, Pentón E, Iglesias E, et al. Development of a nasal vaccine for chronic hepatitis B infection that uses the ability of hepatitis B core antigen to stimulate a strong Th1 response against hepatitis B surface antigen. Immunol Cell Biol 2004;82:539-46.
13. Aguilar A, González CA, Cinza Z, Cabrera J, Veliz G, Moreno SR, et al. Phase I clinical trial in healthy adults of a nasal vaccine candidate containing recombinant hepatitis B surface and core antigens. Int J Infect Dis 2007;11:394-401.
14. Vanlandschoot P, Cao T, Leroux-Roels G. The nucleocapsid of the hepatitis B virus: a remarkable immunogenic structure. Antiviral Res 2003;60(2):67-74.
15. Iglesias E, Franch O, Carrazana Y, Lobaina Y, García D, Sánchez J, et al. Influence of aluminum-based adjuvant on the immune response to multiantigenic formulation. Viral Immunol 2006; 19(4):712-21.
16. Belloni C, Tinelli C, Orsolini P, Pistorio A, Avanzini A, Moretta A, et al. Revaccination against hepatitis B virus of nonresponding and low-responding infants immunised at birth. A parallel evaluation of rubella and tetanus vaccine. Vaccine 1998;16:399-402.
17. De Silvestri A, Pasi A, Martinetti M, Belloni C, Tinelli C, Rondini G, et al. Family study of non-responsiveness to hepatitis B vaccine confirms the importance of HLA class III C4A locus. Genes Immun 2001; 2(7):367-72.
18. Alper CA. The human immune response to hepatitis B surface antigen. Exp Clin Immunogenet 1995;12:171-81.
19. Shokrgozar MA, Shokri F. Enumeration of hepatitis B surface antigen-specific B lymphocytes in responder and non-responder normal individuals vaccinated with recombinant hepatitis B surface antigen. Immunol 2001;104;1:75-9.
20. Bertoletti A, Gehring AJ. The immune response during hepatitis B virus infection. J Gen Virol 2006;87(Pt 6):1439-49.
21. Wieland S, Thimme R, Purcell RH, Chisari FV. Genomic analysis of the host response to hepatitis B virus infection. Proc Natl Acad Sci USA 2004;27;101(17):6669- 74.
22. Lazdina U, Cao T, Steinbergs J, Alheim M, Pumpens P, Peterson DL, et al. Molecular basis for the interaction of the hepatitis B virus core antigen with the surface immunoglobulin receptor on naive B cells. J Virol 2001;75(14):6367-74.
23. Miller MA, Pisani E. The cost of unsafe injections. Bull World Health Organ 1999;77:808-11.
24. World Health Organization. Safety of Injections: Global Facts & Figures. Geneva, 2004.
Received in April, 2010.
Accepted for publication in August, 2010.