My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.28 no.1 La Habana Jan.-Mar. 2011
RESEARCH
Diagnostic value of alpha-fetoprotein for hepatocellular carcinoma
Valor diagnóstico de la alfa-fetoproteína en el carcinoma hepatocelular
Julio C Hernández1, Marcia Samada1, Alejandro Roque1, Yolanda Cruz2, Ivón Howland2, Irma Fernández3
1 Grupo de Hepatología
2 Laboratorio Clínico
3 Bioestadística Centro de Investigaciones Médico Quirúrgicas, CIMEQ Calle 216 y 11B, Reparto Siboney, Playa, CP 12100, La Habana, Cuba
ABSTRACT
Hepatocellular carcinoma (HCC) variously occupies the fifth or sixth position as the most frequent neoplasia worldwide. The present work used alpha-fetoprotein (AFP) determinations on the ultra-micro analytical system (SUMA®) as a tumoral marker in 189 cirrhotic patients evaluated at the Center for Medical and Surgical Research between January 1999 and September 2005. The principal factors associated to increases in AFP were HCC and viral cirrhosis. In all, 22 patients (11.64%) suffered from HCC, with viral cirrhosis caused mainly by hepatitis C virus infections as the most important etiological factor. AFP as a tumoral marker displayed a sensitivity of 68.18% and a specificity of 92.17%, which increased to 86.36 and 100% respectively when combined with abdominal sonography. It is concluded that AFP is valuable for the diagnosis of HCC.
Keywords: hepatocelular carcinoma, cirrhosis, alpha-fetoprotein.
RESUMEN
El carcinoma hepatocelular (CHC) es la quinta y sexta neoplasia más frecuente en el mundo. En este trabajo se empleó la alfa-fetoproteína (AFP) por la técnica del sistema ultramicroanalítico (SUMA®), como marcador tumoral en 189 pacientes cirróticos evaluados en el Centro de Investigaciones Médico-Quirúrgicas (CIMEQ), entre enero de 1999 y septiembre de 2005. Los principales factores que se asociaron a una elevación de la AFP fueron el CHC y la cirrosis viral. Veintidós enfermos presentaron CHC (11.64%) y la causa más importante fue la cirrosis hepática viral, principalmente por el virus de la hepatitis C. Este marcador tumoral mostró una sensibilidad de 68.18% y una especificidad de 92.17%. Al combinarlo con la ecografía abdominal, se incrementó la sensibilidad a 86.36% y la especificidad a 100%. Se concluyó que la AFP tuvo valor en el diagnóstico del CHC.
Palabras clave: carcinoma hepatocelular, cirrosis, alfa-fetoproteína.
INTRODUCTION
Hepatocellular carcinoma (HCC) is a malignant tumor of epithelial origin derived from parenchymal cells of the liver. According to the available statistical data, it is the main cause of death in people with compensated hepatic cirrhosis (HC), and variously alternates between the fifth and sixth position, according to the country, as the most frequent neoplasia (500 000 to 700 000 new cases worldwide per year). In addition, it has very low annual survival rates (3 to 5%), and is considered to be the third most deadly cancer (1, 2).
The diagnosis of HCC is often based on screening and surveillance strategies whose mainstays are the use of imagenological techniques and the measurement of the levels of serum alpha-fetoprotein (AFP) (3-5).
AFP is a 72 kDa onco-fetal glycoprotein with a size of 591 aminoacids (6). It is normally synthesized during fetal life, first in the yolk sac and then in fetal liver; its synthesis is normally repressed in adults (7). High levels of AFP are observed during adulthood only under certain conditions, such as pregnancy, the presence of some neoplasias (e.g. HCC, gastric carcinoma, testicular carcinoma, lung cancer and pancreatic cancer) and some non-neoplastic disorders such as HC and chronic hepatitis (8).
The association between serum AFP and HCC has been widely examined and described by a large number of groups (9). Regardless, its sensitivity and specificity for diagnosing HCC are variable, with Figures ranging from 39 to 73% and 65 to 96%, respectively (3, 10-29), depending on factors such as the specific assay used, the design of the study, the characteristics of the study population, and the designated cut-off level (20).
A new liver transplantation (LT) program began to be implemented and developed at the Center for Medical and Surgical Research (CIMEQ) starting from 1998, a necessary part of which was the evaluation of cirrhotic patients in order to discard the presence of HCC. In order to reach this objective, a screening and surveillance strategy was followed, based on the determination of serum AFP with the Cuban ultra-micro analytical system SUMA®.
Therefore, a study was designed aimed at the identification of the factors associated to increased levels of serum AFP in the target population (cirrhotic patients). The study was also designed to measure the sensitivity and specificity of AFP for the diagnosis of HCC in HC patients.
MATERIALS AND METHODS
This was a descriptive, prospective and longitudinal study that took place at CIMEQ from January 1999 to September 2005. A total of 191 patients with HC of varying etiologies were evaluated and treated by the group specialized in liver transplantation (LT). The exclusion criteria were pregnancy, antecedents of other neoplasias and refusal of the patient to enter the study. Only two prospective patients were eliminated due to previous neoplasias.
The data gathered for the investigation were obtained through the clinical evaluation of the patients using direct interviews, physical examinations, and the results of complementary studies contained in their medical records. A diagnosis of HC was established by compliance with at least one of the following criteria: histology, laparoscopy, and unequivocal clinical signs of the disease, provided mainly by the physical exam, the imagenological elements of the sonograms and the results of the upper GI endoscopy.
The HCC diagnosis took into account the criteria established by the European Association for the Study of Liver Disease (EASLD) for patients with HC and tumoral lesions larger than 2 cm, which produce a typical pattern of hypervascularization for imagenological techniques (11).
The reference values for serum AFP established by the Immunoassay Center using the SUMA® platform were used throughout this work. Although the laboratory facilities at CIMEQ produce AFP results in UI/mL they were converted to ng/mL (conversion factor: 1 UI/mL = 1.24 ng/mL) to guarantee uniformity in the discussion of the work. A test was considered normal if AFP concentration was below 15 UI/mL (i.e. lower than 18.60 ng/mL). The patients were fasted prior to the collection of the blood samples, which were processed by UMELISA®AFP (immunoenzyme assay used for the quantitative determination of alpha-fetoprotein in human serum and amniotic fluid) at the SUMA® laboratory of the Clinical Department from CIMEQ.
The AFP determinations were performed during the first evaluation visit of the patient, and then every 6 months. The serum level of AFP related to the diagnosis, was corresponded with the first value of this tumoral marker at the moment of diagnosis of the disease.
The data were processed with the statistical software package Statistical Package for the Social Sciences (SPSS) version 13.0 and with Epidat version 3.1, an informatics tool for the epidemiological analysis of tabulated data. Mean value, standard deviation (SD) and median were computed for all quantitative variables; using percentages instead for qualitative parameters. Sensitivity, specificity, positive predictive values (PPV) and negative predictive values (NPV) were also calculated and used to estimate Youden’s index.
The comparison of variables in categories was performed using chi-squared and Fisher’s exact probability tests. Mann-Whitney’s U test or the Kruskal Wallis test was used when comparing quantitative variables between two or more independent groups, respectively. The Tamhane test was used for statistically significant differences.
The calculation of the sensitivity and specificity reached when using different serum AFP thresholds for the diagnosis of HCC was carried out by constructing receiver operating characteristic (ROC) curves. The area under the ROC curve (AUROC) was determined and compared in the different study groups.
In all cases, p ≤ 0.005 was taken as the threshold for statistical significance.
The Ethics Committee and the Scientific Council from CIMEQ reviewed and approved the protocol for this investigation before its commencement.
RESULTS
Table 1 shows the demographic and clinical characteristics of the 189 patients included in the study. Viral hepatitis was the main cause of HC, with 83 cases (43.92%). Hepatitis C virus (HCV) was the most frequent viral agent, being involved in 59 of these 83 cases; in addition, two of these patients were co infected with the hepatitis B virus (HBV), and other seven patients had problems with alcohol consumption. The second most common etiology was alcohol consumption, followed by cryptogenic cirrhosis, with 36 (19.05%) and 31 (16.40%) cases, respectively. The remaining causes (autoimmune hepatitis, primary biliary cirrhosis, primary sclerosing cholangitis, secondary biliary cirrhosis and Wilson’s disease) were grouped together as ‘other causes’, with 39 patients (20.63%).
Twenty-two patients were diagnosed with HCC (11.64%); they were predominantly male (male-female ratio was 6.33). The main cause of HCC was viral HC, with 13 patients (59.09%), triggered mainly by HCV infections (8 cases, one of them co-infected with HBV). The remaining etiologies for HCC in this group were alcoholic HC and cryptogenic cirrhosis, with four (18.18%) and five (22.73%) patients respectively. The average age in this patient group was 55 ± 11.99 years (Table 1).
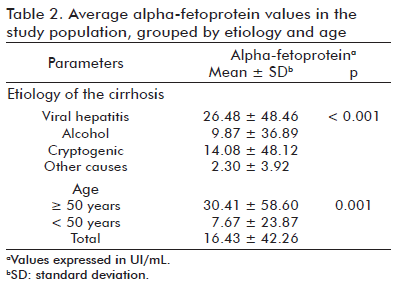
A total of 252 serum AFP determinations by the SUMA® methodology were performed during the study, yielding a mean value of 16.43 ± 42.26 UI/mL. Table 2 shows the mean AFP values divided by HC etiology and age. It is evident that the levels were markedly increased in patients with viral hepatitis (p < 0.001) and in patients with an age of 50 years or older (p = 0.001).
The only statistically significant difference found in the multiple comparisons analysis (Table 3) was that of the mean AFP levels of patients with viral HC compared to patients whose etiology fell under the heading of ‘other causes’ (p < 0.001).
The levels of AFP in patients also suffering from HCC were higher than in the remaining patients (110.78 UI/mL vs. 7.40 UI/mL) (p < 0.001) (Table 4).
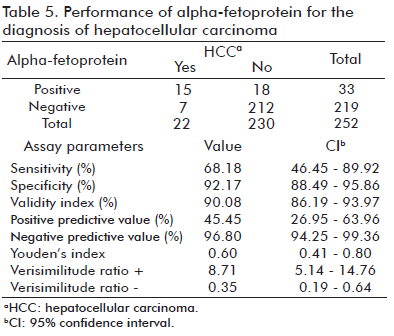
The sensitivity of AFP determinations for diagnosing HCC was 68.18%; the specificity values obtained were 92.17%; 45.45% for the PPV; and 96.80% for the NPV. Youden’s index was low, at 0.60 (Table 5).
The results for the diagnostic application of AFP in the case of HC, divided by etiology, are presented in Table 6. Youden’s index was low for all patient groups, with values of 0.58, 0.50 and 0.60 for viral HC, alcoholic HC and cryptogenic HC, respectively.
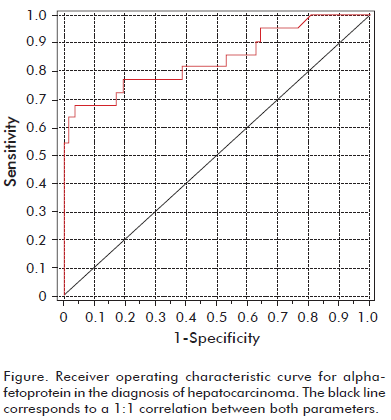
The Figure shows the ROC curve of AFP when used to diagnose HCC. The area under the curve amounted to 0.846 (0.74-0.95). Table 7 presents the sensitivity, specificity, and Youden’s index, according to the specific diagnostic threshold chosen. Youden’s index was also low for each of these groups.
All patients underwent imagenological tests, which supported the HCC diagnosis established with the study criteria. The sensitivity, specificity, PPV and NPV of abdominal sonography were 86.36%, 100%, 100% and 98.71% respectively to diagnose HCC. Youden’s index was very good (0.86). The combination of abdominal sonography and AFP increased sensitivity to 90.91% and NPV to 99.14%. Youden’s index, again, was excellent, at 0.91 (Table 8).
DISCUSSION
Solid data on the real prevalence of HC worldwide is still lacking, and the available statistical figures are always underestimated due to the high prevalence of undiagnosed cirrhoses. This situation is caused mainly by the fact that patients with compensated HC usually have no symptoms or conspicuous clinical signs of liver insufficiency and/or portal hypertension; in addition, they may remain so far considerable periods of time (21).
The etiology of HC, on the other hand, is better known. In developed countries, the causes of most HC cases are HCV infections and alcoholic liver disease (22), while in other parts of the world, such as the Asian Southeast and sub-Saharan Africa, HBV infections constitute the main etiological agent (23).
The results of this study are conformed to our expectations. Viral infections, mainly by HCV, and alcohol consumption, were the first and second most important etiologies for HC, respectively. This is the pattern typical of geographic areas where HBV infection is not endemic (22). The growing prevalence of HCV over HBV in Cuba constitutes the result of the sustained application of blood donor screening programs and, most important, reflects the impact of the National Vaccination Program against HBV, which began in 1991 with the administration of an effective HBV vaccine manufactured in the country (Heberbiovac-HB®) to newborns from carrier mothers and was later extended in 1992 to all newborns, together with the vaccination of the cohorts of children 8 and 14 years old from 1994 onwards (24).
HC has been known for a long time as the most important risk factor for the development of HCC (25). Independently from the etiology of HC, the risk of developing HCC in these patients is estimated to range from 3 to 5% in a year (10, 26).
This investigation demonstrated the presence of HCC in 11.64% of the patients included in the study (Table 4). The principal causes of HCC, ordered by frequency, were the presence of viral HC (HCV-related mainly), the presence of cryptogenic cirrhosis, and the consumption of alcohol. These data are similar to those reported in countries with low prevalence and incidence rates of HBV (11, 27).
Although serum AFP levels have been shown to increase in association with several carcinomas, this parameter has only been employed as a tumoral marker for HCC (28, 29). The present investigation showed that serum AFP levels, measured with SUMA® technology, were significantly higher in patients aged 50 years or older, in patients with viral HC, and in patients with HCC.
The results coincide with previous reports investigating the influence of viral infections on AFP concentrations. Increased levels of AFP are detected in viral hepatitis patients with no detectable HCC. Taking into account the results from different publications, it can be estimated that 10 to 43% of persons with a chronic HCV infection will have increases in this serum marker (8, 30, 31). Searching for an explanation to this finding, several studies have demonstrated a correlation between increased serum AFP levels and the degree of inflammation observed in liver biopsies (30-32).
The investigations examining the usefulness of AFP as a screening and surveillance tool have not been directly comparable: their design and the characteristics of the studied population (type of viral infection, severity of liver disease, demographics) have differed. This fact alone explains the wide variability of their results, with sensitivities ranging from 41 to 69% and specificities between 75 and 94% (3, 9, 12, 16, 17, 20, 33-37).
A publication by Trevisiani et al. (3) has proposed that the best diagnostic threshold level for AFP ranges from 16 to 20 ng/mL. Using a threshold of 20 ng/mL, specificity was indeed high (89.4%), but sensitivity was only 60%; this would produce such a significant number of false negative diagnoses for a disease with the implications of HCC that the marker would not be useful for this purpose. Lowering the threshold identifies a larger number of cases, improving sensitivity at the price of an increased false positive rate. In other words, the higher the threshold, the lower the number of detected cases obtained (sensitivity decreases). A different study by Gambarin-Gelman et al. (12) also used an AFP diagnostic threshold of 20 ng/mL, and obtained a sensitivity of 58% and a specificity of 91%. In exchange, PPV increased from 58 to 75% at higher levels of AFP (50 ng/mL), although it must be underscored that in the latter case, sensitivity dropped from 58 to 47%.
More recent results from Durazo et al. (33) and El-Husseini et al. (34) using diagnostic thresholds of 19.8 and 25 ng/mL have reached the highest sensitivities reported so far in the literature (69 the former and 68.2% the latter). Specificity in both studies was, however, discreetly lower.
The sensitivity and specificity results obtained in this investigation for serum AFP measured with SUMA® technology can be considered good when compared to the results summarized above. Using a diagnostic threshold of 15.38 UI/mL (equivalent to 19.07 ng/mL), sensitivity was 68.2%: a figure higher than the average of all the mentioned reports (60%) and similar to the two most recent ones. Specificity, for this threshold, was also high, at 90%. This value also falls within the published range (78.3 to 94%).
It can be concluded that serum AFP levels of 91.71 UI/mL (113.72 ng/mL) or higher had diagnostic value, with a specificity higher than 99%. In short, less than 1% of the patients without HCC had AFP levels higher than 100 ng/mL. This is a better outcome than that reported by Nguyen et al. (16), who had a lower specificity (97.3%) at the same AFP threshold of 100 ng/mL and had to increase it to 200 ng/mL or higher to obtain a specificity of 100%. In the report from Trevisani et al. (3), levels higher than 200 ng/mL had a specificity of 99.4%.
Reviewing the evidence analyzed above, we support the position stated by Gómez Senent et al. (38) in a recent publication, arguing that the high specificity that can be obtained with elevated diagnostic thresholds for AFP allow its use as a confirmatory test for HCC diagnosis.
The actual usefulness of sonography, as the sole diagnostic test, for the screening and surveillance of HCC in patients with HC is being increasingly contested, as a consequence of the absence of prospective studies and the widely varying sensitivity (35 to 84%) displayed by the technique. This variability has been ascribed not only to methodological differences in study populations, disease severity and non-uniform sampling frequencies, but also to susceptibilities to changes in tumor morphology, operator training and the quality of the measuring instrument (15, 39).
Regardless, abdominal sonography for HCC diagnosis performed, in this study, at levels of sensitivity and specificity much better than those presented in the preceding paragraph. Although we cannot reach a definitive conclusion regarding this result, it may be related to the characteristics of the tumoral lesions and the long experience of the personnel operating the ultrasonographic equipment.
The diagnostic potential of the AFP/sonography combination has been examined before in other countries with good results. This combination aims, mainly, at simultaneously increasing both sensitivity and specificity for the diagnosis of HCC. Some authors refer that with this method sensitivity can even be taken to 100% (40). In the present study, the combination of imagenology with serum AFP measurements by the SUMA® technology at a diagnostic threshold of 15 UI/mL managed to increase sensitivity and NPV to 90.91 and 99.14%, respectively; also increasing Youden’s index. The increase in sensitivity, compared to sonography alone, is 5.7%. Values at the same order of sensitivity were reported by Kang et al. (41).