My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Biotecnología Aplicada
On-line version ISSN 1027-2852
Biotecnol Apl vol.30 no.2 La Habana Apr.-June 2013
TECHNIQUE
Modified T4 Neonatal UMELISA® to 3 mm discs for a more rational use of dried blood newborn samples on filter paper
UMELISA T4 Neonatal® modificado a discos de 3 mm, para un uso más racional de las muestras de sangre seca sobre papel de filtro de recién nacidos
Elisa Castells, Ernesto C González, Amarilys Frómeta, Pedro L Pérez, Pedro Almenares, Lesley del Río, Yileidis Tejeda, Mary T Segura
Laboratorio de Pesquisa Neonatal, Centro de Inmunoensayo, CIE. Calle 134 y Ave. 25, AP 6653, Cubanacán, Playa, La Habana, Cuba.
ABSTRACT
Congenital hypothyroidism (CH) is one of the most common preventable causes of mental retardation. Newborn screening and thyroid therapy started within 2 weeks of age can normalize the cognitive and motor impairment caused by lack of thyroid hormone during the early postnatal phase of brain development. The availability of assays to determine thyroxin (T4) and thyroid stimulating hormone has allowed the establishment of newborn screening programs for CH. The Immunoassay Center (Cuba) developed the T4 Neonatal UMELISA® to determine neonatal T4 levels in dried blood on filter paper using 5 mm discs of samples, standards and controls. The number of diseases which can be diagnosed in Neonatal Screening Programs has increased, so it is necessary to make the maximum use of blood samples collected on filter paper. This work shows the standardization of the T4 Neonatal UMELISA® modified in order to use 3 mm discs. The assay was completed in 3 ½ hours, with a measuring range of 25-400 nmol/L. The intra- and inter-assay coefficients of variation (CV) were 6-10 % and 7-12 % respectively, depending on the T4 concentrations. The recovery ranged from 91.8-115.1 %. The modified assay showed high Pearson correlation (r = 0.877) and concordance correlation (ρc = 0.867) with the T4 Neonatal UMELISA®. The performed modifications do not affect the sensitivity, precision and accuracy of the assay, permitting a more rational use of newborn blood samples and the possibility to increase the number of diseases included in the newborn screening programs.
Keywords: UMELISA, T4, congenital hypothyroidism, dried blood spots, neonatal screening.
RESUMEN
El hipotiroidismo congénito (HC) es una de las causas más frecuentes de retraso mental prevenible en la infancia. La pesquisa neonatal (PN), su diagnóstico y tratamiento, podrían normalizar la discapacidad cognitiva y motora por la falta de hormonas tiroideas durante la etapa temprana de desarrollo del cerebro. Los ensayos para determinar la tiroxina total (T4) y la hormona estimulante del tiroides han permitido establecer programas de PN del HC. El Centro de Inmunoensayo, de La Habana, desarrolló el ensayo inmunoenzimático competitivo UMELISA T4 Neonatal® para determinar los niveles de T4 en neonatos, utilizando discos de 5 mm de muestras, calibradores y controles de sangre seca sobre papel de filtro. Las enfermedades que se pueden diagnosticar mediante esos programas han aumentado, por lo que se deben optimizar los estudios con las muestras de sangre de recién nacidos tomadas en papel de filtro. Este trabajo consistió en la estandarización de un UMELISA T4 Neonatal® modificado para emplear discos de 3 mm. El ensayo dura 3 ½ h, y el análisis abarca entre 25 y 400 nmol/L. Los coeficientes de variación intra e interensayo estuvieron en los rangos 6-10 % y 7-12 %, respectivamente. La recuperación fue de 91.8 y 115.1 %. La correlación de Pearson fue elevada (r = 0.877) y hubo concordancia de la correlación (ρc = 0.867) con el UMELISA T4 Neonatal
Palabras clave: UMELISA, T4, hipotiroidismo congénito, sangre seca, pesquisa neonatal.
INTRODUCTION
Congenital hypothyroidism (CH) is an inherited metabolic disorder, known as the most common preventable cause of mental retardation in children, with a worldwide incidence of 1:3000 to 1:4000 newborns [1, 2]. In Cuba, the estimated incidence is 1:3000 live births [3, 4].
The clinical features of CH are often subtle and many newborn infants remain undiagnosed at birth [5]. The main purpose of neonatal mass screening for CH is early detection and treatment of this disease. All infants with CH should be rendered euthyroid as promptly as possible by replacement therapy with thyroid hormone. An initial dosage of 10 to 15 µg/kg levothyroxin is recommended [6]. The first American Academy of Pediatrics recommendations for newborn screening for CH were published in 1993 [7]. During the past three decades, this screening has been very successful with the vast majority of patients with CH achieving normal neurological outcome [1, 2, 8].
Ideally, universal screening at 3-4 days of age should be done for detecting CH. Two screening strategies for the detection of CH have evolved: a primary thyroid stimulating hormone (TSH)/backup thyroxine (T4) method and a primary T4/backup TSH method. In addition, an increasing number of programs use a combined primary TSH plus T4 approach [6, 9].
In this sense, the T4 neonatal ultramicro enzyme-linked immunosorbent assay (UMELISA®) technique was designed for the determination of T4 in newborns dried blood samples using 5 mm discs [10]. This is a fluorescent enzyme immunoassay that keeps up the sensitivity of the traditional ELISA methods but requires less volume which reduces significantly the cost for determination. Nowadays, with the increase of tests done in neonatal screening programs, it’s necessary to use smaller blood punches in order to make a more rational use of the samples.
The aim of this work was to standardize a high quality assay for T4 determinations in dried blood samples using 3-mm filter paper discs, comparable to the T4 Neonatal UMELISA® used at present in various Latin American countries.
MATERIALS AND METHODS
Chemicals
All the reagents were analytical grade and solutions were prepared in distilled water. Sodium carbonate, sodium bicarbonate, sodium azide, sodium chloride, disodium hydrogen phosphate, potassium chloride, potassium dihydrogen phosphate, magnesium chloride, zinc chloride, Tris-HCl, Tween 20, saccharose, activated charcoal and 5,5-diethylbarbituric acid sodium salt were from Merck. Bovine serum albumin (BSA) and alkaline phosphatase were from Roche. T4 antiserum and glutaraldehyde were from Sigma.
Equipment and accessories
The Ultra Micro Analytic System (SUMA®) technology is a complete system of reagents and instrumentation which comprises a fully computerized spectrofluorimeter (PR-621) for automatic reading, validation, interpretation and quantification of results, a plate washer (MW-2001), a manual punch and reagent kits designed to perform ultramicrotests (10 μL volumes of samples and reagents). The system is manufactured by the Immunoassay Center (Havana, Cuba).
T4-free human plasma
T4 hormone-free plasma was prepared by treating pooled human plasma with 20 % (w/v) of activated charcoal. The plasma was incubated for 4 h at room temperature with stirring followed by repose during 18 h at 2-8 °C. Subsequently, the plasma was centrifuged at 12 000 × g for 1 h, filtered to remove the charcoal, re-centrifuged at 69 700 × g during 4 h at 2-8 °C and re-filtered. The absence of T4 was confirmed by the T4 Neonatal UMELISA® test.
Purification of polyclonal rabbit anti-T4 antibodies
Polyclonal rabbit anti-T4 antibodies were obtained by ion exchange chromatography using a DEAE-Sephacel® matrix (Pharmacia Biotech). The purified rabbit antiserum was stored in aliquots at -20 °C until use.
Immobilization of polyclonal rabbit anti-T4 antibodies
White opaque 96-well polystyrene UMELISA® plates (Tecnosuma International S.A., Havana, Cuba) were coated with 17 μL/well of anti-T4 rabbit polyclonal antibodies at concentrations ranging from 6 μg/mL in 0.05 mol/L sodium carbonate/ bicarbonate buffer (pH 9.6) containing 3 mmol/L of NaN3. The plates were placed for 4 h in a humid chamber at 37 °C and washed with 25 μL/well of 0.15 mol/L of phosphate-buffered saline (PBS) solution containing 1.9 mol/L NaN3 and 1.1 mmol/L Tween 20. The buffer was removed and the plates were treated with 18 μL of a solution containing 14.5 μmol/L of BSA, 0.15 mol/L of saccharose and 0.45 mmol/L of Tween 20 for 18 h at room temperature (23-25 ºC) to increase their stability. Finally, the UMELISA® plates were dried and preserved with desiccant in polyvinyl sealed bags at 2-8 °C, where they are stable for at least 12 months.
Enzyme conjugate
The T4-alkaline phosphatase conjugate was prepared by a modification of the glutaraldehyde method, as previously described [11].
Standards, controls and samples
Erythrocytes from healthy subjects were washed three times with saline solution and then mixed with T4-free human plasma to adjust the hematocrit to 55 %. A T4 sodium hydroxide solution at a concentration of 1 mg/mL was used to prepare blood calibrators with whole blood concentrations between 25 and 400 nmol of T4/L. T4 standards were spotted on filter paper cards Whatman 903 (Whatman 903, Whatman, Maidstone, UK) and dried 24 h at room temperature. Finally, the standards were preserved with desiccant in polyvinyl sealed bags at 2-8 °C, where they are stable for at least 12 months.
Controls with a well known T4 concentration covering a wide range of standard curve activity were used for the assay evaluation. Controls were prepared from whole human blood with a 55 % hematocrit value and dried on filter paper from Whatman 903. One hundred and sixty-six dried blood samples from the National Neonatal Screening Program were evaluated using the T4 Neonatal UMELISA® and the 3 mm discs modified assay. Correlation between both assays was established. Additionally, other 410 dried blood samples were evaluated using the 3 mm discs modified assay. The mean and standard deviation (SD) of T4 concentrations were calculated. The samples were routinely obtained by heel puncture and collected on filter paper cards. Prior to participating in the study, the patient identification of samples was removed. Moreover, six controls from the Centers for Disease Control and Prevention (CDC; USA) were evaluated by both methods.
UMELISA®
T4 Neonatal UMELISA®
For the measurement of T4 concentrations, 5 mm blood discs of standards, controls and samples were punched out of the filter paper and placed into each well of the elution microplates, followed by the addition of 70 μL of the diluted T4-alkaline phosphatase conjugate in PBS solution with 0.12 mol/L of barbituric acid sodium salt, 0.45 mmol/L Tween 20 and 0.15 mmol/L BSA.
After the elution in a humid chamber for 1 h at room temperature, 10 μL of each eluate were transferred into the well of the reaction opaque polystyrene UMELISA® plates previously coated with the specific polyclonal rabbit anti-T4 antibodies. The competitive reaction was developed for 2 h at room temperature in a humid chamber and then, the plates were washed 6 times with 0.37 mol/L of Tris-HCl solution (pH 8.0) containing 3.76 mol/L NaCl, 1.1 mmol/L Tween 20 and 76.9 mmol/L NaN3. The fluorogenic reaction was performed by adding 10 μL of the substrate solution (pH 9.6) containing 5.07 mmol/L 4-methylumbelliferil phosphate, 0.92 mol/L diethanolamine-HCl, 0.7 mmol/L MgCl2 and 7 mmol/L NaN3. UMELISA® plates were further incubated at room temperature in a humid chamber for 30 min. Finally, the fluorescence was measured automatically in the fluorimeterpho-tometer reader. Automatic validation and interpretation of the results were done using specific assay software.
Modified T4 Neonatal UMELISA® using 3 mm discs
For the measurement of T4 concentrations, 3 mm blood discs of standards, controls and samples were punched out of the filter paper and placed into each well of the elution microplates, followed by the addition of 60 μL of the diluted T4-alkaline phosphatase conjugate in phosphate-buffered saline solution with 0.12 mol/L of barbituric acid sodium salt, 0.45 mmol/L of Tween 20 and 0.15 mmol/L of BSA. From this step on, the same procedure of the T4 Neonatal UMELISA® was followed.
Correlation with the T4 Neonatal UMELISA®
The quantitative differences between the modified assay and the T4 Neonatal UMELISA® were assessed using two approaches: the Pearson correlation and the concordance correlation coefficient (ρc). The slope, ordinate intercept of the line-of-best-fit, and regression statistics were obtained using the Microsoft Excel 2000 software, to compare our modified assay results versus those obtained on the same dried blood samples using T4 Neonatal UMELISA®. The concordance correlation coefficient was calculated as a measure of agreement between assays [12]. Concordance was classified as minor (values ranging 0.2-0.7), moderate (0.7-0.85) or high (above 0.85).
RESULTS
Standard curve of the modified T4 Neonatal UMELISA®
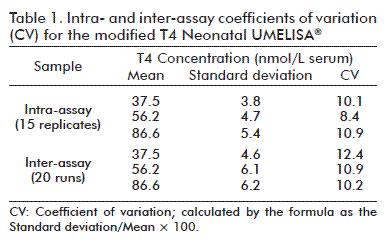
A typical standard curve and a precision profile obtained with the modified T4 Neonatal UMELISA® are shown in figure 1. The adjustment of the standard curve was automatically fitted to a linear function. The calculated values of the samples were interpolated in a graphic of the fluorescence quotient B/Bo (fluorescence for each calibrator over that of the standard calibrator of the curve; expressed in %) versus the T4 concentration, corresponding to the standard curve, getting the concentration values in nmol of T4/L of blood. In the resulting quotient, B/Bo was inversely proportional to the amount of T4 in the sample.
Limit of detection (LOD) and limit of quantitation (LOQ) were determined according to CLSI guidelines [13]. The zero standard and 3 spiked dried blood samples (15, 20 and 25 nmol/L) were analyzed 20 times to estimate LOD and LOQ. Means, SDs, CVs and slope were calculated. LOD was 13 nmol/L, defined as the lowest amount of T4 in a sample that can be detected with a probability of 95 %, and calculated by the formula:
![]()
where:
LOB was the 95 percentile of zero standard measurements; SDs = Pooled SD (estimated from measurements on spiked samples). LOQ was 19 nmol/L; the lowest amount of T4 in a sample that can be quantitatively determined in our UMELISA® with an acceptable precision and a suitable accuracy.
The spiked samples were studied to select the lowest concentration in the assay for which the CV is less than 20 % and the measured value is within 20 % (80-120 %) of true value, calculated by the formula:
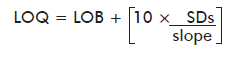
Precision and recovery of the modified T4 Neonatal UMELISA®
The precision of the modified T4 Neonatal UMELISA® is shown in table 1. Samples representing 3 different levels of T4 concentrations were assayed. The intra-assay variability was calculated with 15 replicates of each sample, equidistantly distributed on the same plate. The inter-assay precision was calculated from estimating T4 in the above samples in 10 independent assays. The intra- and inter-assay CVs ranges were 6-10 % and 7-12 %, respectively, depending on T4 concentrations.
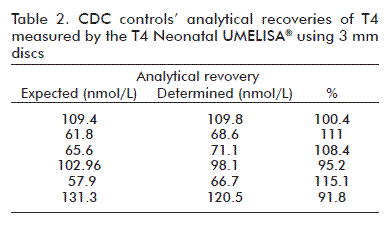
Analytical recovery was evaluated using six CDC controls. Percentage recovery ranged 91-115 % with a mean value of 103 % (Table 2).
Comparison of both methods
Using our modified assay and the T4 Neonatal UMELISA®, we evaluated 83 dried blood samples in duplicates. Results of correlation analyses are shown in figure 2. Additionally, six CDC controls were also evaluated with both methods. The outcomes of these assays highly correlated by the Pearson correlation (r = 0.877) where the slope, ordinate-intercept and linear correlation coefficient were 0.946, 10.06 and 0.769, respectively. The absolute agreement between assays was quantified using the concordance correlation. There was a high overall concordance between the modified assay and the T4 Neonatal UMELISA® (ρc = 0.867).
Analysis of dried blood samples using the modified T4 Neonatal UMELISA®
The distribution of the T4 concentrations using the modified method is shown in figure 3. T4 mean concentration in dried blood samples from 410 healthy neonates (5-to-7 days-old) was 123.2 (SD 31.5), ranging 24.2-220.5 nmol/L. All samples were obtained from apparently healthy full term neonates with birth weights above 2500 g. The modified assay cut-off value (10th percentile) was 100 nmol/L, similar to the one used in the T4 Neonatal UMELISA®.
DISCUSSION
Unrecognized CH leads to severe and irreversible mental retardation. Because signs and symptoms of this disease are often scarce and not easily recognizable, newborns are screened at birth for early CH detection.
Pilot screening programs for CH were developed in Quebec, Canada, and Pittsburgh, Pennsylvania, in 1974 and have now been established in Western Europe, North America, Japan, Australia, and parts of Eastern Europe, Asia, South America, and Central America [14-16]. In North America, more than 5 million newborns are screened and approximately 1400 infants with CH are detected annually. Certainly the main objective of screening, the eradication of mental retardation after CH, has been achieved. In addition to the profound clinical benefit, it has been estimated that the cost of screening for CH is much lower than the cost of diagnosing CH at an older age [6].
At the present time, there are a lot of methods for the diagnosis of CH, either by determining T4 (positive cases are confirmed by TSH quantification) or TSH (positive cases are confirmed by T4 quantification). Both hormones can be detected in human serum or dried blood on filter paper, being this way the most common method used worldwide in the neonatal screening [17].
Early in the experience of screening, most programs undertook an initial T4 test, with a follow-up TSH test on infants below a specified T4 cut-off [2]. With increasing accuracy of TSH measurement, many screening programs now carry out an initial TSH test to detect CH. Each program must develop its own T4 and TSH cut-off for recall of infants with abnormal test results. As there are rapid changes in TSH and T4 in the first few days of life, many programs have developed age-related cutoffs [5].
T4 levels in the neonatal period and brain function of early treated hypothyroid children show the great importance of neonatal screening programs. With screening and neonatal diagnosis, the majority of children who are treated early experience normal growth and neurologic development and normal-range intelligent coefficient values [18-20]. CH treatment involves thyroid hormone replacement with L-levothyroxine. The goals of early thyroid hormone therapy should be to maintain the total or free T4 levels in the upper half of the reference range during the first 3 years of life and to normalize TSH concentrations [6, 7].
This work shows the standardization of a modified T4 Neonatal UMELISA® which has as advantages the use of 3 mm dried blood discs and a 10 µL decrease in the elution volume. With the increment of the number of disorders included in the newborn screening programs, it is necessary the efficient use of newborn dried blood samples. For that reason, the majority of current in-house or commercial assays for detecting inborn errors of metabolism in the neonatal period use smaller blood punches (3 mm in diameter or lower) [21]. Currently, the SUMA® technology ultramicroassays for newborn screening purposes are designed to be performed on 3 mm dried blood discs [22-26].
Overall evaluation of the results shows that the modified T4 Neonatal UMELISA® has similar characteristics to other assays commercially available in terms of precision, accuracy and clinical utility, thus, making it potentially useful in the neonatal screening for CH. The total time required by the new assay using 3 mm discs is about 3 ½ h, similar to T4 Neonatal UMELISA® using 5 mm discs. The assay exhibited good within-run and between-run reproducibilities in the concentration range from 25 to 400 nmol/L, and the results obtained following the evaluation of CDC controls demonstrated its accuracy.
The modified T4 Neonatal UMELISA® is intended for the quantitative measurement of T4 in blood spots dried on filter paper and for the detection of children with CH, where T4 levels are below 80 nmol/L. That is why the LOD and LOQ are more than adequate for population screening.
The modified assay presented a high correlation with the T4 Neonatal UMELISA® (ρc = 0.876) when evaluating 83 newborn dried blood samples and six CDC controls. Additionally, the frequency distribution obtained with the modified assay using 3 mm discs, was similar to other populations [27-29]. For instance, Gruñeiro-Papendieck et al. reported comparable T4 levels measured by fluorimmunoassay (Wallac Inc Turku, Finland) in preterm (117 nmol/L and full term (181 nmol/L) babies [27]. Larsson et al. reported a T4 mean of 142 nmol/L in 19 289 dried blood samples collected on the 5th day as average [28], while González et al., using an ultramicroanalytical fluorescent enzyme immunoassay, obtained a T4 mean concentration of 107 nmol/L (SD 28.0 in 255 healthy euthyroid subjects serum samples and 105 nmol/L (SD 29.0) in 200 cord blood specimens [29].
In summary, the modified T4 Neonatal UMELISA®, as part of SUMA technology, is a simple, precise, accurate and rapid assay that can be used to carry out screening programs in newborns. Modifying the T4 Neonatal UMELISA® to 3 mm discs of dried blood on filter paper permits to make a more rational use of newborn samples and it makes possible to increase the number of diseases included in the newborn screening programs.
ACKNOWLEDGEMENTS
We thank B.A.Ed. Senia Bermúdez for her valuable help in the final language revision of the manuscript.
REFERENCES
1. Fisher DA, Dussault JH, Foley TP, Jr., Klein AH, LaFranchi S, Larsen PR, et al. Screening for congenital hypothyroidism: results of screening one million North American infants. J Pediatr. 1979;94(5):700-5.
2. Fisher DA. Second International Conference on Neonatal Thyroid Screening: progress report. J Pediatr. 1983;102(5):653-4.
3. Aliño M. El programa de pesquisa neonatal en Cuba. In: I Congreso Latinoamericano de Pesquisa Neonatal y Enfermedades Heredometabólicas. Ciudad de La Habana, Cuba; 1997. p. 28.
4. Guell R, Robaina R, Álvarez MA, Fernández JL. Más de 10 años de pesquisaje neonatal de hipotiroidismo congénito. In: I Congreso Latinoamericano de Pesquisa Neonatal y Enfermedades Heredo metabólicas. Ciudad de La Habana, Cuba, 1997. p. 41.
5. Rastogi MV, LaFranchi SH. Congenital hypothyroidism. Orphanet J Rare Dis. 2010;5:17.
6. Rose SR, Brown RS, Foley T, Kaplowitz PB, Kaye CI, Sundararajan S, et al. Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics. 2006;117(6):2290-303.
7. American Academy of Pediatrics AAP Section on Endocrinology and Committee on Genetics, and American Thyroid Association Committee on Public Health: Newborn screening for congenital hypothyroidism: recommended guidelines. Pediatrics. 1993;91(6):1203-9.
8. Fisher DA. Congenital hypothyroidism. Thyroid Int. 2002;3:3-10.
9. van Tijn DA, de Vijlder JJ, Verbeeten B, Jr., Verkerk PH, Vulsma T. Neonatal detection of congenital hypothyroidism of central origin. J Clin Endocrinol Metab. 2005;90(6):3350-9.
10. Almenares P, Lechuga MF, Marrero N, Solís RL, Frómeta A, Robaina R, et al. Development of UMELISA T4 Neonatal for the early diagnosis of congenital hypothyroidism. In: Levy HL, Hermos RJ, Grady GF, editors. Proceedings of third meeting of the international society for neonatal screening; 1996 Oct 20-23; Boston, EUA. Massachusetts: IKON/MAP; 1996. p. 240-1.
11. Marrero N, Zulueta O, González EC, Montalvo G, Pérez PL, Melchor A. Obtention of 17-hydroxyprogesterone/alkaline phosphatase conjugates for use in the diagnosis of congenital adrenal hyperplasia. Rev CENIC Cienc Biol. 2003;34(1):23-8.
12. Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45(1):255-68.
13. Clinical and Laboratory Standards Institute (CLSI). Protocols for determination of limits of detection and limits of quantitation; approved guideline. CLSI document EP17-A. Wayne, PA: CLSI; 2004.
14. Dussault JH, Coulombe P, Laberge C, Letarte J, Guyda H, Khoury K. Preliminary report on a mass screening program for neonatal hypothyroidism. J Pediatr. 1975;86(5):670-4.
15. Revised guidelines for neonatal screening programmes for primary congenital hypothyroidism. Working Group on Neonatal Screening of the European Society for Paediatric Endocrinology. Horm Res. 1999;52(1):49-52.
16. Lafranchi SH, Snyder DB, Sesser DE, Skeels MR, Singh N, Brent GA, et al. Follow-up of newborns with elevated screening T4 concentrations. J Pediatr. 2003;143(3):296-301.
17. Prieto L, Gruñeiro de Papendieck L, Chiesa A, Bergadá C. Screening of congenital hypothyroidism (CH): experience in cord blood. In: Levy HL, Hermos RJ, Grady GF, editors. Proceedings of the third meeting of the international society for neonatal screening; 1996 Oct 22-23; Boston, EUA. Massachusetts: IKON/MAP; 1996. p. 271-2.
18. Derksen-Lubsen G, Verkerk PH. Neuropsychologic development in early treated congenital hypothyroidism: analysis of literature data. Pediatr Res. 1996;39(3):561-6.
19. Álvarez MA, Carvajal-Martínez F, Pérez-Gesén C, Olivares-Torres A, Fernández-Yero JL, Robaina-Álvarez R, et al. Pronóstico de la cognición en el hipotiroidismo congénito tra-tado precozmente. Hipótesis del doble efecto. Rev Neurol. 2004;38(6):513-7.
20. LaFranchi SH, Austin J. How should we be treating children with congenital hypothyroidism? J Pediatr Endocrinol Metab. 2007;20(5):559-78.
21. Mei JV, Alexander JR, Adam BW, Hannon WH. Use of filter paper for the collection and analysis of human whole blood specimens. J Nutr. 2001;131(5):1631S-6S.
22. Gonzalez EC, Marrero N, Frometa A, Herrera D, Castells E, Perez PL. Qualitative colorimetric ultramicroassay for the detection of biotinidase deficiency in newborns. Clin Chim Acta. 2006;369(1):35-9.
23. Herrera D, González EC, Pérez PL, Frómeta A. Normalización del UMELISA TSH NEONATAL® a discos de 3 mm. Rev CENIC Cienc Biol. 2006;37(2):119-24.
24. Gonzalez EC, Marrero N, Perez PL, Frometa A, Zulueta O, Herrera D, et al. An enzyme immunoassay for determining 17alpha-hydroxyprogesterone in dried blood spots on filter paper using an ultramicroanalytical system. Clin Chim Acta. 2008;394(1-2):63-6.
25. Gonzalez EC, Frometa A, del Rio L, Castells E, Robaina MS, Garcia SM, et al. Cuban neonatal screening of phenylketonuria using an ultramicro-fluorometric test. Clin Chim Acta. 2009;402(1-2):129-32.
26. Frometa A, Reyes EC, Castells E, Tejeda Y, Almenares P, Perez PL. Quantitative ultramicrotest for newborn screening of galactosemia in Cuba. J Perinat Med. 2011;39(1):77-81.
27. Gruneiro-Papendieck L, Prieto L, Chiesa A, Bengolea S, Bossi G, Bergada C. Usefulness of thyroxine and free thyroxine filter paper measurements in neonatal screening for congenital hypothyroidism of preterm babies. J Med Screen. 2000;7(2):78-81.
28. Larsson A, Ljunggren JG, Ekman K, Nilsson A, Olin P. Screening for congenital hypothyroidism. I. Laboratory results of a pilot study based on dried blood samples collected for PKU screening. Acta Paediatr Scand.1981;70(2):141-6.
29. Gonzalez RR, Robaina R, Rodriguez ME, Blanca S. An enzyme immunoassay for determining total thyroxine in human serum using an ultramicroanalytical system. Clin Chim Acta. 1991;197(3):159-70.
Received in June, 2012.
Accepted in January, 2013.
Elisa Castells. Laboratorio de Pesquisa Neonatal, Centro de Inmunoensayo, CIE. Calle 134 y Ave. 25, AP 6653, Cubanacán, Playa, La Habana, Cuba. E-mail: iqtsh1@cie.sld.cu.