My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Cuban Journal of Agricultural Science
On-line version ISSN 2079-3480
Cuban J. Agric. Sci. vol.50 no.1 Mayabeque Jan.-Mar. 2016
Cuban Journal of Agricultural Science, 50(1): 77-87, 2016, ISSN: 2079-3480
ORIGINAL ARTICLE
Effect of a raw saponin extract on ruminal microbial population and in vitro methane production with star grass (Cynodon nlemfuensis) substrate
Efecto de un extracto crudo de saponinas en la población microbiana ruminal y en la producción de metano in vitro con sustrato de pasto estrella (Cynodon nlemfuensis)
Juana Galindo,I Niurca González,I A. Luiz Abdalla,II Mariem Alberto,I R.C. Lucas,II K. C. Dos Santos,II M. Regina Santos,II P. Louvandini,II O. Moreira,I Lucía Sarduy,I
IInstituto de Ciencia Animal, Apartado Postal 24, Mayabeque, Cuba.
IICentro de Energía Nuclear para la Agricultura (CENA), Universidad de Sâo Paolo, Piracicaba, Brasil.
ABSTRACT
An experiment was conducted under in vitro conditions for determining the effect of a raw saponin extract on the ruminal microbial population and in vitro methane production with star grass (Cynodon nlemfuensis) substrate. Treatments were designed according to the quantity of saponin extract: 1) control, without saponins, 2) 0.6 %, 3) 1.2 % and 4) 1.8 % of the DM of raw saponin extract. The basal diet was star grass (C. nlemfuensis). The saponin extract was obtained from Sapindus saponaria fruit and its saponin content was of 139.5 mg, equivalent of diogenin.mL-1. There was a reduction in protozoa population, regardless the saponin level. Its effect on the main cellulolytic bacteria, determined by PCR-RT, showed that the amount of Fibrobacter succinogenes was not modified while the values of Ruminococcus albus were 25.92; 26.72; 25.2 and 22.35 CT for the levels 0; 0.6; 1.2 and 1.8 %, respectively. The acetic acid concentration was not modified by the saponins; the propionic was reduced with 1.2 % inclusion. The concentration of valeric acid was 0.68; 0.62; 0.52 and 0.49 mmol.L-1 for 0, 0.6; 1.2 and 1.8 % of saponin extract, respectively. Saponins increased methanogenic representation and methane production. It is concluded that the saponin extract modulates the fermentative process on reducing protozoa, does not modify the presence of F. succinogenes and decreases that of R. albus, probably due to the fact that both utilize the same resource, space and carbon source in the rumen. The quantity of methanogens was higher with 1.2 and 1.8 % coinciding with the highest methane production.
Key words: Fibrobacter succinogenes, Ruminococcus albus, star grass, methanogens.
RESUMEN
Para determinar el efecto de un extracto crudo de saponinas en la población microbiana ruminal y producción de metano in vitro con sustrato de pasto estrella (Cynodon nlemfuensis) se condujo un experimento en condiciones in vitro. Los tratamientos se diseñaron de acuerdo con la cantidad de extracto de saponinas: 1) control, sin saponinas, 2) 0.6 %, 3) 1.2 % y 4) 1.8 % de la MS de extracto crudo de saponinas. La dieta base fue pasto estrella (Cynodon nlemfuensis). El extracto de saponinas se obtuvo a partir del fruto de Sapindus saponaria y su contenido en saponinas fue 139.5 mg, equivalente de diogenina.mL -1. Se encontró reducción en la población de protozoos, independientemente del nivel de saponinas. Su efecto en las principales bacterias celulolíticas, determinadas mediante PCR-RT, mostró que la cantidad de Fibrobacter succinogenes no se modificó, mientras que los valores de Ruminococcus albus fueron 25.92; 26.72; 25.2 y 22.35 CT para los niveles 0; 0.6; 1.2 y 1.8 %, respectivamente. La concentración de acido acético no se modificó por las saponinas, el propiónico se redujo con el 1.2 % de inclusión. La concentración de ácido valérico fue 0.68; 0.62; 0.52 y 0.49 mmol.L-1 para los niveles de 0, 0.6; 1.2 y 1.8 % de extracto de saponinas, respectivamente. Las saponinas incrementaron la representación de metanógenos y la producción de metano. Se concluye que el extracto de saponinas modula el proceso fermentativo al reducir los protozoos, no modifica la presencia de F. succinogenes y disminuye la de R. albus, debido probablemente a que ambas utilizan el mismo recurso, espacio y fuente de carbono en el rumen. La cantidad de metanógenos fue superior con 1,2 y 1.8 %, lo que coincidió con mayor producción de metano.
Palabras clave: Fibrobacter succinogenes, Ruminococcus albus, pasto estrella, metanógenos.
INTRODUCTION
In recent years has been increased the use of trees and shrubs as diet supplement of domestic animals. One characteristics of these plants is that they possess secondary metabolites that can modify the degradation and passage rate of nutrients through the gastrointestinal tract (Pedraza 2000, García et al. 2008 and Delgado et al. 2011), as result of the direct effect on the ruminal ecology.
The presence of secondary metabolites, in particular saponins can act on the protozoa population and produce its lysis. The effect of these metabolites on fungi population, cellylolytic bacteria and methanogens is indirect because protozoa engulf huge amounts of these microbial groups and, consequently, improve the digestive efficiency of the feeds (Coleman 1980, La O et al. 2008 and Galindo et al. 2014).
Saponins are biomolecules that can affect ruminal fermentation in function of its structure, biological activity and concentration (Hart et al. 2008). The majority of the biological effects of these compounds can be attributed to its toxic action and to its defaunating effects in the rumen (Newbold et al. 1997), although there are evidences of the influence of these compounds on other microbial groups.
Probably the saponin capacity of joining to sterols provokes the lysis of the cell membranes of the protozoa, although there are also indications that these compounds affect the mobility of ciliate protozoa and the contraction of the holotrics, Isotricha prostoma and Dasytricha ruminantium. The referred effects are transitory and disappear when animals stop consuming this compound. In other studies it has been demonstrated that several of the main fungi species of the rumen are sensible to saponins and its growth is inhibited at very low concentrations of these secondary metabolites (Wina et al. 2005).
Cunningham (2009) indicated that protozoa ingest large volumes of bacteria and maintain constant its population in the rumen so that defaunation implicate the disappearance of the ecological relationships (predation and competition) affecting the type, genetic distribution and metabolic activity of the fungi and bacterial population of the ruminal ecosystem. The author reports that protozoa of type A, among them Polyplastrom multivesiculatum, act as predators of the cellulolytic bacteria B. fibrisolvens, Ruminococcus flavefaciens regarding the amylolytics Selenomonas ruminantium, Streptococcus bovis or the acidophilic species as Megasphaera elsdenii. However, protozoa type B, including cellulolytic protozoa as Epidinium ecaudatum, Eremoplastron bovis and Eudiplodinium maggii, although acting also as predators of the cellulolytic bacteria are more slow regarding this process.
This study aimed at determining the effect of a raw saponin extract on the ruminal microbial population and in vitro methane production with star grass (C. nlemfuensis) substrate.
MATERIALS AND METHODS
For fulfilling the objective of this research, a saponin extraction process was designed from S. saponaria fruit.
Fruits (30 in total) came from adult trees of approximately 3 m height from the Botanical Garden of Havana. Samples were collected in April-June, 2012. Once collected the fruits were taken to the laboratory of rumen microbiology of the Institute of Animal Science where the preparation process of the sample was carried out. For that, fruits were cut in small pieces with laboratory scissors. The cut material was sun-dried for three days and ground in a hammer mill with a 2 mm sieve until reducing its size to fine dust. This process was executed by ethanol extractions, evaporation, decanting with n-hexane, degreasing with n-butanol water saturated. For determining saponin concentration in the extract, the technique of Hansen et al. (2003) was used. The reaction was made with vanillin solution in acid mean and the determination with the standard curve of diosgenin.
Experimental procedure. The in vitro technique of gas production described by Theodorou et al. (1994) was used. For that, 24 bottles of 160 mL containing 0.5 g of the experimental diets, 50 mL of buffer solution and 25 mL of ruminal liquor (Bueno et al. 2005, Makkar 2005 and Longo 2006) were employed. Also two bottles, without substrate, were utilized as blanks for correcting the effect of the ruminal liquor on the volumes of gas produced. Gas pressure did not exceed 7 psi for avoiding the inhibition of the process of microbial fermentation.
The battery of experimental bottles was integrated by 24 units. For each experimental treatment six bottles were used. There were three replications.
As donor animals of the ruminal liquor three adult female sheep of Santa Inés breed canulated at the dorsal sac of the rumen were employed. The ruminal liquor was extracted to fasting animals through the cannula. With the help of a forceps the solid fraction was extracted and the liquid was collected with a vaccum pump. The ruminal liquor was kept in thermo for guaranteeing the temperature (39 ºC) and anaerobic conditions during the transportation to the laboratory. To the solid fraction a small portion of the buffering solution of Menke and Steingass (1988) was added and agitated for some seconds in a domestic blender for detaching the microorganisms joined to the fiber. Later, the filtrate of this portion is incorporated to the liquid fraction and it was agitated before its use. The ruminal liquor obtained was maintained in CO2 atmosphere until the preparation of the experimental units.
The experimental units were placed for incubation for 24 h in a forced air stove.
Treatments. Consisted of different saponin dosages determined as percentage of the incubation DM:
1) Control, star grass (SG), 0 % saponins
2) SG + 0.6 % saponin extract
3) SG + 1.2 % saponin extract
4) SG + 1.8 % of saponin extract
The saponin dosages were selected according to the results of Abreu et al. (2003), Guo et al. (2008).
The chemical composition of the star grass was established by AOAC (2012). The fibrous fraction was quantified according to the protocol described by van Soest et al. (1991). Its composition in g/kg DM was: OM (947.52), CP (89.47), EE (25.32), ash (52.44), NDF (585.2) and lignin (85.2 g/kg DM), respectively.
Saponin concentration in the raw extract was 139.5 mg equivalent of diogenin.mL-1.
Samplings. Sampling times for the determination of gas production in dynamics were 4, 8, 12 and 24 h. For determining CH4, 2.5 mL of gas were collected in each of the hours in which gas production was measured for forming a pool. These were introduced in a vacuum test tube for establishing methane production during 24 h of fermentation.
Determinations. Gas and methane productions were determined. At 24 of fermentation, bottles containing the samples were placed in ice for stopping the fermentative process. From the supernatant samples were taken for establishing SCFA (total and individual) and N-NH3 and for quantifying the populations of F. succinogenes, R. albus, total methanogens and protozoa.
Molecular monitoring of the microorganism populations. The quantification of F. succinogenes, R. albus and total methanogens were carried out by PCR-RT, according to Makkar and McSweeney (2005). DNA was practiced with the extraction kit (Kit PowerLyzerTMPower Soil. MOBIO). The extracted DNA was amplified for each of the specific primers (total bacteria, fungi, F. succinogenes, R. flavefaciens and methanogenic bacteria). The primers were used at a concentration of 10 mM and the Sybr Green 490 was used. The final volume of the reaction was 10 µL.
The DNA amplification was carried out through the following program: 1 cycle of 95 ºC 10 min, 40 cycles of 95 ºC 15 s, 60 ºC 30 s, 72 ºC 30 s and 1 cycle of 95 ºC 15 s, 60 ºC 1 min and 95 ºC 15 s.
The amount of microbial populations studied was expressed as proportion of total bacteria (ΔCt). These values of ΔCt were calculated by the difference between the Ct value (threshold cycle) of the tarjet gene and the reference gene (16S rRNA of bacterium). The ΔΔCt was determined by difference between the ΔCt of the tarjet groups of the experimental diets and the ΔCt of the tarjet groups of the control diet. The percentage of cellulolytic and methanogenic bacteria relative to the total bacteria population was calculated from the values of ΔCt as 100 x (2ΔCt)-1 and the expression of the tarjet groups, regarding the control treatment as 2 –ΔΔCt (Denman and McSweeney 2006).
Methane determination. Methane was determined by gas chromatography in a Philips PU-4400 chromatograph with a 25 m capillary column and DB-1 stationary phase. A FID detector was used and H2 as carrier (1 mL.min-1). The temperature of the detector and injector was of 200 ºC and the column temperature was 60 ºC. It was injected 1 mL of gas contained in the syringe. Calculations of the methane concentration were realized from the equation obtained in the calibration curve:
y = 0.0001x + 2.8515 (R2 = 0.99)
A completely randomized experimental design was applied. The statistical analysis was made according to the design used. For the particular case of gas production, results were analyzed as random blocks. Each incubation group was considered as a block. The levels of saponin extract were considered as treatments and the average value of the bottles per treatment of each incubation group as the experimental unit. Duncan (1955) multiple range tests weas used for identifying differences between means.
For the variables of molecular indicators the theoretical suppositions of the analysis of variance were verified from the Shapiro’s and Wilk (1965) tests for the normality of the errors and Levene (1960) test for the homogeneity of the variance, the variables analyzed did not fulfill the theoretical suppositions of ANAVA, therefore, the ln transformation was employed for the delta CT variables. The subsequent verification demonstrated the fulfillment of the suppositions. For the CT indicator it was unnecessary to realize transformations owing to the fact that the theoretical suppositions were accomplished.
The statistical INFOSTAT package version 2001 from Di Rienzo et al. (2001) was utilized.
RESULTS AND DISCUSSION
The incorporation of 0.6; 1.2 and 1.8 % of raw saponin extracted from S. saponaria fruit in fermentations with star grass substrate reduced the protozoa population regarding the control treatment without saponins (table 1).
According to Klita et al. (1996) a possible mechanism that could account for the negative effect of the saponins on ciliate protozoa is the mass change produced on the permeability of the cell membrane due to the formation of complexes with the cholesterol and some proteins of this membrane. Protozoa, among the ruminal microorganisms, are especially susceptible to this change in the properties of the cell wall. In studies of Galindo et al. (2000) it was demonstrated that the inclusion of S. saponaria leaves in in vitro fermentations with star grass reduced protozoa population and modified its representation in species. Investigations of González et al. (2007) reported the effect of this plant on gas and methane production coinciding with the studies of Abreu et al. (2003) referring the assessment of the effect of the pericarp and S. saponaria fruit on the ruminal Holotrics ciliate population.
Hess et al. (2003 and 2004) results suggest that the saponin effects on ruminal protozoa could depend on the quality of the diet consumed by the animals, mainly when the CP content is high. In any case, it is important to consider that defaunation reduces CH4 enteric emissions due to the flow of microbial cells from the rumen and to the reduction of acetate/propionate relationship, which are events considered as electron sinks (Leng 2014).
In table 1 is shown that there were no effects of the saponin level on the ruminal pH (P < 0.05). This result agrees with those obtained by Abreu et al. (2003), Díaz et al. (1993) and Navas-Camacho et al. (1994) in vivo, as well as in studies realized in vitro by Hess et al. (2003) who did not find significant changes in the pH of the ruminal liquor by effect of the pericarp and the entire fruit of S. saponaria, respectively.
Ammonia concentration was increased with 0.6 % saponins, 1.2 and 1.8 % did not differ from the control. Similarly there was effect of the saponin extract on the SCFA concentration. Supplementation with 1.2 % saponins reduced the concentration of total SCFA in the rumen, while 0.6 % had no effects. The amount of 1.8 % showed intermediate values between the control without saponins and 1.2 %.
Regarding the concentrations of the different SCFA, the saponins did not produce effect on the concentration of acetic, isobutyric, butyric and isovaleric acids. However, from 1.2 % the concentrations of propionic and valeric acids were reduced. The SCFA decreased from 72.53 mmol.L-1 to 66.55 with 1.2 %. Santoso et al. (2007) assessing the effect of 13, 19.5 and 26 mg of saponins kg LW-1 reported reductions in the SCFA concentration while the percentage of butyrate and isoacids, as well as the number of protozoa was linearly reduced regarding the saponin contents.
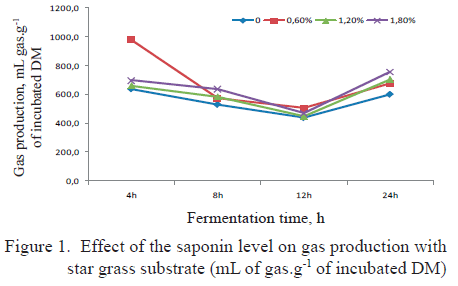
The effect of the saponin level on gas production and total gas production accumulated in 24 h of fermentation on fermenting star grass is shown in figure 1. Net production (mL.g-1 fermented DM) with star grass substrate is shown in figure 2.

In figure 3 can be observed the saponin effect on methane production at 24 h when star grass is used as fermentation substrate. In all treatments including saponins in the diet, methane production was increased.
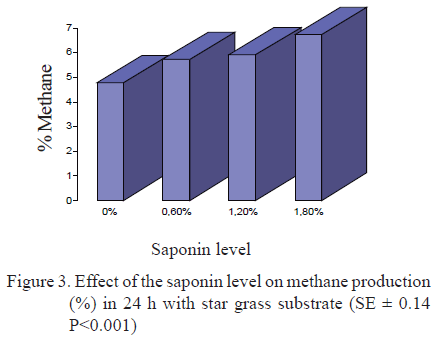
These results do not coincide with the reports of previous research studies in which saponins are indicated to reduce methane production at rumen level (Wang et al. 2006 and 2009). This is related to lower rumen protozoa population (Klita et al. 1996, Lila et al. 2003, 2005, Hess et al. 2004 and Wallace 2004).
In research studies conducted by González et al. (2007) the supplementation with 25 % S. saponaria to a P. purpureum cv. Cuba CT-115, found reductions in ruminal methane production. They attributed this fact to the high concentrations of saponins of S. saponaria.
Investigations of Rodríguez and Fondevilla (2012) who supplemented with Enterolobium cyclocarpum and raw saponin extracts demonstrated that these reduce methane production by direct effect on the protozoa population. Anyway, the complexity of these processes at the rumen led to the continuity of studies directed toward the assessment of saponin inclusion levels different to those used in this experiment, as well as different nitrogen/energy relationships in the diets.
The use of the technique PCR-RT for monitoring two rumen species of cellulolytic bacteria, F. succinogenes and R. albus and the population of cellulolytic and methanogen fungi was previously described by Denman and McSweeney (2005, 2006). Since then, González et al. (2006) and González et al. (2010) used it in Cuba.
In this experiment, the identification and quantification by PCR-RT of the total bacteria and microorganism populations involved in fiber degradation in the rumen (F. succinogenes, R. albus and fungi) and of the total methanogen populations (table 2) showed that saponins did not have effects on the populations of total bacteria, fungi and the cellulolytic bacteria F. succinogenes.
There are study reports, developed by different groups of researchers, referring that saponins have antimicrobial activity against Gram+ bacteria, in relation to Gram- (Patra and Saxena 2009).Wallace (2004) reported that saponins inhibit the growth of Butyrivibrio fibrisolvens and Streptococcus bovis. Patra and Yu (2013) observed that the addition of low doses of saponins, combined with nitrate, increase the population of F. succinogenes, while using high doses, the population is reduced. Zhou et al. (2011), when evaluating saponins extracted from tea (Camellia sinensis), confirmed a decrease of Ruminococcus flavefaciens and Fibrobacter succinogenes, and an increase of B. fibrisolvens, without any effect on Ruminococcus albus.
The 0.6 % level of saponin extract did not modify methanogen population regarding the control without this biomolecule. When 1.2 % of saponins were included there was an increase in the methanogen population. With 1.8 % this microbial group of the rumen attained intermediate population values. R. albus decreased its population in the rumen when 1.8 % saponins was incorporated in the star grass diet.
The representation percentage of methanogens, R. albus and F. succinogenes, regarding the total bacteria population is shown in table 3. As can be observed, there was no effect of the saponins on the methanogens and F. succinogenes. However, there were interesting modifications in the R. albus population. With 0.6 % there was an increase of its representation although not differing from the treatment without saponins. The 1.2 % level produced a decrease regarding the 0.6 % level but no significant differences regarding the treatment without saponins. With the inclusion of 1.8 % saponins this cellulolytic rumen bacteria species reduced its representation.
Values of delta CT (table 4) and the expression related to the control of microbial populations of the rumen identified and quantified through molecular tools (table 5) indicated that all treatments including saponins showed the same difference and expression relative to the control treatment for the microbial populations of methanogens and F. succinogenes. On the contrary there were effects that decreased the R. albus population diminishing in the same magnitude as increasing levels of saponins were included in the diet.
It is concluded that the raw saponin extract modulate the fermentative process by reducing protozoa. It does not modify the presence of F. succinogenes, decreases R. albus, probably due to the fact that both bacteria use the same resource, space and carbon source in the rumen. The quantity of methanogens was higher with 1.2 and 1.8 % coinciding with higher methane production.
REFERENCES
Abreu, A., Fornaguera, J. E. C., Kreuzer, M., Lascano, C. E., Diáz, T. E., Cano, A. & Hess, H.-D. 2003. “Efecto del fruto, del pericarpio y del extracto semipurificado de saponinas de Sapindus saponaria sobre la fermentación ruminal y la metanogénesis in vitro en un sistema RUSITEC”. Revista Colombiana de Ciencias Pecuarias, 16 (2): 147–154, ISSN: 0120-0690.
Bueno, I. C. S., Cabral Filho, S. L. S., Gobbo, S. P., Louvandini, H., Vitti, D. M. S. S. & Abdalla, A. L. 2005. “Influence of inoculum source in a gas production method”. Animal Feed Science and Technology, 123–124: 95–105, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2005.05.003.
Coleman, G. S. 1980. “Rumen ciliate protozoa”. In: Lumsden W. H. R., Muller R. & Bakes J. R. (eds.), Advances in parasitology, vol. 18, London: Academic Press Inc. (London) Ltd., pp. 121–173, ISBN: 978-0-12-031718-9, Available: <http://www.cabdirect.org/abstracts/19812902063.html>, [Consulted: February 21, 2016].
Cunningham, J. G. 2009. Fisiología veterinaria. 4th ed., Barcelona: Elsevier, 720 p., ISBN: 978-84-8086-391-9, Available: <https://www.amazon.es/Fisiolog%C3%ADa-veterinaria-incluye-evolve-Cunningham/dp/8480863919>, [Consulted: May 3, 2016].
Delgado, D. C., Galindo, J., González, R., González, N., Scull, I., Dihigo, L., Cairo, J., Aldama, A. I. & Moreira, O. 2011. “Feeding of tropical trees and shrub foliages as a strategy to reduce ruminal methanogenesis: studies conducted in Cuba”. Tropical Animal Health and Production, 44 (5): 1097–1104, ISSN: 0049-4747, 1573-7438, DOI: 10.1007/s11250-011-0045-5.
Denman, S. E. & McSweeney, C. S. 2005. “Quantitative (real-time) PCR”. In: Makkar H. P. S. & McSweeney C. S. (eds.), Methods in Gut Microbial Ecology for Ruminants, Berlin/Heidelberg: Springer-Verlag, ISBN: 978-1-4020-3790-0, Available: <http://link.springer.com/10.1007/1-4020-3791-0>, [Consulted: February 21, 2016].
Denman, S. E. & McSweeney, C. S. 2006. “Development of a real‐time PCR assay for monitoring anaerobic fungal and cellulolytic bacterial populations within the rumen”. FEMS Microbiology Ecology, 58 (3): 572–582, ISSN: 1574-6941, DOI: 10.1111/j.1574-6941.2006.00190.x.
Díaz, A., Avendaño, M. A. & Escobar, A. 1993. “Evaluation of Sapindus saponaria as a defaunating agent and its effects on different ruminal digestion parameters”. Livestock Research for Rural Development, 5 (2): 1–6, ISSN: 0121-3784.
Di Rienzo, J. A., Casanoves, F., Balzarini, M. G., González, L., Tablada, M. & Robledo, C. W. 2001. InfoStat. version 2001, [Windows], Universidad Nacional de Córdoba, Argentina: Grupo InfoStat, Available: <http://www.infostat.com.ar/>.
Duncan, D. B. 1955. “Multiple Range and Multiple F Tests”. Biometrics, 11 (1): 1–42, ISSN: 0006-341X, DOI: 10.2307/3001478.
Francis, G., Kerem, Z., Makkar, H. P. S. & Becker, K. 2002. “The biological action of saponins in animal systems: a review”. British Journal of Nutrition, 88 (06): 587–605, ISSN: 1475-2662, DOI: 10.1079/BJN2002725.
Galindo, J., Aldama, A. I., Marrero, Y. & González, N. 2000. “Efecto de Sapindus saponaria en los géneros de protozoos y poblaciones de bacterias ruminales”. Cuban Journal of Agricultural Science, 34: 353–358, ISSN: 2079-3480.
Galindo, J., González, N., Marrero, Y., Sosa, A., Ruiz, T., Febles, G., Torres, V., Aldana, A. I., Achang, G., Moreira, O., Sarduy, L. & Noda, A. C. 2014. “Effect of tropical plant foliage on the control of methane production and in vitro ruminal protozoa population”. Cuban Journal of Agricultural Science, 48 (4): 359–364, ISSN: 2079-3480.
García, D. E., Medina, M. G., Cova, L. J., Torres, A., Soca, M., Pizzani, P., Baldizán, A. & Domínguez, C. E. 2008. “Preferencia de vacunos por el follaje de doce especies con potencial para sistemas agrosilvopastoriles en el Estado Trujillo, Venezuela”. Pastos y Forrajes, 31 (3): 1–1, ISSN: 0864-0394.
González, N., Galindo, J., Aldana, A. I., Moreira, O., Abdalla, L. A. & Santos, M. R. 2010. “Evaluación de diferentes variedades de Morera (Morus alba) en el control de la metanogénesis ruminal en búfalos de río in vitro”. Cuban Journal of Agricultural Science, 44 (1): 37–41, ISSN: 2079-3480.
González, N., Galindo, J., González, R., Sosa, A., Moreira, O., Delgado, D., Martín, E. & Sanabria, C. 2006. “Utilización de la técnica de PCR en tiempo real y de la producción de gas in vitro para determinar el efecto del ácido bromoetano sulfónico en la metanogénesis y la población microbiana ruminal”. Cuban Journal of Agricultural Science, 40 (2): 183–189, ISSN: 2079-3480.
González, R., Delgado, D. & Cairo, J. 2007. “Efecto de la inclusión de Sapindus saponaria en la producción de gas y metano en la fermentación in vitro de Pennisetun purpureum cv Cuba CT-115”. Cuban Journal of Agricultural Science, 41 (1): 39–41, ISSN: 2079-3480.
Guo, Y. Q., Liu, J. X., Lu, Y., Zhu, W. Y., Denman, S. E. & McSweeney, C. S. 2008. “Effect of tea saponin on methanogenesis, microbial community structure and expression of mcrA gene, in cultures of rumen micro-organisms”. Letters in Applied Microbiology, 47 (5): 421–426, ISSN: 1472-765X, DOI: 10.1111/j.1472-765X.2008.02459.x.
Hansen, I., Brimer, L. & Mølgaard, P. 2003. “Herbivore-deterring secondary compounds in heterophyllous woody species of the Mascarene Islands”. Perspectives in Plant Ecology, Evolution and Systematics, 6 (3): 187–203, ISSN: 1433-8319, DOI: 10.1078/1433-8319-00077.
Hart, K. J., Yáñez-Ruiz, D. R., Duval, S. M., McEwan, N. R. & Newbold, C. J. 2008. “Plant extracts to manipulate rumen fermentation”. Animal Feed Science and Technology, 147 (1–3): 8–35, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2007.09.007.
Hess, H. D., Beuret, R. A., Lotscher, M., Hindrichsen, I. K., Machmuller, A., Carulla, J. E., Lascano, C. E. & Kreuzer, M. 2004. “Ruminal fermentation, methanogenesis and nitrogen utilization of sheep receiving tropical grass hay-concentrate diets offered with Sapindus saponaria fruits and Cratylia argentea foliage”. Journal of Animal Science, 79 (1): 177–189, ISSN: 0021-8812, 1525-3163.
Hess, H. D., Kreuzer, M., Dı́az, T. E., Lascano, C. E., Carulla, J. E., Soliva, C. R. & Machmüller, A. 2003. “Saponin rich tropical fruits affect fermentation and methanogenesis in faunated and defaunated rumen fluid”. Animal Feed Science and Technology, 109 (1–4): 79–94, ISSN: 0377-8401, DOI: 10.1016/S0377-8401(03)00212-8.
Klita, P. T., Mathison, G. W., Fenton, T. W. & Hardin, R. T. 1996. “Effects of alfalfa root saponins on digestive function in sheep”. Journal of Animal Science, 74 (5): 1144–1156, ISSN: 0021-8812, DOI: /1996.7451144x.
La O, O., García, R., Ruiz, O., Castillo, Y., Muro, A., Rodríguez, C., Arzola, C., González, H. & Ortiz, B. 2008. “In vitro ruminal fermentative potential of two trees (Pithecellobium dulce and Tamarindos indica) of importance for livestock rearing in fragile, saline ecosystems with high drought, located in the eastern part of Cuba”. Cuban Journal of Agricultural Science, 42 (1): 57–59, ISSN: 2079-3480.
Latimer, G. W. 2012. Official Methods of Analysis of AOAC International. 19th ed., Gaithersburg, Md.: AOAC International, ISBN: 978-0-935584-83-7, Available: <http://www.amazon.com/Official-Methods-Analysis-OFFICIAL-ANALYSIS/dp/0935584838/ref=pd_sim_sbs_14_1?ie=UTF8&dpID=31iikC-xl2L&dpSrc=sims&preST=_AC_UL160_SR160%2C160_&refRID=101AB94246X0EM9N7XMW>, [Consulted: April 1, 2016].
Leng, R. A. 2014. “Interactions between microbial consortia in biofilms: a paradigm shift in rumen microbial ecology and enteric methane mitigation”. Animal Production Science, 54 (5): 519–543, ISSN: 1836-0939, DOI: http://dx.doi.org/10.1071/AN13381.
Levene, H. 1960. “Robust tests for the equality of variance”. In: Olkin I., Contributions to Probability and Statistics: Essays in Honor of Harold Hotelling, Stanford University Press, pp. 278–292, ISBN: 978-0-8047-0596-7, Available: <https://books.google.com.cu/books?hl=es&lr=&id=ZUSsAAAAIAAJ&oi=fnd&pg=PA3&dq=Contributions+to+Probability+and+Statistics&ots=GchNfCzOVQ&sig=-W3BeBmuWhTzgRCkktOdQjq0Di4&redir_esc=y#v=onepage&q=Contributions%20to%20Probability%20and%20Statistics&f=false>, [Consulted: April 19, 2016].
Lila, Z. A., Mohammed, N., Kanda, S., Kamada, T. & Itabashi, H. 2003. “Effect of Sarsaponin on Ruminal Fermentation with Particular Reference to Methane Production in vitro”. Journal of Dairy Science, 86 (10): 3330–3336, ISSN: 0022-0302, DOI: 10.3168/jds.S0022-0302(03)73935-6.
Lila, Z. A., Mohammed, N., Kanda, S., Kurihara, M. & Itabashi, H. 2005. “Sarsaponin effects on ruminal fermentation and microbes, methane production, digestibility and blood metabolites in steers”. Asian Australasian Journal of Animal Sciences, 18 (12): 1746, ISSN: 1011-2367.
Longo, C. 2006. “Methane production from tannin rich plants incubated in vitro”. In: British Society of Animal Science Meeting, York: British Society of Animal Science.
Makkar, H. P. S. 2005. “In vitro gas methods for evaluation of feeds containing phytochemicals”. Animal Feed Science and Technology, 123: 291–302, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2005.06.003.
Makkar H. P. S. & McSweeney C. S. (eds.). 2005. Methods in Gut Microbial Ecology for Ruminants. Berlin/Heidelberg: Springer-Verlag, 225 p., ISBN: 978-1-4020-3790-0, Available: <http://link.springer.com/10.1007/1-4020-3791-0>, [Consulted: April 19, 2016].
Menke, K. H. & Steingass, H. 1988. “Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid”. Animal Research and Development, 28 (1): 7–55, ISSN: 0340-3165.
Newbold, C. J., el Hassan, S. M., Wang, J., Ortega, M. E. & Wallace, R. J. 1997. “Influence of foliage from African multipurpose trees on activity of rumen protozoa and bacteria”. The British Journal of Nutrition, 78 (2): 237–249, ISSN: 0007-1145, PMID: 9301414.
Patra, A. K. & Saxena, J. 2009. “The effect and mode of action of saponins on the microbial populations and fermentation in the rumen and ruminant production”. Nutrition Research Reviews, 22 (02): 204–219, ISSN: 1475-2700, DOI: 10.1017/S0954422409990163.
Patra, A. K. & Yu, Z. 2013. “Effective reduction of enteric methane production by a combination of nitrate and saponin without adverse effect on feed degradability, fermentation, or bacterial and archaeal communities of the rumen”. Bioresource Technology, 148: 352–360, ISSN: 0960-8524, DOI: 10.1016/j.biortech.2013.08.140.
Pedraza, R. 2000. Contribución al estudio del valor nutritivo de Gliricidia sepium en la alimentación de rumiantes. Ph.D. Thesis, Instituto de Ciencia Animal, La Habana, Cuba.
Rodríguez, R. & Fondevila, M. 2012. “Effect of saponins from Enterolobium cyclocarpum on in vitro microbial fermentation of the tropical grass Pennisetum purpureum”. Journal of Animal Physiology and Animal Nutrition, 96 (5): 762–769, ISSN: 1439-0396, DOI: 10.1111/j.1439-0396.2011.01161.x.
Santoso, B., Kilmaskossu, A. & Sambodo, P. 2007. “Effects of saponin from Biophytum petersianum Klotzsch on ruminal fermentation, microbial protein synthesis and nitrogen utilization in goats”. Animal Feed Science and Technology, 137 (1–2): 58–68, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2006.10.005.
Shapiro, S. S. & Wilk, M. B. 1965. “An analysis of variance test for normality (complete samples)”. Biometrika, 52 (3-4): 591–611, ISSN: 0006-3444, 1464-3510, DOI: 10.1093/biomet/52.3-4.591.
Theodorou, M. K., Williams, B. A., Dhanoa, M. S., McAllan, A. B. & France, J. 1994. “A simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds”. Animal Feed Science and Technology, 48 (3–4): 185–197, ISSN: 0377-8401, DOI: 10.1016/0377-8401(94)90171-6.
Van Soest, P. J., Robertson, J. B. & Lewis, B. A. 1991. “Methods for Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition”. Journal of Dairy Science, 74 (10): 3583–3597, ISSN: 0022-0302, DOI: 10.3168/jds.S0022-0302(91)78551-2.
Wallace, R. J. 2004. “Antimicrobial properties of plant secondary metabolites”. Proceedings of the Nutrition Society, 63 (04): 621–629, ISSN: 1475-2719, DOI: 10.1079/PNS2004393.
Wang, C. J., Wang, S. P. & Zhou, H. 2009. “Influences of flavomycin, ropadiar, and saponin on nutrient digestibility, rumen fermentation, and methane emission from sheep”. Animal Feed Science and Technology, 148 (2–4): 157–166, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2008.03.008.
Wang, C., Wang, S., Zhou, H. & Glindemann, T. 2006. “Effects of forage composition and growing season on methane emission from sheep in the Inner Mongolia steppe of China”. Ecological Research, 22 (1): 41–48, ISSN: 0912-3814, 1440-1703, DOI: 10.1007/s11284-006-0191-9.
Wina, E., Muetzel, S., Hoftman, E., Makkar, H. P. S. & Becker, K. 2005. “Effect of secondary compound in forages on rumen microorganisms quantified by 16S and 18S rRNA”. In: Makkar H. P. S. & Viljoen G. J. (eds.), Applications of Gene-Based Technologies for Improving Animal Production and Health in Developing Countries, Dordrecht: Springer Netherlands, ISBN: 978-1-4020-3311-7, Available: <http://link.springer.com/10.1007/b105256>, [Consulted: February 21, 2016].
Zhou, Y. Y., Mao, H. L., Jiang, F., Wang, J. K., Liu, J. X. & McSweeney, C. S. 2011. “Inhibition of rumen methanogenesis by tea saponins with reference to fermentation pattern and microbial communities in Hu sheep”. Animal Feed Science and Technology, 166–167: 93–100, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2011.04.007.
Received: December 18, 2014
Accepted: January 28, 2016
Juana Galindo, Instituto de Ciencia Animal, Apartado Postal 24, Mayabeque, Cuba. Email: jgalindo@ica.co.cu














