My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Cuban Journal of Agricultural Science
On-line version ISSN 2079-3480
Cuban J. Agric. Sci. vol.50 no.1 Mayabeque Jan.-Mar. 2016
Cuban Journal of Agricultural Science, 50(1): 151-159, 2016, ISSN: 2079-3480
ORIGINAL ARTICLE
Nutritive quality of foxtail millet (Setaria italica) grass, hay and silage, and seed fatty acid profile
Calidad nutritiva del mijo de cola de zorra (Setaria italica), heno y ensilaje, y perfil de ácidos grasos de las semillas
P.G. Peiretti,I S. Tassone,II
IInstitute of Sciences of Food Production, National Research Council, Largo Braccini 2, 10095 Grugliasco (TO), Italy.
IIDepartment of Agriculture, Forestry, and Food Sciences, University of Torino, Largo Braccini 2, 10095 Grugliasco (TO), Italy.
ABSTRACT
Foxtail millet (Setaria italica) is grown for green grass, hay and silage and is good forage for livestock. The aim of this study was to determine the nutritive quality of foxtail millet grass, hay and silage harvested in the Po valley (northern Italy) at cut or at wilted level. The forage samples used for the trial were cut at budding stage on 8 subplots of 1 m2, randomLy located in 2 x 4 m plots. Two replicates were collected for each wilting levels: at cut (299 g/kg FM) and after 24 h of wilting (487 g/kg FM). Finally, two replicates of hay samples were collected after 36 h of wilting (918 g/kg FM) on the same two subplots, in order to determine chemical composition, gross energy, in vitro true digestibility (IVTD), indigestible neutral detergent fibre (INDF) of hay and grass samples. Moreover, herbage samples for ensiling were taken at cut and after 24 h of wilting, respectively. Foxtail millet forage was ensiled in sterile 2–litre laboratory glass silos. Two replications were performed at each dry matter level, in order to determine chemical composition, gross energy, IVTD, INDF, and fermentation characteristics of silages. The variability in herbage quality characteristics after different wilting periods and the variability in silage characteristics were analyzed for their statistical significance via analysis of variance (ANOVA) to test the effect of wilting level. Herbage wilting had no effects on nutritional parameters, reflecting excellent drying conditions. The results of ensiling indicate that the fermentation of foxtail millet is characterized by the presence of some alcohols and volatile fatty acids. No significant differences were found in crude protein, fibre, gross energy, pH or fermentation characteristics, with the exception of ethanol and butyric acid. The values of IVTD and INDF were improved in wilting silage. The good results obtained in lab–scale silos, with silage characterized by restricted fermentation at high wilting level, would seem to suggest that foxtail millet has the potential for large–scale ensiling, if it is harvested at budding stage and wilted to a DM level of more than 470 g/kg FM. Moreover, because few data are available in literature, the fat content and fatty acid (FA) profile of the seeds used in the agronomic trial were also determined. The lipid content of the foxtail millet seeds was 31.9 g/kg FM, while the predominant FAs were linoleic, oleic, and palmitic acid. The contents of stearic and linolenic acids were 32 and 27 g/kg total FA, respectively. The contents of palmitoleic, arachidic, gadoleic and behenic acid were lower than 7 g/kg total FA.
Key words: Setaria italica, chemical composition, fatty acid, ensilage, forage.
RESUMEN
El mijo de cola de zorra (Setaria italica) se cultiva para pasto verde, heno y ensilaje, y es bueno como forraje para el ganado. El objetivo de este estudio fue determinar la calidad nutritiva del pasto, heno y ensilaje de mijo de cola de zorra, cultivado en el valle del Po (norte de Italia), a nivel de corte o marchitos. Las muestras de forraje utilizadas se cortaron en etapa de yema, en ocho subparcelas de 1 m2, ubicadas al azar en parcelas de 2 x 4 m. Se recolectaron dos réplicas para cada nivel de marchitez: en el momento del corte (299 g/kg MV) y después de 24 horas de marchitez (487 g/kg MV). Por último, se recolectaron réplicas de las muestras de heno después de 36 h de marchitez (918 g/kg MV) en las mismas subparcelas para determinar la composición química, energía bruta, digestibilidad verdadera in vitro (DVIV), fibra detergente neutra no digerible (FDNND) de las muestras de pasto y heno. Además, las muestras de plantas para ensilaje se tomaron en el momento del corte y después de 24 h de marchitez, respectivamente. El forraje de mijo cola de zorra se almacenó en silos de cristal de laboratorio de 2 litros esterilizados. Se realizaron dos réplicas en cada nivel de la materia seca para determinar la composición química, energía bruta, DVIV, FDNND y las características de la fermentación de los silos. La variabilidad en las características de la calidad de las plantas, después de diferentes períodos de marchitez y la variabilidad en las características del ensilaje se analizaron para establecer su significación estadística con el uso de el análisis de varianza (ANOVA) para probar lo efecto del nivel de marchitez. La marchitez de las plantas no influyó en los indicadores nutricionales, lo cual reflejó unas condiciones excelentes para el secado. Los resultados del ensilado indican que la fermentación del mijo de cola de zorra se caracteriza por la presencia de algunos alcoholes y ácidos grasos volátiles. No se encontraron diferencias significativas en la proteína cruda, fibra, energía bruta, características de fermentación o pH, con excepción del etanol y el ácido butírico. Los valores de DVIV y FDNND se mejoraron en el ensilaje de plantas marchitas. Los buenos resultados obtenidos en silos a escala de laboratorio, con ensilaje caracterizado por la fermentación restringida a alto nivel de marchitez, parecieran sugerir que el mijo de cola de zorra tiene el potencial para su ensilaje a gran escala, solo si se cosecha en la etapa de yema y se marchitó a un nivel de MS superior a 470 g/kg MV. Además, debido a que existen pocos datos disponibles en la literatura, se determinaron también el contenido de grasas y el perfil de ácidos grasos (AG) de las semillas utilizadas en la prueba agronómica. El contenido de lípidos de las semillas de mijo de cola de zorra fue 31.9 g/kg MV, mientras que los AG predominantes fueron el linolénico , oleico y palmítico. Los contenidos de los ácidos esteárico y linolénico fueron 32 y 27 g/kg AG totales, respectivamente. Los contenidos de los ácidos palmitoleico, araquídico, gadoleico y behénico fueron menores que 7 g/kg de AG totales.
Palabras clave: Setaria italica, composición química, ácido graso, ensilaje, forraje.
INTRODUCTION
Foxtail millet (Setaria italica) is a summer annual plant belonging to the Labiatae family and is one of the earth’s oldest cultivated crops. It is an important small millet crop, which grows rapidly during warm weather and is one of the most efficient water–use crops, and is therefore widely cultivated in semi–arid regions of the subtropics. This crop germinates comparatively well in drought–prone areas and in very poor soils, where other crops fail to grow (Torbatinejad et al. 2009).
The principal use of foxtail millet, including its forage and silage, is to feed cows and sheep. It is an important grain crop and is grown for silage and hay in North and South America, Australia and North Africa (Anderson 1999). As a short–season forage crop may have the potential to be grown for forage in dry–land cropping systems in the central Great Plains region of North America (Nielsen et al. 2006). Foxtail millet produced dense growth and excellent quality pasture for mid-late summer or hay, which can either be stored or left in windrows (Koch 202). In Italy, the importance of these crops has been diminishing, since production is intended for feeding birds, and it can also be used as a fast–growing meadow grass in summer, to be used from milk to waxy ripeness. Baltensperger (2002) indicates the opportunities for foxtail millet as crops, but also points out the lack of publications in this area. Svirskis (2009), found that threshed straw of foxtail millet according to chemical composition was nearly identical to conventional hay, since their stems with leaves remain green until seed harvesting and concluded that this crop can be used as forage for many animal species as green mass, hay or silage. Fresh forage crops, such as foxtail millet, can be preserved by ensiling, because the major advantages are the retention of the biological properties of green plants and an increase in the nutrient value of the finished silage (Bergero & Peiretti 2011).
The aim of this study was to determine the nutritive quality of foxtail millet grass, hay and silage harvested at cut or at wilted level. Moreover, because few data are available, the fatty acid profile of the seed used in the agronomic trial was also determined.
MATERIALS AND METHODS
Plant material and environmental conditions. The foxtail millet seed was obtained from the Ornitalia Product Service s.a.s. (Colleredo di Monte Albano, Italy). The trials were carried out in the western Po Valley (Italy). No irrigation or fertilisers were applied after spring sowing. The herbage samples (whole plant) were cut at budding stage to a 1–2 cm stubble height with edging shears (0.1 m cutting width) on 8 subplots of 1 m2, randomLy located in 2 x 4 m plots. Sampling for chemical analysis occurred only in favourable weather conditions and in the morning after the disappearance of dew.
Due to the limited dimension of plots only two replicates were collected on two subplots for each wilting level: at cut (299 g/kg FM) and after 24 h of wilting (487 g/kg FM), respectively. Finally, two replicates of hay samples were collected after 36 h of wilting (918 g/kg FM) on the same subplots, in order to determine chemical composition, gross energy, in vitro true digestibility (IVTD), indigestible neutral detergent fibre (INDF) of hay and grass samples. Moreover, herbage samples (whole plant) for ensiling were taken on the other four subplots: two subplots harvested at cut and two subplots harvested after 24 h of wilting, respectively.
Foxtail millet sample was chopped with a paper slicer to a 1–2 cm length and then ensiled in sterile 2–litre laboratory glass silos each, equipped with a lid that enables gas release only. All the silages were preserved by spontaneous fermentation and stored at 20±2 °C in a dark room for 190 days. Silage samples were then immediately frozen and kept at -30 °C until analysis was carried out. Two replications were performed at each dry matter level for a total of 4 laboratory glass silos, in order to determine chemical composition, gross energy, IVTD, INDF, and fermentation characteristics of silages.
Chemical analysis. The herbage and silage samples were immediately dried in a forced draft air oven to a constant weight at 65 °C. Samples were then brought to air temperature, weighed, ground in a Cyclotec mill (Tecator, Herndon, VA, USA) to pass through a 1 mm screen and stored for qualitative analyses. They were analyzed by AOAC (1995) for dry matter (DM), crude protein (CP), and ash by ignition to 550 °C. Neutral detergent fibre (NDF), acid detergent fibre (ADF) and acid detergent lignin (ADL) were determined with the Ankom 200 Fiber Analyser, following the method of Van Soest et al. (1991). The NDF of herbage samples was analyzed without sodium sulfite or α–amylase. Gross energy (GE) was determined using an adiabatic calorimeter bomb (IKA C7000, Staufen, Germany) according to Meineri and Peiretti (2005). All analyses were performed in duplicate. Hemicellulose was calculated as NDF-ADF and cellulose as ADF-ADL.
In vitro digestibility. The herbage, silage and hay samples were also analyzed to determine their in vitro true digestibility (IVTD) and indigestible neutral detergent fibre (INDF) using the DaisyII Incubator (Ankom Technology Corp. Fairport, NY. USA) as described by ANKOM Technology (2005).
IVTD was calculated using the following equation:
IVTD = 1000-(W3-(W1*C1))*1000/(W2*DM),
where W1 was the filter bag weight, W2 was the sample weight, W3 was the final weight (filter bag+residue) after in vitro and sequential treatment with NDF solution, C1 was a comparison of the blank filter bag weight after and before digestion treatment and DM was the dry matter content of the samples.
INDF was calculated using the following equation:
INDF = NDF-dNDF
where NDF was neutral detergent fibre content of the sample and dNDF was digestible neutral detergent fibre (NDF*NDFD/1000).
NDFD was calculated using the following equation:
NDFD = 1000-(W3-(W1*C1))*1000/(W2*NDF)
where W1 is the filter bag weight, W2 is the sample weight, W3 is the final weight (filter bag+residue) after in vitro and sequential treatment with NDF solution, C1 is a comparison of the blank filter bag weight after and before digestion treatment and NDF is neutral detergent fibre content of the sample.
Silage conservation quality analysis. Ammonia, alcohol, volatile fatty acids, and lactic acid concentrations were determined on acid silage extracts: samples of chopped frozen silage were weighed (50 g) in a 400 mL poly–ethylene bag and extracted with 200 mL of 0.1 N H2SO4 at 20 °C for 4 min in a Lab Blender Stomacher 400. The mixture was centrifuged for 5 min at 3000 x g and then filtered through a Schleicher and Schull membrane filter (BA–83, 0.2 µm).
pH was determined on the water extracts of chopped frozen silage using a Crison portable pH meter (Crison Instruments, S.A., Alella, Spain). Ammonia was measured using an ammonia gas–sensing combination electrode (Orion, Model 95–12, Boston, MA) connected to an ion analyzer (Orion, Model 920A, Boston, MA).
A 1 µL aliquot of the extract was injected, using an on–column technique with an auto–sampler (Dani Instruments SpA, ALS 1000, Cologno Monzese, Italy), into a wide–bore capillary column (SGE BP21 25m x 0.53 mm internal diameter and 0.5 µm film thickness; P/N 054474, SGE International, Ringwood, Victoria, Australia) installed in a gas chromatograph (Dani GC 1000 DPC), running in a temperature–programmed mode and equipped with a flame ionization detector and a PTV injection port, used in split mode, with a split vent flow of 100 mL/min. The detector port was set to 240 °C, hydrogen was used as the carrier gas and the oven temperature was programmed to 50 °C for 2 min and from 50 °C to 190 °C at 5 °C per min giving a run time of 30 min. The peak area was measured using a Dani Data Station DDS 1000. Each peak was identified and quantified according to pure standards (Sigma Chemical, St. Louis, MO, USA).
Seed fatty acid analysis. Seeds obtained from the Ornitalia Product Service s.a.s. were ground to pass through a 1 mm screen. Lipid extraction, transesterification and fatty acid methyl esters gas–chromatography determination was performed according to Peiretti et al. (2013).
Statistical analysis. The data shown are mean values of the duplicate analyses performed for each replications sample. The variability in foxtail millet (direct cut, wilted and hay) quality characteristics and the variability in the silage (direct cut and wilted) characteristics were analyzed with analysis of variance (ANOVA) using the Statistical Package for Social Science, v. 11.5 (SPSS 2002) to test the effect of wilting level and using Duncan’s New Multiple Range Test for post-hoc analysis. The significance was established at P<0.05.
RESULTS AND DISCUSSION
Results on different wilting treatments (0, 24, and 36 hours) on foxtail millet herbage are reported in table 1. No significant differences were found in any parameters, with the exception of DM and ash content. Foxtail millet harvested at budding stage is relatively poor in protein, that ranged from 52 to 61 g/kg DM, and rich in fibre (NDF: 674–678 g/kg DM, ADF: 394– 421 g/kg DM, ADL: 49–58 g/kg DM). In agreement with Feedipedia (2012) IVTD varied from 637 to 678 g/kg DM, while INDF ranged from 322 to 363 g/kg of DM. The gross energy ranged from 16.9 to 17.1 MJ/kg DM.
Wilting the plant had little effect on its chemical composition and nutritive quality, as demonstrated for other forages (Rymer 2008). Extending the duration of wilting increased the DM concentration of foxtail millet, but had no impact on protein content, fibre fractions, gross energy and digestibility. Wilting for 36 hours resulted in a higher ash concentration than wilting for 24 hours and fresh herbage (P<0.05), in agreement with Keles et al. (2009). The slight rise in ash concentration with wilting may reflect the respiration of organic matter and/or some level of soil contamination.
In order to avoid border effects, 20 cm of uncutted herbage was left between the neighbouring plots; therefore the fresh matter mass was sufficient only for two replicates for each collected sample. In a similar work, Peiretti et al. (2010), sampled two replicates of white lupin (Lupinus albus L.) for each herbage sample with the aim to determine the effect of harvest date on the chemical composition, gross energy, organic matter digestibility, nutritive value and amino acid content of this plant.
The chemical composition, gross energy, in vitro digestibility and fermentation characteristics of the foxtail millet silages at the two DM levels are reported in table 2.
As far as the chemical composition of foxtail millet silages is concerned, ash and crude protein did not show any significant differences between the two types of silage. Also the fibre fractions were not affected by wilting level and this is in disagreement with other authors (Repetto et al. 2005 and McEniry et al. 2007). The hemicellulose content, which is the most digestible component of NDF and represents a fermentable substrate for lactic acid bacteria during ensiling (McDonald et al. 1991), showed similar values of about 240 g/kg DM. Aerobic exposure resulted in an increase in IVTD (P=0.053) and decrease in INDF (P=0.053).
Forage preservation (wilting, drying and ensiling) of the crop may initiate or accelerate biological processes such as plant respiration, enzymatic and microorganism activities, that are responsible for carbohydrate oxidation, protein degradation and amino acid deamination. A consequence of these processes may be a reduction in the nutritive value of forage preserved compared with standing fresh forage (Repetto et al. 2005 and Bakken et al. 2011). However, foxtail millet preservation had no significant effect on its chemical composition, probably due to the rapid drying to hay under dry sunny weather conditions.
In particular NDF, the main chemical constituent of foxtail millet forage (Torbatinejad et al. 2009), was stable among preservation methods, and wilting had little effect on NDF concentration, in accordance with McEniry et al. (2007). Keady et al. (2000) demonstrated that NDF was more labile in crops harvested at early developmental stages than in more mature crops. Moreover, the study by Torbatinejad et al. (2009), on foxtail millet indicated that a delay in the sowing date and reducing the period of growth cause a reduction in fibre content and an increase in its nutritional value. Indeed, the stage of growth, sowing date and plant density are the most important factors that influence the composition and nutritive value of forages (Howell 1990 and Fulkerson et al. 2008). Unlike other grasses (Bakken et al. 2011), foxtail millet showed no changes in fibre concentrations, although some soluble compounds were respired during wilting. On the contrary, it improved IVTD and reduced INDF, the latter being a major factor limiting the value of forages for animals. ADL is generally the best single predictor of INDF content and hemicellulose the worst (Jančik et al. 2008).
The results obtained in lab–scale silos indicate that the fermentation of foxtail millet is characterized by the presence of alcohols, volatile fatty acids and a lack of lactic acid. No significant differences were found in the fermentation characteristics, with the exception of ammonia, ethanol and butyric acid (P<0.05), which decreased with increasing DM content. Silage quality was characterized by restricted fermentation and low content of butyric acid at the high wilting level and this would seem to suggest that foxtail millet has the potential for large–scale ensiling, if it is harvested at the budding stage and wilted to a DM level of more than 470 g/kg fresh matter.
Further research is required to define the conservation quality in farm–scale silos or in lab silos with additives, that improve silage fermentation when foxtail millet is ensiled at cut or at low wilting levels. Arbabi and Ghoorchi (2008), found that without additives, foxtail millet can be turned into acceptable silage, but the quality, nutrition value, DM digestibility and aerobic stability of silage were improved with the addition of 7.5% molasses. The poor nutritive value of the silage is attributed to the nature and composition of the material ensiled rather than to any defect in the ensilage process itself, and may be a feature of most silage made from tropical grasses (Nussio 2005).
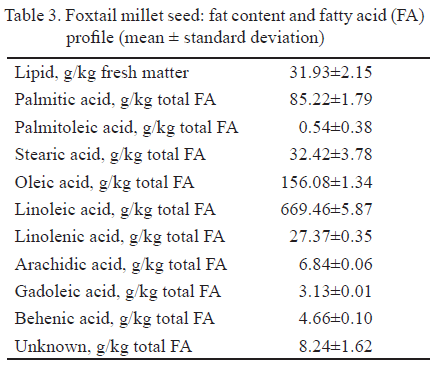
The lipid content of the foxtail millet seeds was 31.9 g/kg fresh matter. The present results are in agreement with the observation of Léder (2009).
The predominant fatty acids in seed were linoleic, oleic, and palmitic acid. The contents of stearic and linolenic acids were 32 and 27 g/kg total fatty acid, respectively. The contents of palmitoleic, arachidic, gadoleic and behenic acid were lower than 7 g/kg total fatty acid (Table 3). Liang et al. (2009), studied the fatty acid content of foxtail millet bran oil and found that it was rich in linoleic acid (665 g/kg total fatty acid) and oleic acid (130 g/kg total fatty acid) and poor in saturated fatty acids such as palmitic acid (64 g/kg total fatty acid) and stearic acid (63 g/kg total fatty acid).
Although foxtail millet is not a high–quality forage, results have shown that it could be considered as an alternative or complement to traditional forage resources. Herbage wilting did not affect nutritional parameters, reflecting excellent drying conditions. The good conservation quality of wilted silage showed that foxtail millet is suitable for conservation through ensiling, provided that silage is well preserved with moderate ensiling losses. Further research is required to define the most appropriate agronomic techniques to follow and the conservation quality in farm–scale silos.
REFERENCES
AOAC. 1995. Official methods of analysis of AOAC. 16th ed., vol. 1, Washington, D.C.: AOAC International, ISBN: 978-0-935584-54-7, Available: <http://www.cabdirect.org/abstracts/19951414840.html>, [Accessed: February 16, 2016].
Arbabi, S. & Ghoorchi, T. 2008. ‘‘The Effect of Different Levels of Molasses as Silage Additives on Fermentation Quality of Foxtail Millet (Setaria italica) Silage’’. Asian Journal of Animal Sciences, 2 (2), pp. 43–50, ISSN: 18191878, DOI: 10.3923/ajas.2008.43.50.
Bakken, A. K., Randby, Å. T. & Udén, P. 2011. ‘‘Changes in fibre content and degradability during preservation of grass–clover crops’’. Animal Feed Science and Technology, 168 (1–2), pp. 122–130, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2011.03.016.
Baltensperger, D. D. 2002. ‘‘Progress with proso, pearl and other millets’’. In: Janick, J. & Whipkey, A. (eds.), Trends in New Crops and New Uses, ASHS Press, pp. 100–103, ISBN: 978-0-09-707565-5.
Bergero, D. & Peiretti, P. G. 2011. ‘‘Intake and Apparent Digestibility of Permanent Meadow Hay and Haylage in Ponies’’. Journal of Equine Veterinary Science, 31 (2), pp. 67–71, ISSN: 0737-0806, DOI: 10.1016/j.jevs.2010.12.006.
Fulkerson, W. J., Horadagoda, A., Neal, J. S., Barchia, I. & Nandra, K. S. 2008. ‘‘Nutritive value of forage species grown in the warm temperate climate of Australia for dairy cows: Herbs and grain crops’’. Livestock Science, 114 (1), pp. 75–83, ISSN: 1871-1413, DOI: 10.1016/j.livsci.2007.04.013.
Howell, T. A. 1990. ‘‘Grain, Dry Matter Yield Relationships for Winter Wheat and Grain Sorghum-Southern High Plains’’. Agronomy Journal, 82 (5), pp. 914–918, ISSN: 0002-1962, DOI: 10.2134/agronj1990.00021962008200050014x.
IBM Corporation. 2002. IBM SPSS Statistics. version 11.5, [Windows], U.S, Available: <http://www.ibm.com> .
Jancik, F., Homolka, P., Cermak, B. & Lád, F. 2008. ‘‘Determination of indigestible neutral detergent fibre contents of grasses and its prediction from chemical composition’’. Czech Journal of Animal Science-UZPI (Czech Republic), 53 (3), pp. 128–135, ISSN: 1212-1819, 1805-9309.
Keady, T. W. J., Mayne, C. S. & Fitzpatrick, D. A. 2000. ‘‘Prediction of silage feeding value from the analysis of the herbage at ensiling and effects of nitrogen fertilizer, date of harvest and additive treatment on grass silage composition’’. The Journal of Agricultural Science, 134 (04), pp. 353–368, ISSN: 1469-5146.
Keles, G., O’Kiely, P., Lenehan, J. J. & Forristal, P. D. 2009. ‘‘Conservation Characteristics of Baled Grass Silages Differing in Duration of Wilting, Bale Density and Number of Layers of Plastic Stretch-Film’’. Irish Journal of Agricultural and Food Research, 48 (1), pp. 21–34, ISSN: 0791-6833.
Léder, I. 2009. ‘‘Cultivated Plants, Primarily as Food Sources’’. In: Fuleky, G. (ed.), Encyclopedia of Life Support Systems, vol. 1, Oxford ,UK: Eolss Publishers Company Limited, ISBN: 978-1-84826-550-9.
Liang, S., Yang, G. & Ma, Y. 2009. ‘‘Chemical Characteristics and Fatty Acid Profile of Foxtail Millet Bran Oil’’. Journal of the American Oil Chemists’ Society, 87 (1), pp. 63–67, ISSN: 0003-021X, 1558-9331, DOI: 10.1007/s11746-009-1475-3.
McDonald, P., Henderson, A. R. & Heron, S. J. E. 1991. The Biochemistry of Silage. 2nd ed., Kingston: Scholium Intl, 340 p., ISBN: 978-0-948617-22-5.
McEniry, J., O’Kiely, P., Clipson, N. J. W., Forristal, P. D. & Doyle, E. M. 2007. ‘‘The relative impacts of wilting, chopping, compaction and air infiltration on the conservation characteristics of ensiled grass’’. Grass and Forage Science, 62 (4), pp. 470–484, ISSN: 1365-2494, DOI: 10.1111/j.1365-2494.2007.00602.x.
Meineri, G. & Peiretti, P. G. 2010. ‘‘Determination of gross energy of silages’’. Italian Journal of Animal Science, 4 (2s), pp. 147–149, ISSN: 1828-051X, DOI: 10.4081/ijas.2005.2s.147.
Nielsen, D. C., Vigil, M. F. & Benjamin, J. G. 2006. ‘‘Forage Yield Response to Water Use for Dryland Corn, Millet, and Triticale in the Central Great Plains’’. Agronomy Journal, 98 (4), p. 992, ISSN: 1435-0645, DOI: 10.2134/agronj2005.0356.
Nussio, L. G. 2005. ‘‘Silage production from tropical forages’’. In: Park, R. S. & Stronge, M. D. (eds.), Proceedings of the XIV International Silage Conference, a satellite workshop of the XX International Grassland Congress, Belfast, Northern Ireland: Wageningen Academic Publishers, pp. 97–107, ISBN: 978-90-76998-75-6.
Peiretti, P. G., Daprà, F., Zunino, V. & Meineri, G. 2010. ‘‘The effect of harvest date on the chemical composition, gross energy, organic matter digestibility, nutritive value and amino acid content of white lupin (Lupinus albus L.)’’. Cuban Journal of Agricultural Science, 44 (2), pp. 169–173, ISSN: 2079-3480.
Peiretti, P. G., Gai, F. & Tassone, S. 2013. ‘‘Fatty acid profile and nutritive value of quinoa (Chenopodium quinoa Willd.) seeds and plants at different growth stages’’. Animal Feed Science and Technology, 183 (1), pp. 56–61, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2013.04.012.
Repetto, J. L., Cajarville, C., D’Alessandro, J., Curbelo, A., Soto, C. & Garín, D. 2005. ‘‘Effect of wilting and ensiling on ruminal degradability of temperate grass and legume mixtures’’. Animal Research, 54 (2), p. 8, ISSN: 1627-3583, 1627-3591, DOI: 10.1051/animres:2005007.
Robinson, P. H., Mathews, M. C. & Fadel, J. G. 1999. ‘‘Influence of storage time and temperature on in vitro digestion of neutral detergent fibre at 48 h, and comparison to 48 h in sacco neutral detergent fibre digestion’’. Animal Feed Science and Technology, 80 (3), pp. 257–266, ISSN: 0377-8401, DOI: 10.1016/S0377-8401(99)00062-0.
Rymer, C. 2008. ‘‘The effect of wilting and soaking Eupatorium adenophorum on its digestibility in vitro and voluntary intake by goats’’. Animal Feed Science and Technology, 141 (1–2), pp. 49–60, ISSN: 0377-8401, DOI: 10.1016/j.anifeedsci.2007.04.013.
Svirskis, A. 2009. ‘‘Prospects for non-traditional plant species cultivated for forage in Lithuania’’. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 37 (1), p. 215, ISSN: 0255-965X.
Torbatinejad, N., Galeshi, S. & Ghoorchi, T. 2009. ‘‘Evaluation by Chemical and in vitro Gas Production Techniques of Foxtail Millet Grown in Northern Iran’’. Journal of Animal and Veterinary Advances, 8 (12), pp. 2662–2667, ISSN: 1680-5593.
Received: June 11, 2014
Accepted: March 28, 2016
P.G. Peiretti, Institute of Sciences of Food Production, National Research Council, Largo Braccini 2, 10095 Grugliasco (TO), Italy. Email: piergiorgio.peiretti@ispa.cnr.it
 All the contents of this journal, except where otherwise noted, is licensed under a Creative Commons Attribution License
All the contents of this journal, except where otherwise noted, is licensed under a Creative Commons Attribution License













