My SciELO
Services on Demand
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Cuban Journal of Agricultural Science
On-line version ISSN 2079-3480
Cuban J. Agric. Sci. vol.50 no.3 Mayabeque Jul.-Sept. 2016
Cuban Journal of Agricultural Science, 50(3): 455-463, 2016, ISSN: 2079-3480
ORIGINAL ARTICLE
Evaluation of polyethylene glycol phosphate aqueous two-phase systems in purifying pepsin from residues of the slaughter house
Evaluación de los sistemas bifásicos acuosos polietilenglicol fosfato en la purificación de pepsina proveniente de residuos de matadero
Maryen Alberto Vázquez,I Beatriz Farruggia,II
IInstituto de Ciencia Animal, Apartado Postal 24, San José de las Lajas, Mayabeque, Cuba.
IILaboratorio de Bioseparación, Departamento de Físico-química, Universidad Nacional del Rosario.
ABSTRACT
In order to determine the most suitable aqueous two-phase system in pepsin separation, different purification systems formed by polyethylene-glycol (PEG) and potassium phosphate were evaluated. The study was conducted on enzyme homogenates of porcine gastric mucus (PH) and bovine abomasum homogenates (BH). Different molecular mass of polyethylene - glycol (1500 and 3000) and distribution of temperature (8 and 20 °C) were evaluated. The distribution of the pure enzyme was first determined, and then, the performance of the enzyme in the homogenates. The enzyme yield and the purification factor were determined for each system. The pepsin distribution in both homogenates showed similar performance to the pattern enzyme and distribution constants higher than the unit (PH 2.7 and BH 1.5) were obtained. The purification indicators for both preparations showed a purification factor higher than 3(HP 3.25 and BH 3.81) and yields higher than 60 % (PH 74.16 and BH 61. 54). The system formed by PEG 1500/phosphate at a distribution temperature of 20 ºC was the best variant. This method of separation and purification of pepsin is a simple and rapid alternative, also allows obtaining an adequate purity for further extractives steps of the enzyme.
RESUMEN
Para determinar el sistema bifásico acuoso más adecuado en la separación de pepsina, se evaluaron diferentes sistemas de purificación formados por poli etilenglicol (PEG) y fosfato de potasio. El estudio se realizó en homogenados enzimáticos de mucosa gástrica porcina (HP) y homogenados de abomaso bovino (HB). Se evaluaron diferentes masas moleculares de poli etilenglicol (1500 y 3000) y temperaturas de reparto (8 y 20 ºC). Se determinó primeramente el reparto de la enzima pura y luego, el comportamiento de la enzima presente en los homogenados. Se determinó el rendimiento de enzima y el factor de purificación para cada sistema. El reparto de la pepsina en ambos homogenados mostró comportamiento similar a la enzima patrón y se obtuvieron constantes de reparto superiores a la unidad (HP 2.7 y HB 1.5). Los indicadores de la purificación para ambos preparados mostraron un factor de purificación superior a 3 (HP 3.25 y HB 3.81) y rendimientos superiores a 60 % (HP 74,16 y HB 61, 54). El sistema formado por PEG 1500/fosfato a una temperatura de reparto de 20 ºC resultó la mejor variante. Este método de separación y purificación de pepsina es una alternativa simple y rápida, además permite obtener una pureza adecuada para posteriores pasos extractivos de la enzima.
INTRODUCTION
In the agricultural industry, the residues treatment and the search of new enzymes are aspects of great importance (Saval 2012). In Cuba, few proteins that are isolated from a bacterial culture or are purified from their natural source to supply the industry, which are mostly imported (Montes de Oca et al. 2013). The obtaining of enzyme preparations to replace commercial enzymes in different uses represents a significant cost savings for the country and, if the obtaining source of these is a waste, the benefit is double, because when used decreases its negative effect on the environment.
By the shown, the study of new isolation forms and enzymes purification from natural substrates and industrial waste is of interest. One of the enzymes most used in research laboratories are the proteases, such as pepsin, which initiates protein degradation in the stomach (Morillo et al. 2015). Pepsin is widely used in food and pharmaceutical industries and is a useful tool for in vitro digestibility simulations of many foods. However, their purification procedure, like that of any other enzyme, is a highly complex process.
At present there are dissimilar extraction and isolation techniques, but science increasingly moving towards the use of simple, rapid and inexpensive methods that provide a final product in fewer steps. Among these methods it is highlighted the purification in aqueous two- phase systems (ATPS), characterized by being simple and provide an environment of high stability to macromolecules. In addition, unlike other methods, these can be scaled without major complications. For these reasons, this research aims to study the aqueous two-phase systems in the separation and purification of pepsin from the slaughter house wastes.
MATERIALS AND METHODS
Obtaining enzymatic crudes. The gastric mucus of three stomachs per species (pigs and cattle) for obtaining the homogenates was taken. The animals that were extracted the organ intake the same diet and belonged to the same category (fattening).
Immediately after removing the stomach of the animal just slaughtered, the mucus was separated. It was washed with saline solution and specific regions were taken. In the pig, the mucus from the fundic area of the stomach was taken and in the cattle, the abomasum gastric mucus, up to collect a total weight of 100 g of mucus. The collected mucus was cut into small portions, fat was manually removed and mixed with 300 mL of potassium phosphate buffer 50 mM at pH 7.0 using a blade homogenizer, with short and repeated cycles of 1 min. each. The raw extract was filtered with glass fiber to remove cell remains, and then centrifuged at low temperature (5 °C) for twenty minutes at 15000 rpm. The supernatant was divided into small fractions and stored at -20 °C until their further use (Boeris et al. 2009).
Before use, the homogenate was thawed and the stored fat was removed at the top by using Pasteur pipettes.
Preparation of two phase systems. The two – phase systems (TPS) were formed with polyethylene glycol (PEG) of different molecular mass (1500 and 3000) and potassium phosphate (Pi), as showed in table 1. The concentration of these compounds was selected according to partitions reported by Boeris et al. (2009) and according to binodial diagrams proposed by Lei et al. (1990) and Tubío et al. (2007). An amount of 2 g of each system were prepared at pH 6.0 by direct weight of the components. The constituents were mixed and equilibrated for 30 min. at 20 °C. Then they were agitated by inversion and were let to rest for 24 h in separating funnel at constant temperature. Once reached the balance, the upper and lower phases were separated and six new smaller ATPS were reconstituted to later test the pepsin distribution.
Determination of distribution coefficient. To determine the coefficient of enzyme distribution, 250 μL of a solution pepsin concentration pattern 10 g/L were added to the preformed two-phase system, which contained 2 mL of each balanced phase.
To determine the effect of temperature, the coefficient of enzyme distribution in the different systems (PEG 1500 and PEG 3000) at 8 and 20 °C was evaluated. After mixing by inversion for a minute, it was let to rest 30 min. in bath with controlled temperature, prior to phase separation to allow the balance distribution was reached. There was not variation in the volume of each phase when adding the protein. Subsequently, it was removed with micropipette the maximum amount of both phases discarding the interface.
The coefficient of pepsin pattern distribution was calculated as the ratio of enzyme activity in the upper and lower phases respectively.
For the distribution of porcine (PH) and bovine (BH) homogenates it was proceeded to prepare the two-phase systems the same way as for the distribution of pure enzyme. The constituents were mixed and equilibrated for 30 min. at 20 °C, shake by inversion and then, let to rest for 24 h in separating funnels, at constant temperature of 25 °C.
To prevent the disappearance of ATPS by dilution, BH and PH masses corresponding at 15% of the total mass of the system were used. The volume ratio between the phases, once the balance was reached(R = 1), did not affect the distribution constant.
The distribution was carried out in rest for 2 h at 20 ºC. Separated the upper and lower phases, three samples of each phase per each two- phase system were taken and enzyme activity and total protein concentration was determined.
The distribution constants of total protein and enzyme, for each homogenate were determined by the ratio of protein concentration and enzyme activity between the phases, respectively.
Determination of proteins concentration. The protein concentration of homogenates, and their purified fractions by the aqueous two- phase system, was determined by absorbance readings at 280 nm. For calculating a pattern curve of serum bovine albumin with concentration range between 0.05 and 0.5 mg/mL was used. All analyzes were performed in triplicate. To correct interference by phases, target systems without protein were used.
In the distribution of the enzyme pattern (pepsin), the concentration was determined by calibration curves on the upper and lower phases of the two-phase systems under study. Pepsin concentration interval between 0. 05 and 0.5 mg/mL for each phase were used and targets without proteins to correct possible interference. All analyzes were performed in triplicate.
Determination of enzyme activity. The enzyme activity of crudes and their purified fractions was determined by the technique of milk coagulation, described by Tubío et al. (2007).
Increasing volumes (50, 75, 100, 150 and 200 uL) of commercial pepsin solution (0.1 g / L) were added to the tubes with 2 mL of milk, so that the aggregate volume did not exceed 10% of the total volume for measures of coagulation time (Ct) not be affected by dilution. To perform a calibration curve the Ct for each of the added volumes of pepsin were calculated and Ct was showed in a graph versus 1/mass (1/μg) of pepsin aggregates.
The concentration of pepsin average in g/L in the different samples was calculated by interpolation of the values of Ct in the calibration curve.
The inverse of the slope of the graph Ct vs. 1/μL allows to obtaining the coagulase activity –total activity in U/mL. An enzymatic unit is defined as the amount of enzyme that coagulates 2 mL of skim milk at 35 °C per minute. All measurements were performed in triplicate.
Determination of purification factor. It was determined by the ratio of enzymatic activity obtained in the separation process with respect to the initial enzymatic activity (Bravo et al. 2011).
Yield (y %) of total proteins and pepsin in superior phase of each system was calculated using the following equation (Bravo et al. 2011):
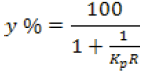
Where:
Kp is the constant of pepsin distribution
R is the volume ratio between the upper and lower phases, respectively
Statistical analysis. To study the effect of temperature and molar mass in the distribution constant of pepsin pattern an analysis of variance was carried out, according to simple classification model with factorial arrangement 2 x 2. The Duncan test (1955) was applied for comparing means.
The determinations of the enzymatic activity, protein concentration and distribution constants, were analyzed according to simple classification model. The mean values were compared by the Duncan test (1955) in necessary cases. The data were analyzed using computer statistical package InfoStat, version 2012 (Di Rienzo et al. 2012).
RESULTS AND DISCUSSION
Table 2 shows the effect of temperature and the molecular mass of PEG on the distribution constant of pepsin pattern.
The best pepsin distributions were found in systems with PEG 1500 and for a temperature of 20 °C. These results are in agreement with that of pointed by Spelzini et al. (2006, 2011), who reported that the systems better adapted to separate the pepsin are those with low molecular masses of PEG.
In this study a satisfactory separation of the enzyme towards the upper phase of the two systems was obtained, with values of coefficient distribution higher to the unit, which indicates a favorable interaction between the protein and PEG. This performance of pepsin in the two-phase system is due to, mainly, to its structure, because although most of the hydrophobic residues are inside the molecule, this has a high hydrophobic character in the surface exposed to the solvent, generating interactions with the system under study (Spelzini et al. 2005).
The PEG has a flexible molecule, which can acquire a compact structure that is stabilized by intramolecular hydrophobic unions. This compact structure has lower interaction with the solvent than the molecules fully extended, allowing interacts with the protein domain (Marini et al. 2011). The ability to interact with proteins is also mediated by their molecular mass and the effect of temperature (Bassani et al. 2010).
According to Tubío et al. (2007), the increase in the molecular mass of PEG induces the displacement the distribution equilibrium towards the phase where the polymer is in lower concentration, that is, towards the lower phase. The volume excluded by the PEG increases as its molecular mass increases, causing a transfer of pepsin to the salt- rich phase, with the consequent decrease of the distribution constant. This effect agrees with the results which are obtained in this study.
Arakawa and Timasheff (1985) studied the interaction between PEG and lacto globulin β, depending on the molecular mass of PEG. These authors observed that the preferential exclusion increases when the size of the PEG increases. This result is in favor of the steric exclusion as one of the factors which determines the interaction of PEG with proteins.
In the case of the effect of temperature on the distribution enzyme, it is observed that an increase in it induces a displacement of pepsin to the polymer-rich phase, informed by Spelzini et al. (2008) .This effect is manifested for most of proteins, where the temperature increase leads to the distribution coefficient increase.
Several authors stated (Imelio et al. 2008, Spelzini et al. 2008) that in the systems formed by PEG / Pi, the pepsin transfer from the salt- rich phase towards the PEG -rich phase is favored by increase of temperature, mainly to low molecular mass of PEG. Spelzini et al. (2011) studied the heat associated with the pepsin distribution and found that enthalpy changes are associated with the transfer of pepsin from the salt-rich phase to the PEG- rich phase.
When the transfer of protein from one phase to the other is carried out, a large enthalpy change suggests the breaking of many intermolecular unions (structured water in the protein domain and in the polymer). This effect may be due to the strong interaction PEG-protein that induces the displacement of water in the protein domain. It is also considered, that the structured water around the hydrophobic chain (ethylene) of PEG molecule release the ordered water (Tubío et al. 2007).
As it is shown in figure 1, high separation of contaminating proteins was achieved. The enzyme was distributed mainly towards the upper PEG- rich phase and most of their impurities towards the interface and the lower phase.
Figure 2 show that the values of enzymatic activity in the upper phase of each system have higher values to those of the lower phase and to the original preparations.
Spelzini et al. (2011) refers that PEG produces interaction not destabilizing in the secondary structure of protein. Therefore, to separate the enzymes in the upper PEG-rich phase is a benefit, in terms of stability for subsequent purification steps.
As figure 3 shows, there is greater enzyme distribution than the total proteins, suggesting high recovery of enzymatic activity in the upper phase of the system in relation to the rest of the proteins present in the homogenates.
Figure 4 shows that for both homogenates a purification factor exceeding three was obtained, which indicate that the target protein of a complex mixture was extracted. Although the porcine homogenate showed higher yield of enzymatic activity, for both homogenates best performance of enzyme was achieved than of protein, which shows the separation of pepsin of much of the contaminating proteins.
As for the purification factor, values higher than three were reached. Differences between homogenates are associated with higher concentration of contaminating proteins in the porcine homogenate.
Previous studies of pepsin purification from their natural sources with the use of PEG/Pi two-phase systems described by Rito (2004) and Boeris et al. (2009) refers yield values and purification factors similar to this study.
It is known that the yield of a pure enzyme is very high (between 80 and 95 %). However, when the method is used in the purification of the same enzyme in a complex mixture, such as the animal tissue homogenate or plant tissue homogenate, the yield is lower. This is due to the presence of other macromolecules such as nucleic acids and proteins, which can interact with the enzyme and cause its precipitation on the interface (Bassani et al. 2010).
Pepsin is purified from their natural sources by different methods, which it combines the fractional precipitation with addition of salts (Páez de Leon et al. 1995) and polyelectrolytes of opposite load (Boeris et al. 2009), the anionic exchange chromatography (Rocha et al. 2010), gel filtration (Brier et al. 2007), among others. Most of these methods allow obtaining only a very few amount of the enzyme. Furthermore, in many cases the process is complex and expensive.
Pepsin purification with the use of aqueous two-phase systems provides a simple, fast and inexpensive alternative besides of having the possibility of being widely applied (Du et al. 2007). The use of this method to obtain preparations that do not require high degrees of purity is of choice, as the characteristics of the system provide a suitable environment that preserves the biological activity of materials and provides high stability to macromolecules (Kayitmazer et al. 2007).
It is concluded that the two-phase system composed by PEG1500/potassium phosphate at a distribution temperature of 20 °C was the variant that achieved the best pepsin distribution towards the upper PEG rich- phase. This system allow eliminates most of the contaminating proteins in a stage, obtain a purification factor higher three for each extract and ensure an acceptable purity for subsequent extractive steps of the enzyme.
REFERENCES
Arakawa, T. & Timasheff, S. N. 1985. “Mechanism of polyethylene glycol interaction with proteins”. Biochemistry, 24(24): 6756–6762, ISSN: 0006-2960, 1520-4995, DOI: 10.1021/bi00345a005.
Bassani, G., Fuciños, P., Picó, G. & Farruggia, B. 2010. “Candida rugosa lipase Lip1–polyethyleneglycol interaction and the relation with its partition in aqueous two-phase systems”. Colloids and Surfaces B: Biointerfaces, 75(2): 532–537, ISSN: 0927-7765, DOI: 10.1016/j.colsurfb.2009.09.031.
Boeris, V., Spelzini, D., Farruggia, B. & Picó, G. 2009. “Aqueous two-phase extraction and polyelectrolyte precipitation combination: A simple and economically technologies for pepsin isolation from bovine abomasum homogenate”. Process Biochemistry, 44(11): 1260–1264, ISSN: 13595113, DOI: 10.1016/j.procbio.2009.07.001.
Bravo, M. K. E., Muñoz, D. K., Calderón, G. J. & Osorio, D. E. J. 2011. “Desarrollo de un método para la extracción de polifenol oxidasa de uchuva (Physalis peruviana L.) y aislamiento por sistemas bifásicos acuososo”. Vitae, 18(2): 124–132, ISSN: 0121-4004, 2145-2660.
Brier, S., Maria, G., Carginale, V., Capasso, A., Wu, Y., Taylor, R. M., Borotto, N. B., Capasso, C. & Engen, J. R. 2007. “Purification and characterization of pepsins A1 and A2 from the Antarctic rock cod Trematomus bernacchii”. FEBS Journal, 274(23): 6152–6166, ISSN: 1742464X, DOI: 10.1111/j.1742-4658.2007.06136.x.
Di Rienzo, J. A., Casanoves, F., Balzarini, M. G., González, L., Tablada, M. & Robledo, C. W. 2012. InfoStat. version 2012, [Windows], Universidad Nacional de Córdoba, Argentina: Grupo InfoStat, Available: <http://www.infostat.com.ar/> .
Du, Z., Yu, Y. L. & Wang, J. H. 2007. “Extraction of Proteins from Biological Fluids by Use of an Ionic Liquid/Aqueous Two-Phase System”. Chemistry - A European Journal, 13(7): 2130–2137, ISSN: 09476539, 15213765, DOI: 10.1002/chem.200601234.
Duncan, D. B. 1955. “Multiple Range and Multiple F Tests”. Biometrics, 11(1): 1–42, ISSN: 0006-341X, DOI: 10.2307/3001478.
Imelio, N., Marini, A., Spelzini, D., Picó, G. & Farruggia, B. 2008. “Pepsin extraction from bovine stomach using aqueous two-phase systems: Molecular mechanism and influence of homogenate mass and phase volume ratio”. Journal of Chromatography B, 873(2): 133–138, ISSN: 15700232, DOI: 10.1016/j.jchromb.2008.07.006.
Kayitmazer, A. B., Strand, S. P., Tribet, C., Jaeger, W. & Dubin, P. L. 2007. “Effect of Polyelectrolyte Structure on Protein−Polyelectrolyte Coacervates: Coacervates of Bovine Serum Albumin with Poly (diallyl dimethyl ammonium chloride) versus Chitosan”. Biomacromolecules, 8(11): 3568–3577, ISSN: 1525-7797, 1526-4602, DOI: 10.1021/bm700645t.
Lei, X., Diamond, A. D. & Hsu, J. T. 1990. “Equilibrium phase behavior of the poly(ethylene glycol)/potassium phosphate/water two-phase system at 4.degree.C”. Journal of Chemical & Engineering Data, 35(4): 420–423, ISSN: 0021-9568, 1520-5134, DOI: 10.1021/je00062a014.
Marini, A., Imelio, N., Picó, G., Romanini, D. & Farruggia, B. 2011. “Isolation of a Aspergillus niger lipase from a solid culture medium with aqueous two-phase systems”. Journal of Chromatography B, 879(22): 2135–2141, ISSN: 15700232, DOI: 10.1016/j.jchromb.2011.05.042.
Montes de Oca, O. A., Bermúdez, R. C. & Serrat, D. M. 2013. “Evaluación del boniato tetuanado como sustrato para la obtención de proteína microbiana”. Tecnología Química, 33(3): 340–352, ISSN: 2224-6185.
Morillo, O. T., García, L. P. J., Guerrero, C. B. R., Torres, V. Y. & Castañeda, R. R. 2015. “Evaluación de la producción experimental de enzimas coagulantes de leche utilizando cepas de Rhizomucor spp.”. Revista Colombiana de Biotecnología, 17(1): 54–60, ISSN: 1909-8758, 0123-3475, DOI: 10.15446/rev.colomb.biote.v17n1.50701.
Páez de León, L., Pinzón, G. & Otaiza, V. E. 1995. “Purification and assay of chicken pepsin”. Acta Cientifica Venezolana, 46(4): 237–241, ISSN: 0001-5504, PMID: 9334437.
Rito, P. M. 2004. “Practical application of aqueous two-phase partition to process development for the recovery of biological products”. Journal of Chromatography B, 807(1): 3–11, ISSN: 1570-0232, DOI: 10.1016/j.jchromb.2004.01.008.
Rocha, G. F., Fernández, G. & Parisi, M. G. 2010. “Estudios de Caracterización Cinética y Fisicoquímica de una Proteinasa Aspártica Aislada de Frutos Maduros de Salpichroa origanifolia”. Información Tecnológica, 21(2): 21–28, ISSN: 0718-0764, DOI: 10.4067/S0718-07642010000200004.
Saval, S. 2012. “Aprovechamiento de Residuos Agroindustriales: Pasado, Presente y Futuro”. BioTecnología, 16(2): 14–46.
Spelzini, D., Farruggia, B., Guerra, N. P., Rua, M. L. & Pastrana, L. 2011. “Production of aspartic peptidases by Aspergillus spp. using tuna cooked wastewater as nitrogen source and further extraction using aqueous two phase system”. African Journal of Biotechnology, 10(68): 15287–15294, ISSN: 1684-5315, DOI: 10.4314/ajb.v10i68.
Spelzini, D., Farruggia, B. & Picó, G. 2005. “Features of the acid protease partition in aqueous two-phase systems of polyethylene glycol–phosphate: Chymosin and pepsin”. Journal of Chromatography B, 821(1): 60–66, ISSN: 15700232, DOI: 10.1016/j.jchromb.2005.04.007.
Spelzini, D., Peleteiro, J., Picó, G. & Farruggia, B. 2008. “Polyethyleneglycol–pepsin interaction and its relationship with protein partitioning in aqueous two-phase systems”. Colloids and Surfaces B: Biointerfaces, 67(2): 151–156, ISSN: 09277765, DOI: 10.1016/j.colsurfb.2008.06.013.
Spelzini, D., Picó, G. & Farruggia, B. 2006. “Dependence of chymosin and pepsin partition coefficient with phase volume and polymer pausidispersity in polyethyleneglycol–phosphate aqueous two-phase system”. Colloids and Surfaces B: Biointerfaces, 51(1): 80–85, ISSN: 09277765, DOI: 10.1016/j.colsurfb.2006.03.023.
Tubío, G., Nerli, B. & Picó, G. 2007. “Partitioning features of bovine trypsin and α-chymotrypsin in polyethyleneglycol-sodium citrate aqueous two-phase systems”. Journal of Chromatography B, 852(1–2): 244–249, ISSN: 15700232, DOI: 10.1016/j.jchromb.2007.01.025.
Received: 14/09/2015
Accepted: 11/07/2016
Maryen Alberto Vázquez, Instituto de Ciencia Animal, Apartado Postal 24, San José de las Lajas, Mayabeque, Cuba. Email: mvazquez@ica.co.cu














