According to Potter and Hotchkiss (1995), the definition of "quality and safety" of a food is based on knowledge of the nutritional and hygienic-toxicological processes on which its production is based. That is why, the physical- chemical evaluation of food products, according to Doorenbos et al. (2004), is one of the most important aspects in the nutritional assessment. This will allow knowing the possible effects on the gastrointestinal and metabolic functions of the animal organism, which will contribute to formulate more balanced and adequate diets to obtain better productive efficiency.
In Cuba, products, by-products and waste products are studied that, due to their chemical and nutritional properties, can constitute a nutritional complement and provide nutrients for the animals' diet. Among these is distillery vinasse, waste of alcohol production, which stands out for its organic acids, yeasts, vitamins and minerals (Mc-Pherson et al. 2002). These components can increase resistance to stress and infectious diseases, by functioning as substances stimulating the innate immune response.
In response to its use in pigs and ducks, decreases in feeding costs and more efficient production results are reported (Lewicki 2001 and Stemme et al. 2005). It also refers to a stimulating effect on intake and animal performance, in response to the high content of B vitamins in the vinasse (Gohl 1991).
Sarria and Serrano (2008) describe the variability of the vinasse composition, taking into account the different points of the productive chain. However, they are referred to as animal food. However, the existing literature is not comprehensive in terms of its chemical-physical composition.
Therefore, it is necessary to deepen in the study of the chemical-physical and microbiological composition of the concentrated distillery vinasse, produced in the International Havana Club Rum factory, located in San José de las Lajas, Mayabeque, for its use as additive in animal feeding.
Materials and Methods
Biological material. Samples of concentrated vinasse were collected from the International Havana Club Rum factory, located in San José de las Lajas. The samples (table 1) were taken in four storage tanks, vertical factory silo (P-1), tank truck (P-2), horizontal tank from the Instituto de Ciencia Animal (P-3) and laboratory tank (P-4).
The sampling was randomly conducted in the outlets faucets and every 10 min. for P-1 and P-2. In the case of P-3 and P-4, the content was removed for 15 min. and the surface, the center and the bottom were sampled. The samples were taken in quintuple at each moment, stored in 250 mL sterile plastic bottles and kept at room temperature until further analysis.
Physical-chemical analysis of concentrated vinasse. The composition and fractionation of the chemical compounds was carried out in samples prepared for this purpose. The indicators of dry matter (DM), crude protein (CP) and ash were determined, according to AOAC (2016). Neutral detergent fiber (NDF) and acid detergent fiber (ADF) were determined according to van Soest (1994) and the true protein (TP) according to Bernstein et al. (1977).
Determination of pH. Digital potentiometer with glass electrode, buffer solutions of pH 4 and 7, and an electromagnetic stirrer were used.
Measurement of Brix degrees (ºBx). Three drops of each sample were taken and placed in the hand-held refractometer, with a scale of 0 to 80 %.
Table 1 Sampling for the characterization of the concentrated vinasse.
*Sample used in the stability study
Determination of minerals (macro and micro). The identification and quantification of P, Ca, Na, Fe, As and Pb was performed by atomic emission with inductive plasma, K by atomic absorption and S by turbidimetry at 10 % and glycerin, according to the techniques described in the AOAC (2016).
Determination of amino acids. It was carried out in two samples of concentrated vinasse (P-1 and P-4) and in the soybean cake (as reference standard). The analysis was performed by high pressure liquid chromatography (HPLC) according to the Krishnamurti et al. (1984) technique. The hydrolysis of the sample was carried out with 6N HCl at 110 °C temperature (Knecht and Chang 1986).
Determination of direct reducing carbohydrates. The technique described by Dubois et al. (1956) and Mancilla (2006) was used and the absorbance was read on an UV-Visible spectrophotometer at 490 nm.
Microbiological characterization. Two samples were used (P-1 and P-4), of which 1 mL of each was used and 9 mL of the dilution medium (1:10). It was shaked for one minute and then three samples of each dilution were taken for culturing under anaerobiosis or aerobiosis conditions, taking into account that there are groups of facultative microorganisms that can live in the presence or absence of oxygen.
For counting total viable, it was cultured on silver agar and for Escherichia coli on MacConkey agar, both under aerobiosis conditions at 36 °C. For the yeasts and fungi, the malt extract was used under aerobic conditions at 25 °C. For Lactobacillus spp. Agar M.RS was used under anaerobiosis conditions at 36 °C and the methodology described by the United States Department of Agriculture (USDA-FSIS 2008) was used.
Stability indicators. The sample P-4 was used, in which the pH values and specific weight (weight / volume) were analyzed. For the specific weight reading, a digital scale with an accuracy of ± 1 g and a graduated cylinder was used.
Data registration and statistical analysis. The data record was monthly performed for five months for physical- chemical studies. For stability, data was recorded during twelve months of storage.
For the analysis of the results, the Infostat computer system was applied, according to Di Rienzo et al. (2008). In the physical-chemical characterization, the variance analysis method was used, according to one- way model, and Duncan's test for P <0.05 was applied. In the case of the stability study, a descriptive analysis was carried out.
Results and Discussion
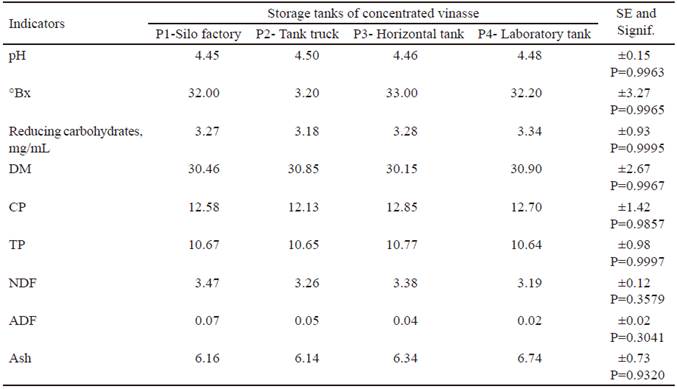
The results (table 2) show that there are no differences in the composition of the vinasse between the different storage tanks. They corroborate that the concentrated vinasse from the International Havana Club Rum factory has a low pH value, characteristic of this type of waste. These data coincide with those reported by Christofoletti et al. (2013), Moraes et al. (2014), Vázquez et al. (2014) and Camacho et al. (2016) and show that its storage is possible for a prolonged period and its use in food preservation.
Brix degrees in the samples were between 32 and 33 %, higher values than those reported in the literature (Aristizábal 2015). These values are related to the concentration of total soluble solids in the vinasse. That is to say, with the sugars coming from the walls of the yeasts and with the salts in the ashes, unlike other vinasses in which the yeast is collected for the protein production.
The vinasse showed low content of direct reducing carbohydrates, which could be related to the presence of glucans and mannan from the yeast used in the alcohol fermentation process. Similar results were described by Tungland and Meyer (2006), who suggest direct evaluations in animals, since vinasse contains oligosaccharides resistant to digestion, which reach the caecum and provoke a positive physiological response.
These compounds could have a positive effect on the immune system of animals, in addition to preventing the colonization of pathogenic bacteria in the gastrointestinal tract. According to Urias and López (2006), the fermentation of these in the caecum generates short chain fatty acids (SCFA), which are attributed positive health effects, such as increased absorption of inorganic nutrients and decreased glucose, triglycerides and blood cholesterol (Gibson and Roberfroid 2008).
The chemical indicators show that the DM values are higher than those reported in the vinasses from other distilleries in Cuba. Bermúdez et al. (2000) refer ranges of DM between 5 and 8 %, which is why we call concentrated vinasse to that obtained in the International Havana Club Rum factory.
Similar results were found by Sarria and Serrano (2008), who describe the concentration of these residuals in Valle del Cauca (15.4 to 47.6 % of DM). These authors report that the concentration process allows increasing the content of nutrients that the vinasse would contribute to the animals diets. They also emphasize that this characteristic is one of the most important for its management, since it reduces transportation costs and facilitates its conservation and storage.
Refering to the values for crude and true protein (table 2), they are similar to those obtained by Scull et al. (2012) when studying the vinasse coming from this factory during the process of technology standardization. However, they are superior to those reported in Cuba until now and are related to the content of yeast in this residual.
García et al. (1991) point out that this waste allows making important contributions to the diet of adult animals, if it is used as dehydrated meal at 90 %.
The ash content in the samples has values between 6.14 and 6.74 %, which represents low variability. These values are directly related to the degree of concentration of vinasse. According to Lugo (2017), the ash content can be 6 % in vinasse, with 22 % of DM and 16.4 % in vinasse with 47.1 % of DM.
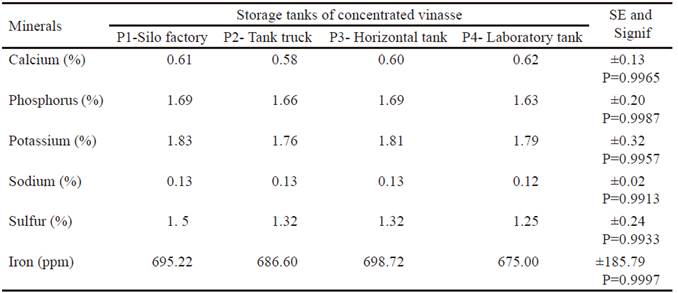
Another important aspect is the mineral content in the vinasse (table 3). This showed low values, except for K, Fe and P. The P could be available almost entirely by coming from the yeast used in the fermentation process. Conversely, Berón (2005) found that the mineral content in the vinasses studied in Colombia showed lower results, in terms of phosphorus content, but higher in relation to potassium, being this, one of the biggest problems for the use of high volumes in animal feeding. However, Zuñiga and Gandini (2013) describe the vinasse as a residual that has low or adequate mineral content, which does not lead to physiological disorders in the animals when performing low intake. Ponce de León (2008) emphasizes in the contribution of minerals of great importance in animal feeding for the formation of organs, tissues, body fluids or as controllers of metabolic, enzymatic and hormonal reactions.
The amino acid composition of the vinasse under study determines that it is a low quality protein source (table 4). The lowest values corresponded to the sulfurized aa: methionine (0-0.08) and cysteine (0.03-0.07), which coincides with those described by Gallo et al. (1986). However, they mention that the rest of the aminoacid were in relatively high concentrations, unlike those recorded in this study.
According to the Fundación Española para el Desarrollo de la Nutrición Animal (FEDNA 2017), the aa that are contributed by beet vinasse has real low digestibility in birds and pigs. It is inferred that they must have the same performance in the vinasses obtained from the cane molasses. Therefore, this disadvantage, join to the low level of inclusion in the diet, allows concluding that the aminoacid included in the diet do not influence on the results.
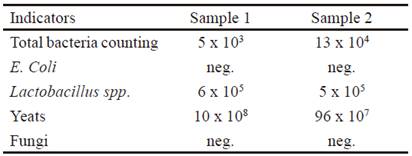
The microbiological results are shown in table 5. The safety of the product should be highlighted, as the presence of Escherichia coli and fungi are not recorded in the samples. De Blas et al. (2010) commented that the microbiological evaluation of the products or foods to be used in animals is of great importance, since it allows confirming the sanitary quality of them.
The lactobacilli in the vinasse are found in low concentrations (5 to 6 x 105) and, on the contrary, the yeasts are in high concentrations (10 x 108).
Regarding the use of yeasts in animal production, it can be mention that they offer great advantages. These are used as micro-ingredients due to their prophylactic action (Anon 2007) and among the main advantages it is highlighted that they are sources of nucleotides, a natural source of the B complex, including inositol, which is considered a growth promoter. It is also mentioned that they improve the quality of the ration and the intestinal integrity due to the presence of mannan oligosaccharides (MOS) and have a high mycotoxin absorption capacity.
Yeasts are composed between 20 and 40 % of carbohydrates in the cell wall represented mostly by glucans and mannan, which have an impact on the immune system and the ability to prevent the colonization of pathogenic bacteria in the gastrointestinal tract (Gibson and Roberfroid 2008).
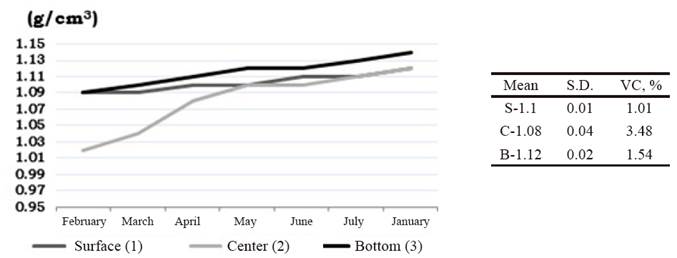
The indicators under study to determine the stability of the vinasse (pH and specific weight) did not show variation, being an aspect of great importance, since the low stability of the liquid compounds that are used in animal feeding constitutes a disadvantage when using alternative sources (Valdivié et al. 2010).
For the pH, the values fluctuated between 4.2 and 5.21 (figure 1), which is a characteristic feature of the distilleries vinasse (Saura 2008). This stability could be related to the presence of short-chain acids, which according to Leeson (2006) are considered food preservatives.
According to Gómez (2009), pH control is an important aspect in the elaboration of food products, as an indicator of hygienic conditions and for the control of transformation processes. It is also comments that pH, joins to temperature and humidity is important for the preservation of food and for the inhibition of the multiplication of pathogens. This author reports that the fermented products have better stability, due to the formation of final products in the oxidation that takes place in the fermentation process, among which are quinones and acetaldehydes (Gómez 2009).
The study of the specific weight was related to the variation of the DM due to the possible evaporation of the liquid content of the vinasse, taking into account the high temperatures that characterize Cuba (figure 2).
It should be highlighted that the composition of the vinasses is similar regardless of the raw material. However, the variability in the values will always be related to the source of sugars used to produce alcohol or aguardiente (juice or molasses of sugarcane, beet, agave, corn or barley). To this aspect is added the efficiency of the industry and the collection or not of the yeast used in the fermentation process.
In general, the results described allow classifying the waste as concentrated vinasse, taking into account the DM values. It also highlights the safety of vinasse and its stability over time. Likewise, the physical-chemical charac- terization of concentrated vinasse allows its evaluation as a candidate for additive, since it contains compounds and microorganisms that could cause positive effects on health and animal performance.











 text in
text in