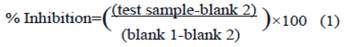Soybean meal in its untreated form contains a number of anti-nutritional factors (ANF) that have the potential of seriously diminishing its nutritional value to the point of decreasing animal health and performance (Gemede and Ratta 2014 and Rocha et al. 2014). Treatment of soybeans to eliminate these anti-nutritional factors is thus necessary especially in the case of monogastric diets. The deleterious components in soybean meal include several protease inhibitors generally called trypsin inhibitors (Rada et al. 2017), hemaglutinins or lectins (Udeogu and Awuchi 2016), goitrogens (Dourado et al. 2011 and Bajaj 2016), saponins (Omizu 2011 and Chaturvedi, 2012) and urease (Real-Guerra et al. 2013 and Khan et al. 2013). The presence of these factors in relatively large amounts places a restriction on the use of soybeans and their products in poultry diets. These anti-nutritional factors give reason why processing techniques are applied to soybeans and the most important of all these ANF is trypsin inhibitors.
It has been reported that trypsin inhibitors interfere with the digestion of proteins in the intestine of young chicks (Goebel and Stein 2011) and raw soybean meal causes enlargement of the pancreas of growing chicks because of the presence of deleterious components in large amounts (Beuković et al. 2012 and Dei 2011 and Foley et al. 2013). Effects of raw soybean meal on growth inhibition of chicks could not be overcome by supplementing the diet with methionine, lysine, etcetera (Loeffler et al. 2013). Hence, the need for suitable process and process conditions for adequate denaturing of trypsin inhibitor while also conserve the nutritional quality of soybean meal.
Nigeria, the leading producer of soybean in Sub - Saharan Africa (Mubichi 2017), has an estimated installed annual soybean crushing capacity of 600,000 MT to obtain oil for industrial use and soybean meal (USDA 2011). The soybean meal (SBM) utilized in Nigeria for poultry feed is a by-product of roasting operation and oil extraction either by screw press or hydraulic press (Moses 2014). It is desirable to produce soybean meal for poultry feed by other processes, examine the anti-nutritional content of the soybean meal produced and make comparison of the quality of soybean meal from these processes. Poultry feed meal with very low trypsin inhibitor content will certainly be safer for poultry in Nigeria.
The objectives of this study were to produce soybean meal using three heat treatment processes, namely: roasting, autoclaving and steaming, followed by solvent extraction of oil; carry out trypsin inhibitor analysis, proximate analysis and quality tests of the soybean meal; and make comparisons of the results from the processes and identify the best process and process conditions.
Materials and Methods
Preparation of Soybean. Foreign materials were separated from soya beans using double sieve method. The cleaned beans were then placed in the oven at 38 ºC for 24 h. After this step, the beans were crushed into large fragments, de-hulled, placed in oven at 55 ºC for 20 min and grinded into powder using hammer milling machine (Hanssen 2003).
Heat treatment of samples. Soybean was subjected to three heat treatments at varied conditions. The samples were properly labelled and studies were carried out in triplicates.
Steaming. For each run, 50 g of soybean powder was spread on the screen of a steamer. The soybean was heated by direct conduction for the period of 10, 12, 15 and 18 minutes using steam at a temperature of 105 ºC. The cake was cooled and dried (Gilbert et al. 2000).
Autoclaving. In this study, four runs of autoclaving were done for a period of 20, 30, 40 and 50 minutes, respectively. For each of the runs, 50 g of soybean powder was wrapped in hard polythene and was cooked in an autoclave operated at 121 ºC and 15 psi. The autoclaved samples were kept in cool and dry condition (Herrman 2001).
Roasting. In this study, 50g of soybean powder was spread on a thin layer of tray and placed in an oven. Four runs of roasting was done and the oven was operated at 120 ºC for 5 minutes, 130 ºC for 7 minutes, 135 ºC for 8 minutes and 140 ºC for 9 minutes, respectively (Gill 2003).
Defatting of soya bean meal. Each sample of the steamed, autoclaved and roasted soybean powder was defatted using Soxhlet extractor. N-hexane was used as the solvent and was gently heated for 3 hr. At the end of each run, the defatted cake was removed, de-solventised by oven drying at 37 ºC. The extracted oil along with the hexane was boiled in order to remove the hexane and the percentage of extracted oil was calculated (Herrman 2001).
Analytical methods. The effects of heat treatments on nutrient availability, quality of protein and trypsin activity on the soybean meals were analyzed.
Trypsin inhibitor analysis. 2g each of the treated soybean powder was homogenized with 12 mL phosphate buffer. The mixtures were centrifuged and the aliquots were taken to study their inhibitory activity. For each of the inhibitory activity studies, 0.1mL of soybean aliquot was mixed with 0.1 mL of 1 mg/mL trypsin solution. This was allowed to stand at 0 ºC for 10 min before 0.3 mL of 2 % casein was added. The mixture was incubated in the water bath for 20 min at 37 ºC. The reaction was stopped and undigested casein was precipitated by the addition of 0.5 mL of 2.9 M perchloride acid (Iwamoto and Abiko 1970). The undigested casein, larger inhibitor fragments and enzyme protein were removed by centrifuging for 5 min. In addition to the test samples, two control experiments were run simultaneously. Control experiment 1 was done without the addition of any inhibitor and control experiment 2 was done with casein only. The amounts of soluble inhibitors in the supernatant acid were estimated using the method of Lowry et al. (1951). The absorbances of the clear supernatant were measured using a UV spectrophotometer (Camspec and Model M201) and the percentage inhibition was calculated using the following equation:
Where, blank 1 is control without the addition of any inhibitor and blank 2 is control with casein only. Proximate analysis. The moisture content of the treated soybean meal was determined according to AOAC (2010) official method and expressed as a percentage of total weight of original sample. The ash content of the soybean meal was determined according to AOAC (2010) official method. The crude protein of the soybeans was estimated using the Kjeldahl method, the nitrogen content of the digested sample was determined using Kjeldahl automated nitrogen analyzer (Foss Model 2300) and the percentage crude protein was estimated from the nitrogen content as previously reported by van Eys et al. (2004). The crude fiber of the soybean meal was determined using method described by AOAC (2010) official method. The ether extract was determined by following procedure described by van Eys et al. (2004). The urease index level was determined by method described by AOCS (1993). The KOH protein solubility was determined using method previously reported (Araba and Dale 1990). To determine the protein dispersability index (PDI), the nitrogen content of soybean meal sample was analyzed using the Kjeldahl method and expressed as a percentage of the original soybean protein content of the sample that was dispersed in water (ASA 2005).
Statistical analysis. Results obtained from the laboratory were subjected to statistical analysis using IBM SPSS Statistics 23. One-way ANOVA test was run on the values and means were separated using Duncan multiple range test (DMRT). The samples were done in triplicate
Results and Discussion
The table 1 revealed that at p<0.05, there is a significant difference in the mean values of trypsin. The results generally showed the action of heat to denature trypsin inhibitor. The absorbance is a measure of activity. At p<0.05, it was observed that the highest inhibitor activity of 3.54 mg/g occurred in beans roasted at 120 ºC for 5 minutes. This could be as a result of low roasting temperature and short time of roasting. Furthermore, at significant level set at 5%, soybean meal processed with steam at 105ºC for 18 minutes gave the best result with inhibitor activity of 1.20 mg/g. The untreated beans gave a very high trypsin activity of 6.01 mg/g. Roasting at low temperatures (120 ºC - 135 ºC) for short period (5min - 8min) is inadequate to sufficiently denature trypsin inhibitor in soybean to obtain SBM with trypsin inhibitor between 1.75 - 2.50 mg/g which is taken to be safe (NOPA, 2011). However, for the preservation of the nutritional contents of SBM, especially, the protein content, prolonged heating and high temperature of heating is not desirable because excessive heat denatures protein.
The proximate analysis results are presented in table 2. The data presented in Table 2 was analyzed statistically with ANOVA and mean was separated using DMRT. Significance level was set at 5 %. The result revealed that at p<0.05, there was a significant difference in the mean values of moisture content, ash content, crude fiber, crude protein and ether extract.
The result of the statistical analysis showed that the moisture content of soybeans meal for all the processes yielded good results which could enhance long storage without growth of mould. The result showed that steam treatment had significant increasing effect on the moisture content of SBM which could be as a result of the introduction of steam from water. Moisture content which is an index of water activity is also used as a measure of stability (Alagbaoso et al. 2015) and susceptibility to microbial contamination (Rawat 2015), indicating that all the heat treated samples can be kept for an extended period without spoilage as their moisture content were significantly low. The highest moisture content of 4 % was recorded for the steam process which is considered low and good enough for long shelf life. The lowest moisture content was recorded for the roasting process with 1 % moisture content which showed that very small amount of moisture was present.
Table 1 Trypsin inhibitor activity

abcdefghValues having different superscripts are significantly different (p<0.05). The data presented in the table are mean of the triplicate determination ± standard deviation
Table 2 Proximate analysis

abcdefValues in the same column having different superscripts are significantly different (p<0.05). The data presented in the table are mean of the triplicate determination ± standard deviation
Also, at p<0.05, it was observed that all the processes gave good results for the ash content. The highest value of 5.5 % was obtained for autoclave process and the lowest of 3.5 % for the steam process. The value of 5.5 % for the autoclave process is considered non-hazardous for poultry feed. Ash content is an indicator of mineral content and mineral type in any sample (Enwereuzoh et al. 2015). The results showed that autoclaved samples had significantly higher ash content (p<0.05) indicating that it may have more mineral content than the other two heat treatment method although the treated samples all had fair ash content.
The crude fiber of the soybeans meal for all the processes showed consistency. The highest value of 4.4 % was obtained for the steam process and autoclave process and the lowest value of 3.8 % was obtained for the roasting process. Fibre has been shown to have some physiological effect in the gastrointestinal tract and low fibre in feed is undesirable as it may result in constipation (Zhang et al. 2013). The steam treatment result showed a significant increase in the crude fibre content of soybean meal (p≥0.05). The crude fibre in the roasted samples was similar to that of the raw sample.
The crude protein analysis showed that all the processes were capable of giving the good yield required for poultry feed production. Raw beans (without application of heat) gave 44.60 % crude protein content. The roasting process at 120 ºC for 5 min gave 40.58 % which could be as a result of low heat applied in the roaster. The lowest value of 39.38 % was obtained for the steam process operated at 105 ºC for 18 minutes which is acceptable and considered normal for poultry feed production.
The ether extract analysis showed that more oil cells were broken in some processes than others and consequently more oil was released in such processes. The highest percentage oil extracted (22.8 %) was obtained from the steam process and this is because more oil cells were broken by this process. Surprisingly, a greater variation was noticed in the autoclave process and the lowest percentage oil extracted (18.6 %) was for the autoclave process.
In comparison to NOPA specifications (NOPA 2011) which recommends maximum moisture content of 12 %; minimum protein content of 44 % and 47.5-49% for solvent extracted and dehulled SBM, respectively; minimum fat content of 0.5 %; maximum crude fiber of 7 % and 3.3-3.5 % for solvent extracted SBM and dehulled SBM, respectively; and ash content of less than 7.5 %, the result of the proximate analysis for all SBM produced by processes described in this study can be considered good. The slight difference between the protein content of the SBM from this study and the NOPA recommendation may be as a result of the heat treatment given to the SBM during solvent extraction of the oil. The slight difference from the NOPA protein recommendation may be considered a necessary trade off to obtain SBM with adequately low trypsin inhibitor.
The statistical analysis results of the quality tests are presented in table 3. ANOVA test was run on the mean and DMRT was used to separate the means and significance level was set at p<0.05. The result revealed that p<0.05, there was significant different in the values of urease activity index, protein solubility in KOH, and protein dispersability index (PDI). The result showed that at p<0.05, the urease activity index was at the highest level of 2.07 for beans processed by roasting at 120 ºC for 5 minutes.
Table 3 Quality test

abcdefValues in the same column having different superscripts are significantly different (p<0.05). The data presented in the table are mean of the triplicate determination ± standard deviation.
The lowest urease value of 0.11 was obtained for the beans processed by steam at 105 ºC for 18 minutes. In comparison with the standard prescription of 0.01 - 0.35 pH unit rise (NOPA, 2011), it was observed that the steam process gave the best results followed by the autoclave process. The roasting process did not produce good results. The raw beans gave the highest value of urease activity index of 2.11 at p<0.05.
The adequacy of processing is estimated by the protein solubility in KOH solution (Căpriţă, et al. 2010). SBM with KOH solubilities between 70 and 85 % are considered adequately heat processed (van Eys et al. 2004). The KOH protein solubility test when evaluated statistically at p<0.05 using one way Anova showed that the autoclave processed soya beans meal at 40 minutes and 50 minutes processing time had adequate heat treatment with solubilities of 72.8 % and 73.1 %, respectively. The results obtained for the roasting process showed under processing which could be as a result of inefficient and insufficient heating from the roaster. The steam process for 15 minutes and 18 minutes gave high protein solubility of 70.0 % and 72.1 %, respectively. The raw beans gave a solubility of 49.1 %.
The protein dispersability index (PDI) test predicts the quality of soybean meal more accurately (Dozier and Hess, 2011). The results were generally encouraging with the exception of the roasting process which showed complete deviation from PDI values between 15 and 30 % recommendation for soybean meals (van Eys et al. 2004). The beans processed by steam at 105 ºC for 18 minutes showed the best result.
The general increase in protein solubility and protein dispersability index with processing time and the inverse relationship between urease activity and processing time agree with those presented by Navicha et al. (2017). In all cases, process temperatures and duration had effect on nutritional quality of soybean meal. From the results presented in tables 1 - 3, SBM produced by roasting had the highest crude protein composition, however, the quality of this protein was the lowest using the urease index, protein KOH solubility and protein dispersability index as a bench mark, and the trypsin inhibitor activity was also the highest. Although SBM produced from the autoclaving process had higher crude protein content with higher KOH solubility, a major demerit of the autoclave processed SBM with respect to steam processed SBM, is the higher trypsin inhibitor activity of autoclave processed SBM. The inhibitor activity of steam processed SBM is the lowest, the SBM have crude protein within the range considered normal for poultry birds and the protein KOH solubility falls within the recommended standard. With other parameters falling within the recommended range, the steam processed SBM can be considered as the best.
Conclusion
The steaming process was found to be the best of the three heat treatment processes considered. The steam treated soybean meal gave low trypsin inhibitor of 1.20 mg/g and good urease level of 0.11. This shows an adequacy of the steaming process that destroyed the trypsin inhibitor without compromising the nutrients standard to produce quality SBM for poultry feed. Also the parameters from the proximate analysis showed that the steaming process had the best overall performance.
Recommendetion
The application of the results from this study is that steam process should replace the current roasting process used in producing SBM for poultry feed in Nigeria. The oil extraction from SBM by screw press or hydraulic press should be retained for commercial production of SBM. This will remove the effect of heat treatment on SBM during solvent extraction. The cost implications for this change will not be excessive. The benefits of the change on the health of the birds far outweigh the cost implications.











 text in
text in