Stizolobium spp. (common name mucuna) is a legume native from India, China and Malaysia, places where it has great acceptance as animal and human food not only for its low production cost and adaptability to different types of climates, but for its excellent nutritive properties (Chandra et al. 2016). It is a multipurpose species, known and used as a cover crop worldwide (Castillo and Caamal 2011), it is used as a nutritional supplement for animals (Caro and Dihigo 2012) and in traditional medicine for its hypoglycemic, anti-inflammatory and aphrodisiac properties (Tavares et al. 2015).
The mucuna seeds are consumed in some regions of Africa, as an alternative food to satisfy the growing protein demand of its inhabitants and improve its nutrition due to its high content of protein and other nutrients essential for the organism (Tresina and Mohan 2013). Forages are less used for these purposes; however, they also refer formidable nutritional properties because they are a source rich in proteins, minerals and fibers. They also have a variety of secondary compounds such as flavonoids, phenolic acids, saponins and alkaloids that can have benefits for health and welfare, more than the basic diet can provide, these compounds give value added to meals as a functional food. In this context, a new relation between nutrition and health appears, which are the nutraceutical products. These can be defined as active chemical or biological components of a food or part of it that provides an added benefit to health including the prevention and treatment of diseases (Celis 2009).
The presence of phytochemical components linked to other bioactive ingredients of the plant such as fatty acids, carotenes and phytosterols are those can determine their nutraceutical properties and their significant role in the control and prevention of diseases and in the productive indicators of animals.
The objective of this study was to evaluate and compare the nutraceutical value of the forage meal of Stizolobium niveum and Stizolobium aterrimum for their use in animal feeding.
Materials and Methods
Research area. The study was carried out in September, in an area of Ayala farm, from the Instituto de Ciencia Animal, located at 22°81' north latitude and 82°01' west longitude in San José de las Lajas Mayabeque province (Academia de Ciencias de Cuba 11989). Plants of Stizolobium niveum and Stizolobium aterrimum were used, which were planted in brown soil with carbonate (Hernández et al. 2015), in a land of two hectares. No fertilization or irrigation was applied.
Meals preparation. The cut of the plants was carried out after 80 days of sowing, was manually done using knives, at 5 cm above the soil level when 100 % of the plants that were in the area were flowered (Díaz et al. 2003). The sampling was performed by the total cut of the plot and 50 cm of border effect was eliminated. Five points were randomly sampled for each of the varieties and ten plants were taken at each point, which constituted the samples for the analysis. The whole plant (leaves and stems) was cut and chopped, then dried at room temperature, in a ventilated room until approximately 20 % DM was obtained. The samples (n=5) were milled at a particle size of 1 mm and stored in amber bottles until the time of analysis.
Bromatological analysis. The dry matter (DM), crude protein (CP) and ash were determined according to AOAC (1995). The true protein was analyzed according to Bernstein (1983) cited by Meir (1986).
Mineral composition. The macro and microelements were analyzed by atomic absorption spectrophotometry according to the procedures of the data book (Atomic Absortion Data Book 1991).
The analysis was carried out on Philips equipment from the Pye Unican firm with serial number PV 9100. The height of the burner was 10mm. The composition of gases was air-acetylene and the flow was 4.5 mm/min. The determination of phosphorus was performed according to the Amaral (1972) methodology.
Fractionation of the fiber. The neutral detergent fiber (NDF), acid detergent fiber (ADF), lignin (Lig) and cellulose (Cel) were determined according to the procedure described by Van Soest et al. (1991).
Determination of the profile of long chain fatty acids (LCFA). The extraction of the meal lipids was carried out using a Soxhlet extractor. Diethyl ether was used as solvent and the extraction time was 4 hours (AOAC 1995).
The determination of the fatty acid profile was carried out in the Centro de Productos Naturales from the Centro Nacional de Investigaciones Científicas (CENIC), Havana, Cuba. The oils were analyzed as methyl esters according to method 108.003 of the Institute for Nutraceutical Advancemen.
An Agilent 7890 A gas chromatograph was used, with capillary column: BPX-70 (30 m x 0.25 di), provided with Flame Ionization Detector (FID). Initial temperature: 80°C (2 min isothermal) at 10 °C/ min up to 180°C, from 180 °C at 1°C/min up to 200 ºC, 200°C to 10°C/ min up to final temperature: 240 °C (60 min isothermal). Detector temperature equal to the injector temperature (260 °C). Flow: 0.8 mL/ min, hydrogen flow: 35 mL/min and air flow: 350 mL/min. The chromatographic profile of each sample was compared with the chromatograms of the corresponding standards to perform the identification of the LCFA.
Determination of phenolic compounds. To determine the concentration of total polyphenols (TP) and total tannins (TT) a reference solution of tannic acid (Sigma Aldrich) concentration (0.5 g/L) was used. The TP and TT analysis was carried out using the Folin-Ciocalteau reagent, before and after the treatment of the extracts with polyvinylpolypyrrolidone (Makkar 2003). The total condensed (TCT) and fiber- bound tannins (FBT) were expressed in leucocyanidin and catechin equivalents respectively, according to Makkar's (2003) methodology and the flavonoids were determined according to the procedure proposed by Kostennikova (1983), modified by Méndez (1996).
Total carotenes. The procedure for extracting the carotenes was performed in a bain- marie (60 °C) with stirring and a mixture of tetrahydrofuran-methanol (1:1) v/v was used according to the Odriozola(2009) methodology.
Total saponins. The content of total saponins was quantified by measuring in a spectrophotometer the color developed during blood hemolysis by this type of compound. The technique described by Guerra et al. (2001) was used.
Statistical analysis. Analysis of variance was carried out according to a completely randomized design. For the analysis of the results, the statistical system Infostat (Di Rienzo et al. 2012) was used.
Results and Discussion
To evaluate the nutraceutical components of the forage meal of Stizolobium niveum and Stizolobium aterrimum and its possible use as a functional food it is necessary to consider its nutritional composition (table 1).
Table 1 Nutritional composition of the forage meal of Stizolobium niveum and Stizolobium aterrimum (n=5)
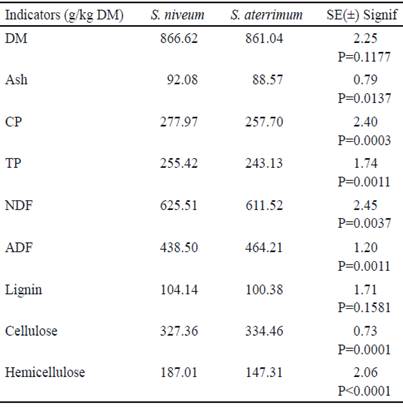
The concentration of crude protein (CP), true protein (TP) and ash (As) showed differences between the S. niveum and S. aterrimum varieties. A greater amount of ash was found in the S. niveum cultivar, this is an indicator of the proportion of inorganic compounds present in the plant. In the meals, from both plant materials were values above those found by Ujowundu et al. (2010) and Ihedioha and Okoye (2011), which may be due, among other factors, to the characteristics of the soil in which the crops were grown, the variety, management of the cultivars and the climatic conditions.
The average CP was higher for S. niveum. However, in both materials the CP was higher than 200 g/kg DM, so it can be considered as protein-rich materials, comparable with other tropical legumes from Cuba and Mexico with potential for use in animal production systems (Delgado et al. 2007 and Cab et al 2015). The high content of protein is related to the characteristic of legumes to fix atmospheric nitrogen in their radical nodules and, through metabolism, store it in its forage component (leaves, young stems and fruits) which allows to calculate the crude protein with help of a factor (generally 6.25), this content varies between 10 to 35 %.
The CP values found agree with those reported by Delgado et al. (2007) and Chikagwa-Malunga et al. (2009) when determining the bromatological composition of Mucuna pruriens forage. Ujowundu et al. (2010) and Ifemeje (2016) showed protein contents higher to those obtained in this study, while the protein levels reported (133.6 g/kg DM) by Ihedioha and Okoye (2011) are considered less and lower for this legume. These differences can be related to the variety used, phenological state of the plants, crop conditions and with variations in the environmental conditions.
As can be seen in the table, the major component of meals is insoluble fiber. The concentration of NDF, ADF and cellulose in forages showed differences between the species, being S. niveum the one that showed the highest NDF value, higher to that indicated by Fluck et al. (2013) for the foliage of three varieties of this plant. The NDF is constituted by three structural polymers fundamentally; cellulose, lignin and hemicellulose, while the ADF is composed of cellulose and lignin, hence the main difference between these fractions is the inclusion of hemicellulose in the calculation of the NDF. These indicators allow determining the amount of energy that the food contains and its quality. The obtained results are similar to those found by Savón et al. (2004), when evaluating a group of legumes and other forage tree plants. These authors report that the high fiber content in this legume can be due to complexes formed between carbohydrates and phenolic compounds. These associations may be the cause of analytical interference in the measurement of cell wall components.
In spite of the fact that fiber in foods is related to the indigestible fraction of the diet, it promotes beneficial physiological effects such as laxation, attenuation of cholesterol and blood glucose. This fiber could also be associated with phytochemical compounds or other components present in the food such as polyphenols, carotenoids and phytosterols, which can give antioxidant capacity (Saura-Calixto and Jiménez-Escrig, 2001).
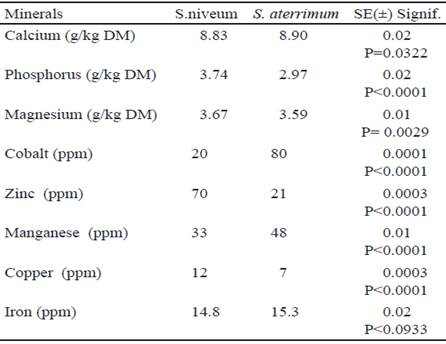
Minerals are other compounds of great nutraceutical importance since they intervene in important functions in the enzymatic metabolism and strengthen the immune system, which makes them essential nutrients for the animal organism (Li et al. (2016). The previous reaffirms the need to know its content when it is intended to use meals for this purpose. From the results of the mineral composition of the forage meal of Stizolobium niveum and Stizolobium aterrimum (table 2) it can be pointed out that both species have high amounts of minerals, a typical characteristic of tropical legumes (Román et al. 2014).
It was found that the concentrations of calcium (Ca) and of the cobalt (Co) and manganese (Mn) microelements were higher for S. aterrimum meal, which could be due to differences in the absorption of soil ions, due to variations in cell metabolism and different growth forms (Raven et al.1992). However, the results show that both species could make a significant contribution of minerals to the animals diets.
Results similar to those in this experiment were found by Chikagwa-Malunga et al. (2009) when they evaluated plants with 77 days of sowing, while the values of the Fe, Zn and Cu microelements were lower than those indicated by Ifemeje (2016) in plants grown in Nigeria. The differences could be related to the type of soil, the varieties used and the climatic conditions in which the plants were developed in each experiment.
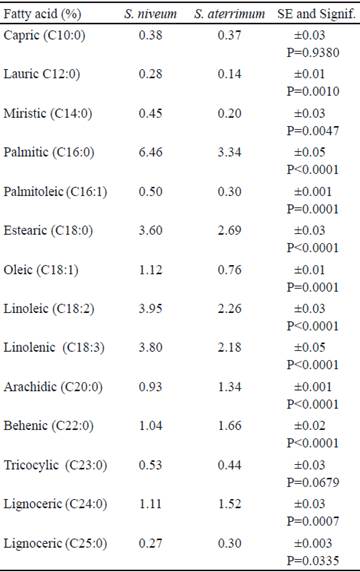
Among the compounds that can provide added benefits to the animals diets are fatty acids, which are related to the decrease of cholesterol and blood triglycerides (Valenzuela and Sanhueza 2009). In this study, differences were found in the FA composition among the forage meals under study, except for capric and tricyclic acids (table 3).
The palmitic was the most abundant FA in both species, and reached higher values for S. niveum. Other characteristic compounds in the FA profile of the forage meal were linoleic, linolenic and stearic acid, the highest values of these acids were obtained for S. niveum meal. Tavares et al. (2015) when analyzing flowers of plants of this same species found the palmitic, linoleic and linolenic acids representative in their FA composition, results similar to those of this study.
Carrillo et al. (2012) stated that linoleic acid is recommended in diets of laying hens for its effect on egg quality and its high reducing power of total cholesterol, both in eggs and in poultry meats. This acid is also recognized for its antioxidant, immunomodulatory and antimutagenic properties. These same authors pointed out that the relation between health and fat intake depends on the type of FA that predominates in the diet, that is, on the quality.
Numerous beneficial effects are attributed to these compounds, polyunsaturated fatty acids are considered of great importance in the prevention and treatment of several chronic diseases such as diabetes and cancer, while medium chain FA are responsible for the antimicrobial effect (Abuelfatah et al. 2016).
As shown in table 4, the concentrations of total polyphenols (TP), total tannins (TT), condensed tannins (CT), flavonoids (Flav), carotenoids and saponins were higher for S. aterrimum, while the values of condensed tannins join to the neutral detergent fiber (CT-NDF) and the acid detergent fiber (CT-ADF) were lower for this species.
Table 4 Content of phenolic compounds, carotenoids and saponins in the forage meal of S. niveum and S. aterrimum species
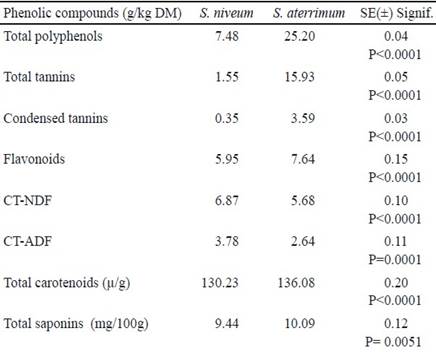
The differences in the total of phenolic compounds may be due that the synthesis and accumulation of polyphenols during the flowering period is different in both species (Chikagwa-Malunga et al. 2009). These same authors evaluated these secondary metabolites in the Mucuna pruriens with different vegetative states of the plant and found TP values between 5.85- 30.9 g/kg DM, levels similar to those obtained in this study.
The contrasts in the content of phenolic compounds may also be related to variations in the metabolism of these compounds due to genetic differences between the varieties. Valares (2011) argued the importance of these metabolites in the physiological processes of the plant that allows it to adapt to environmental change in accordance with its hereditary characteristics. In general, if a species shows variability in the amount of these compounds, it would imply a better response to environmental changes, which is favored.
The results found for the TP suggest that the meals have antioxidant potential and it is recommended to carry out other studies to evaluate this property and promote its proper use in the diet.
Ifemeye (2016), when evaluating the chemical composition of Mucuna pruriens found values for TT and flavonoids of 32.5 and 28.6 mg/g respectively, however Motta et al. (2013) reported a flavonoid concentration of 18.3 mg/g in this same species, while lower values were found in the evaluated species. These divergences in the results could be due to environmental differences. Plants vary quantitatively and qualitatively in their synthesis of tannins in response to environmental conditions. According to Santacoloma and Granados (2012), the climatic season, luminosity and humidity are the elements with the highest incidence in the synthesis of tannins. These authors when studying the concentration of tannins in the Gliricidia sepium legume cultivated in several zones of Colombia found higher content of this metabolite in high temperature zones.
The activity of some enzymes such as phenylalanine ammonia lyase (PAL) and polyphenol oxidase (PPO) in the plants may also be related to the content of these compounds, either by the induction of polyphenol synthesis or by oxidation to other compounds, events that may occur during the plant development process (Quiñones et al. 2012). These authors, in addition, show the participation of the PAL in the formation of phenylpropanoid, intermediate involved in the production of flavonoids and other phenolic compounds and involved with plant responses to stress situations.
The CT associated with fiber represents part of the non-extractable polyphenols, since they remain in the fibrous residue after extraction. This fraction of polyphenols is not usually taken into account in nutritional studies, however, it may have important biological activity and be responsible for part of the beneficial properties attributed to fiber (Kumar and Pandey 2013 and Saura-Calixto 2010).
The total carotenoids showed differences in their content in the forage meal of Stizolobium species under study, the higher values were in S. aterrimum. The synthesis and accumulation of carotenoids is genetically determined, but the composition and content depend on the environment and culture conditions. The levels found in this experiment were lower than those of Otitoju et al. (2014) on fresh and dried leaves of Mucuna pruriens.
The presence of these compounds in the diet is of great importance because the animals cannot synthesize it and some of them have provitamin A activity in the animal organism. On the other hand, carotenoids are used to improve the pigmentation of products such as chickens, meats and eggs. These compounds are considered beneficial because they provide higher immune capacity in the organism, so that it could reduce the incidence of diseases in animals (Cothran et al. 2015).
García and Medina (2006), reported that the content of saponins in plants for forage use was considered as high when the values were higher than 3.5 %. The levels of this metabolite in the meal under study were similar to the values reported by these authors in foliages of other plants that are habitually intake by cattle. However, the values in the evaluated species were higher than the results found by Ifemeje (2016) when studying the phytochemical composition in the Mucuna pruriens and finding a concentration of 3.50 mg/100g of saponins.
The different structures with which saponins are presented in plants are related to the diversity of biological activity attributed to them, which increases the use of these compounds as nutraceuticals. Ahumada et al. (2016), showed antifungal and antioxidant activity, among others, of the saponins of a quinoa extract, used to inhibit the growth of Candida albicans (fungus of medical interest), so that these compounds could be used against diseases caused by fungi and bacteria. The ability to lower cholesterol in serum is one of the most significant characteristics of saponins, however, some of these nitrogen compounds may have toxic effects on the physiology of monogastric animals (Arabski et al. 2012).
According to the results obtained in this study, the S. niveum forage meal of showed higher protein, fiber and fatty acid content, while in the S. aterrimum were the highest concentrations of saponins and carotenoids, with marked differences between total polyphenols and total tannins, an aspect in which it must be deepened because of the biological activity that these compounds can contribute to meal. The condensed tannins linked to fiber must serve as a precedent for future researches that allow corroborating whether fiber benefits from phenolic compounds.
The forage meal of these species has a high nutraceutical value when contributing to the diet chemical compounds important for nutrition and bioactive ingredients that can contribute to the control and decrease of diseases in animals, as well as improve their productivity and welfare. It is proposed to continue in vivo researches that include meals in different formulations of diets.











 text in
text in