INTRODUCTION
Heavy metal contamination of soils is wide spread due to human, agricultural and industrial activities (Beladi et al. 2011). It results in the presence of heavy metal traces in agricultural soils that lead to a threat to food safety and public health (Dary et al. 2010). This accumulation of heavy metals leads to abnormal loss of soil fertility and microbial activities (Krujatz et al. 2011). One of the stressful environmental conditions is contamination of soils in which they grow with metals such as copper (Cu), zinc (Zn), cobalt (Co) and manganese (Mn). All these metals are essential for plant and microbial growth, but at high concentrations they are toxic and adversely affect plant and microbial growth, nodulation and nitrogenase activity of plants (Ahmad et al. 2012). Copper (Cu) is not only a heavy metal but also an essential micronutrient for plant growth (Szira et al. 2014) and development (Thounaojam et al. 2012).
The influence of soil microorganisms on soil quality and plant health has recently received more attention. These microorganisms can promote plant acquisition of nutrients (Vimal et al. 2017), mineralization of organic phosphorus (Meyer et al. 2017) and production of phytohormones (Kurepin et al. 2015) decrease the negative effects of environmental stress. Plant growth promoting bacteria have been widely used to increase the capacity of host plants to tolerate and absorb heavy metals from soil (Kamran et al. 2017). Some metal resistant microorganisms could also promote plant growth under heavy metal stress conditions, thus more biomass increases the efficiency of phytoremediation.
The legume-Rhizobium symbiosis has been proposed as a promising technique for the phytoremediation of contaminated soils due to its beneficial activity in symbiotic nitrogen fixation (Jian et al. 2019). Some rhizobia have survived under heavy metal contamination. In spite of their symbiotic association their nitrogen fixing ability is lost (Broos et al. 2004). Application of heavy metal tolerant Rhizobium species with the plant provides high efficiency for phytoremediation (Dary et al. 2014). It also has the additional advantage of providing N-compounds to the soil by biological nitrogen fixation in root nodules even in metal polluted soils (Hao et al. 2014). This enhances soil fertility also.
Horse gram is an important pulse crop and it is extensively cultivated on light red and gravel soils of peninsular India. The significance of this crop is its adaptability to poor and adverse climatic conditions, which are unsuitable for other pulse crops. It is widely cultivated as a grain legume and fodder crop in the states of Tamil Nadu, Karnataka, Andhra Pradesh and Orissa of South India.
The current studies target to analyze the effect of Cu tolerant rhizobia-plant symbiosis on photosynthetic performance, symbiotic efficiency and biosorption potential of Horse gram plants upon inoculation with the chosen rhizobial strains.
MATERIALS AND METHODS
Horse gram [Macrotyloma uniflorum (Lam.) Verdc.], belongs to the species Macrotyloma uniflorum of the genus Macrotyloma in the family Fabaceae/Leguminoseae of the order Fabales in the Kingdom Plantae. Seeds of Horse gram were obtained from local fields of Andhra Pradesh, India were used for nodulation studies.
Experimental design and treatments. Inoculation with rhizobial strains grown under different concentrations of Copper (Cu): The experiment was carried out during Kharif season at the Botanical garden, Acharya Nagarjuna University, Andhra Pradesh, India. Horse gram seeds were surface sterilized with 70 % ethanol for 3 min followed by sodium hypochlorite for 3 min. Then seeds were washed in sterile water and dried. The rhizobial suspension of isolates HGR-4 (GQ483457), HGR-6 (GQ483458), HGR-13 (GQ483459) and HGR-25 (GQ483460) were grown in Yeast Extract Mannitol (YEM) broth in flasks shaken at 120 rpm at 28±2ºC for 3 days (cell density of 6×109 cells ml-2). These suspensions were inoculated to Horse gram plants. The inoculation was performed on sterilized seeds were coated with the rhizobial strains (by soaking the seeds in liquid culture medium for 2 h using 10 % (wt/vol) gum Arabic as adhesive to deliver approximately 109 cells seed-1). The inoculated seeds (20 seeds pot-1) were sown in clay pots using 2 kg sterilized soil (autoclaved at 121ºC for 3 h for three successive days). Controls were maintained with seeds treated in sterilized distilled water. Cu tolerance of Horse gram plants without rhizobial inoculation was also analyzed with various Cu supplements i.e. 50, 100, 300 and 500 µg g-1 of kg-1 in unsterilized soil. Triplicates were maintained for each treatment. The pots were watered when required and were maintain separately in open field conditions and allowed to grow.
Analysis of plants for Photosynthetic (PS) performance and symbiotic efficiency: After 40 d treatment, the Net photosynthetic rate (PN) was determined with a Portable Photosynthetic System ADC Bioscientific, U.K. The measurements were made at ambient CO2 concentrtions between 09.00 and 10.00 a.m. on a clear sky day. These plants were observed for nodulation regularly after the seedlings came out. Five plants in each treatment were picked up randomly and nodulation characteristics viz., number, size, shape, colour, distribution of the nodules were noted after 40 days of sowing, as it was previously observed that highest nodulation of Horse gram occurred at 40 days.
Biochemical analysis: For biochemical analysis, plants raised in different concentrations of Cu were collected, leghaemoglobin content was estimated after 40 (d) of sowing (Tu et al. 1970). Soil pH, organic matter, total nitrogen (N) (Jackson 1973) and total phosphorus (P) (Olsen et al. 1954) were estimated. The amount of sand, silt and clay present in the soil were also analyzed (Black 1965). Cu concentration present in control as well as in inoculated soil and nodule samples were determined by ICP-AAS (THERMO AAS Model No: ICE 3000).
Statistical Analysis: Statistical analysis was done in three replicates for each treatment. The mean and standard error (SE) were calculated using Microsoft Office Excel 2007. To know the statistical significance all the values were analyzed by ANOVA, using IBM SPSS Statistics, Version 20 (Armonk 2011). Bars indicate means of ± SE and were significant at 5% level of significance (LOS). P value < 0.05.
RESULTS AND DISCUSSION
Horse gram plants have shown significant changes in their net photosynthetic rate (PN) upon inoculation with Rhizobium strains grown under different concentrations of Cu. Plants exposed to Cu have shown a decline in PN (Ahmad and Naim 2008). At 20 µM concentration Cu decreased the PN of about 55 % over the control (Burzynski and Zurek 2007). Horse gram plants inoculated with the strain HGR-6 have shown their maximum PN [10.44 µmol (CO2) m-2s-1] at 100 µg g-1. HGR-25 inoculated plants also have shown maximum PN values at this concentration. But, the values were low when compared to HGR-6 (figure 1). The plants inoculated with the strain HGR-4 have enhanced PN values with increase in Cu concentration up to 500 µg g-1. CuCl2 at low concentrations (0.1 mM) enhanced maximum CO2 assimilation rate in sweet sorghum (Poor et al. 2015). The plants inoculated with the strain HGR-13 have shown maximum PN only at 50 µg g-1. PN was significantly inhibited (100 %) and most similarly affected by two concentrations of Cu2+ i.e. 0.062 and 0.33 M (González-Mendoza et al. 2013). Cu has shown strong negative effect on PN in barley plants. At 20 mg Cu kg-1 it was decreased to 56-57 % when compared to control plants (Vassilev et al. 2003). That effect is found to be low below 20 %. Cu initially destroys the photosynthetic carbon reduction cycle and subsequently influences the photosynthetic electron transport (Burzynski and Klobus 2004). Plants kept (96 h) in Hoagland nutrient solution without Cu, in full Hoagland solution (0.5 µM Cu) and in Hoagland medium with Cu from 1 to 100 µM. The absence and the presence of Cu at above to 1 µM inhibited PN (Costa et al. 2018). PN declined 24, 77 and 89 % at 0.1, 1 and 3 m mol L-1 Cu treatment when compared to control (Nisha et al. 2016). It clearly shows that Cu enhanced PN values of Horse gram plants with prior inoculation of rhizobia.
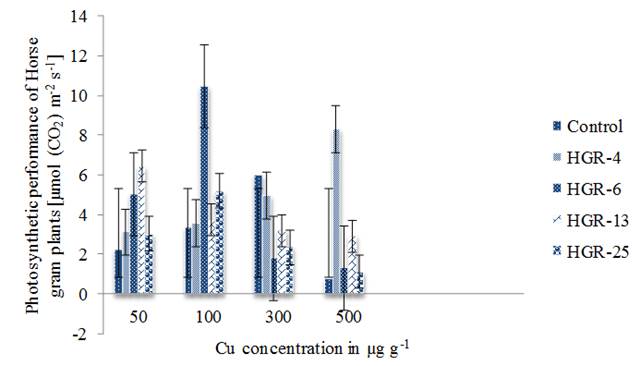
Figure 1 Copper effect on photosynthetic performance of Horse gram plants inoculated with four Rhizobium stains. Bars indicate mean ± SE. All the values were analyzed by ANOVA using IBM SPSS Statistics Version 20 and were significant at 5% level of significance (LOS). P value < 0.05.
Horse gram rhizobial strains have shown their ability to form effective nodules at all the metal concentrations tested. In our study, nodules were observed after 13 days of sowing and were formed both on tap root and as well as on lateral roots. The total number of nodules formed per plant ranged from 12 to 18 (figure 2). The plants inoculated with the strain HGR-6 inoculated plants have shown maximum nodulation at 300 µg g-1. But, the strains HGR-4, 13 and 25 have shown at 100 µg g-1. After that the number of nodules was decreased with increasing Cu concentration. The increasing concentrations of Cu in N-free solution or potted soil reduced fresh nodule mass and volume (Sánchez-Pardo et al. 2012). Adverse effect of excessive Cu, on symbiotic biological nitrogen fixation (BNF) have shown inhibitory growth effect on the microsymbiont (Stan et al. 2011) nodule growth, development and function (Sanchez-Pardo et al. 2012). Cu at 192 µM in N-free nutrient solution reduced nodule weight along with nodule number in Lupines albus L. and Glycine max L. (Sánchez-Pardo et al. 2012). Heavy metals at high concentration reduce the efficiency of symbiotic nodulation with Rhizobium and also inhibit plant growth (Jian et al. 2019).

Figure 2 Nodulation of Horse gram plants inoculated with Rhizobium strains in response to Copper. Bars indicate mean ± SE. All the values were analyzed by ANOVA using IBM SPSS Statistics Version 20 and were significant at 5% level of significance (LOS). P value < 0.05.
There are some variations in the ability to tolerate Cu toxicity among rhizobia-legume associations. The rhizobia-Acacia auriculaeformis association has high tolerance to Cu2+ than the Rhizobium-Lespedeza formosa association (Nie et al. 2002). Excess Cu reduced the number of functional nodules and the infected cells in nodules (Hao et al. 2015). Inoculation of green gram with Bradyrhizobium sp. under Cu stress decreased the number of nodules by 23 % at 1338 mg kg-1 soil, when compared with control (Wani et al. 2007). High levels of Cu reduced nodule number and weight in Vigna unguiculata (Kopittke et al. 2007). The number of nodules formed were high at low concentration of Cu (50 mg kg-1) after that, they were decreased with increasing Cu concentration (Manivasagaperumal et al. 2011). The reduction in nodule number under Cu stress was also observed in Lupin, soy bean and Vigna (Sanchez-Pardo et al. 2012). Pot experiments have shown that 400 mg kg-1 Cu2+ greatly inhibited plant growth of Medicago lupulina but dual inoculation of Sinorhizobium meliloti CCNWSX0020 and Agrobacterium tumefaciens CCNWGS0286 significantly increased the number of nodules (Jian et al. 2019).
The amount of leghaemoglobin was maximum in Horse gram plants inoculated with the strains HGR-4, 6 and 25 at 300 µg g-1 of Cu (figure 3). But, the plants inoculated with the strain HGR-13 have shown at 500 µg g-1. Cu at 192 μM concentration, in N-free nutrient solution reduced total plant nitrogen in Lupinus albus L. and Glycine max L. (Sanchez-Pardo et al. 2012). Cu causes reduction in protein content of nodule cytosol, bacteroid fractions and leghaemoglobin concentration of nodule cytosol (Younis 2007). Cu toxicity to plant on the production of leghaemoglobin was independent of inoculated rhizobia (Hao et al. 2014). A significant reduction in total shoot nitrogen was observed when Cu was increased from 0 to 100 mg Cu L-1 in vitro and in vivo. In the potted soil based studies, increasing Cu concentration from 0-100 mg g-1 resulted in drastic decrease in total shoot nitrogen of 39.2 and 36.2 % for GM3 and CP1 compared to 17.2 and 18.2 for PV1 and PV2 respectively (Hamisi Tindwa et al. 2014). The reduction in nitrogen fixation under Cu stress is due to its accumulation on the inner cortex and infected zones of the nodule there by inducing abnormalities in the structure and ultrastructure of the nodules (Sanchez-Pardo et al. 2012). Cu tolerant S. meliloti CCNWSX0020 can establish a normal symbiotic relationship with the host plant under Cu stress and the nitrogenase activity indicated that it has formed effective nodules under Cu stress (Jian et al. 2019).
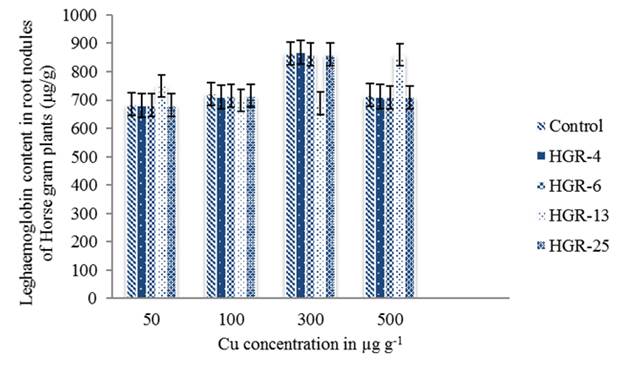
Figure 3 Leghaemoglobin content in root nodules of Horse gram plants inoculated with Rhizobium strains in response to Copper. Bars indicate mean ± SE. All the values were analyzed by ANOVA using IBM SPSS Statistics Version 20 and were significant at 5% level of significance (LOS). P value < 0.05.
The production of leghaemoglobin was quite sensitive to Cu stress in nodules generated by both wild-type and mutant strains. Nitrogen fixation in white clover was halved at 428 mg Cu kg-1 (Broos et al. 2004) but, these concentrations were above the normal value in Woburn for total Cu (99 mg kg-1). The inoculated Medicago lupulina plants grown in the presence of excess Cu have shown a slight increase in N content in both shoots and roots (Kong et al. 2015). The nitrogen content in shoots could be considered as the supply of N through N-fixation in root nodules. Cu-resistant strain S. meliloti CCNWSX0020 is able to survive under the Cu concentration used and promotes a normal level of plant nitrogen (Kong et al. 2015). A significant increase in nitrogen content in plants was detected after co-inoculation of S. meliloti and Agrobacterium tumefaciences which significantly increased the N content of the above ground portion by 19% compared to that of control in the presence of 400 mg kg-1 Cu2+ (Jian et al. 2019). The amount of total nitrogen (%) and phosphorus (%) present in the soil are 0.85 and 1.24 respectively. The total content of organic matter in soil is 1.20, sand 18, silt 16, clay 42 and the pH of the soil is 6.44.
In addition to nitrogen fixation, metal-resistant rhizobia demonstrate the production of plant growth-regulating substances or effects on metal solubility and bioavailability which affects plant metal uptake (Pajuelo et al. 2011). Bacterial biosorption/bioaccumulation mechanism together with other plant growth promoting features accounted for improved plant growth in metal contaminated soils (Zaidi et al. 2006). In the present study, biosorption potential of the strain HGR-6 was determined by Atomic Absorption Spectroscopy (AAS), as the amount of metal present in the medium after the treatment with the isolate. The results have shown that the isolate was able to absorb Cu at a concentration of 500 µg g-1 in root nodules and also in soil samples inoculated with the strain HGR-6 (figure 4 and 5). Therefore, the use of microbes for reduction/detoxification of heavy metals is one of the preferred choices and is considered a cost-effective approach in remediation technologies.
These bacteria can also protect the plants from the toxic effects of metals through an adsorption/desorption mechanism. The decrease in metal concentration after rhizobial inoculation suggested the role of strain RP5 in the removal of these metals through adsorption-desorption mechanism (Mammaril et al. 1997). The dead cells of Mesorhizobium amorphae CCNWGS0123 are an effective absorbent for removal of Cu from aqueous solution. The removal of Cu2+ increased significantly by decreasing the pH up to 5.0 (Mohamad et al. 2012).
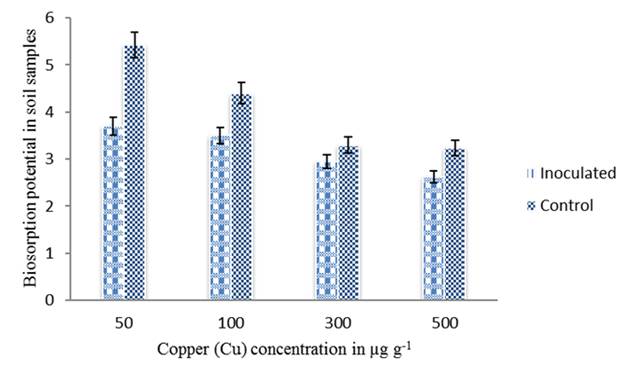
Figure 4 Metal (Cu) biosorption by the strain HGR-6 in soil determined by AAS. Bars indicate mean of ± SE. All the values were analyzed by ANOVA using IBM SPSS Statistics Version 20 and were significant at 5% level of significance (LOS). P value < 0.05.
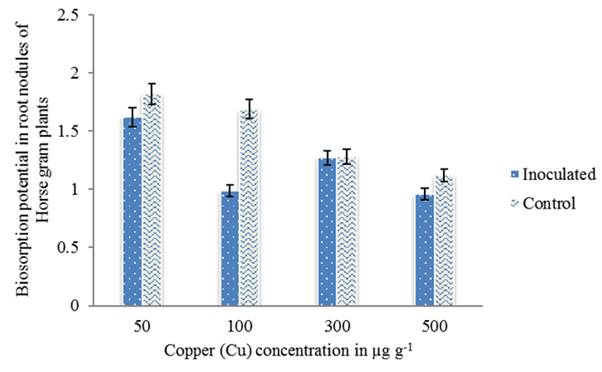
Figure 5 Metal (Cu) biosorption in root nodules of Horse gram plants inoculated with the strain HGR-6 determined by AAS. Bars indicate mean of ± SE. All the values were analyzed by ANOVA using IBM SPSS Statistics Version 20 and were significant at 5% level of significance (LOS). P value < 0.05.
This study demonstrates that the Cu tolerant rhizobia i.e. HGR-4, 6, 13 and 25 enhanced the photosynthetic performance, symbiotic efficiency and biosorption potential in the rhizobia inoculated Horse gram plants. Hence, Horse gram plants associated with rhizobia could be used in the phytoremediation of metal (Cu) contaminated soils.











 text in
text in 


